Which Element Has The Following Ground State Electron Configuration

Okay, picture this: it’s a sleepy Tuesday afternoon, and I’m nursing a lukewarm cup of tea, staring blankly at a particularly gnarly chemistry problem set. My brain feels like it’s been through a tumble dryer on high heat, and the words are all starting to blur. Suddenly, my cat, Archimedes (yes, that Archimedes, he’s very intellectual for a creature that spends 18 hours a day napping), saunters over and decides my textbook is the perfect new napping spot. He’s meticulously arranged himself right over a diagram of electron shells, his fluffy tail flicking right over the n=2 level. And then it hit me, not with a lightning bolt of genius, but more like a gentle nudge from a furry, purring companion: the arrangement of electrons. It’s not just random scribbles on a page, is it? It’s the identity of an element!
Seriously, think about it. Every single element on that shiny Periodic Table, from the simplest hydrogen to the most ridiculously named superheavy element, has its own unique fingerprint. And that fingerprint, my friends, is its ground state electron configuration. It’s like their cosmic social security number, but way cooler and much more fundamental.
So, the next time you’re wrestling with a problem that asks you to identify an element based on its electron arrangement, don’t despair. Just channel your inner Archimedes (or your favorite nerdy scientist) and let’s dive into this fascinating world. Today, we’re going on a little adventure to figure out: Which Element Has The Following Ground State Electron Configuration?
The Electron Orchestra: A Symphony of Energy Levels
Before we start naming names, let’s quickly recap what we’re even talking about. Electrons, those tiny, zippy things that orbit the nucleus of an atom, aren’t just milling about willy-nilly. Oh no. They’re organized. They hang out in specific energy levels, often called shells, and within those shells, they have even more specific neighborhoods called orbitals. Think of it like a ridiculously elaborate apartment building, where electrons are the tenants. The higher the floor (energy level), the more energy the electrons have.
We’ve got the principal energy levels, numbered 1, 2, 3, and so on. Level 1 is the coziest, closest to the nucleus, and has the least energy. Level 2 is a bit bigger, a bit further out, and has more energy. And it just keeps going. Inside these levels, we have different types of orbitals: s, p, d, and f. Each type of orbital can hold a maximum of two electrons, and they have specific shapes, which is a whole other can of worms we won’t open today unless you promise to buy me more tea.
The s orbitals are spherical, like tiny little tennis balls. The p orbitals are dumbbell-shaped, and they come in sets of three, oriented along the x, y, and z axes. The d orbitals get a bit fancier with cloverleaf shapes, and the f orbitals are… well, let’s just say they’re complicated. The key takeaway here is that electrons fill these energy levels and orbitals in a specific order, based on their energy. It’s called the Aufbau principle, which basically means “building up” in German. They fill the lowest energy levels and orbitals first, like filling up your grocery cart with the cheapest, most essential items before splurging on the fancy stuff.
Decoding the Notation: More Than Just Letters and Numbers
So, when you see a ground state electron configuration like, say, 1s² 2s² 2p⁶ 3s¹, what does that actually tell you? Let’s break it down, piece by piece. It’s like a secret code, and once you crack it, you’re golden.
The number at the beginning of each term (like the ‘1’ in 1s², the ‘2’ in 2s²) tells you the principal energy level. So, ‘1’ means the first shell, ‘2’ means the second shell, and so on. Easy peasy, right?
The letter (like ‘s’, ‘p’, ‘d’, ‘f’) tells you the type of orbital. Remember those s, p, d, f neighborhoods? This is where they live.
And the superscript number (like the ‘²’ in 1s²) tells you how many electrons are in that specific orbital. So, 1s² means there are 2 electrons in the 1s orbital. 2s² means 2 electrons in the 2s orbital. And 2p⁶ means 6 electrons in the 2p orbitals. Since a p subshell has three orbitals (px, py, pz), and each can hold 2 electrons, 6 electrons is the maximum capacity for a p subshell.
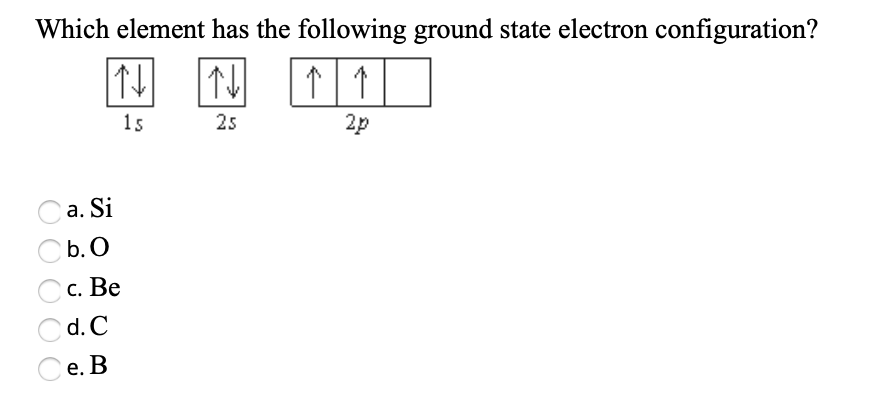
So, when you add up all those superscript numbers – the electrons in each part of the configuration – you get the total number of electrons in the atom. And here’s the kicker: in a neutral atom, the number of electrons is exactly equal to the number of protons in the nucleus. And the number of protons? That’s the atomic number! Bingo!
The Grand Total: Counting Those Electrons
Let’s take our example, 1s² 2s² 2p⁶ 3s¹. To find the total number of electrons, we just add up the superscripts:
2 (from 1s²) + 2 (from 2s²) + 6 (from 2p⁶) + 1 (from 3s¹) = 11 electrons.
Since we’re talking about the ground state electron configuration, we’re assuming a neutral atom. So, this element has 11 electrons. And because it’s neutral, it must also have 11 protons.
Now, pull out your trusty Periodic Table (or if you’re like me, squint at a blurry photo of one on your phone). Find the element with atomic number 11. Go on, I’ll wait. (Go get it. Seriously. It’s part of the fun!)
Did you find it? That’s right! Element number 11 is Sodium (Na)!
See? It’s like solving a tiny, cosmic puzzle. The electron configuration is the map, and the Periodic Table is the treasure chest.
But Wait, There’s More! The Nuances of Filling
Now, you might be thinking, “Okay, that was pretty straightforward. But what if the configuration is more complicated?” And you’d be absolutely right to think that! Chemistry, bless its heart, always has a few extra twists and turns. There are rules governing how those electrons fill up those orbitals. It’s not just a free-for-all.
We’ve already touched on the Aufbau principle. Then there’s Hund’s Rule, which states that electrons will individually occupy each orbital within a subshell before doubling up. Think of it like people on a bus: everyone gets their own seat before they have to sit next to someone. This is why you’ll see configurations where the p orbitals are filled like: one electron in each of the three p orbitals before any of them get a second electron.
And then there’s the Pauli Exclusion Principle, which essentially says that no two electrons in an atom can have the same set of four quantum numbers. In simpler terms, if two electrons are in the same orbital, they have to have opposite spins. So, if one electron is spinning “up,” the other in the same orbital has to be spinning “down.” It's like they're giving each other a high-five, but in opposite directions.
These rules are crucial because they determine the exact electron configuration. Without them, we might have multiple possibilities, and that would be utter chaos. Imagine trying to identify an element if its electron configuration was ambiguous. The whole system would collapse! (Okay, maybe not collapse, but it would be a lot harder.)
Let’s Try Another One: Building Confidence
To solidify this, let’s try another example. What element has the ground state electron configuration 1s² 2s² 2p⁶ 3s² 3p⁴?
First, let’s count the electrons:
2 (from 1s²) + 2 (from 2s²) + 6 (from 2p⁶) + 2 (from 3s²) + 4 (from 3p⁴) = 16 electrons.

In a neutral atom, this means 16 protons. So, we’re looking for the element with atomic number 16 on the Periodic Table.
Go ahead, take a look. Element 16 is Sulfur (S)! Pretty neat, huh?
Notice how the configuration fills up the energy levels sequentially. We’ve got the first energy level (n=1) completely filled with 1s². Then the second energy level (n=2) is completely filled with 2s² and 2p⁶. And then we start filling the third energy level (n=3) with 3s² and then move into the 3p orbitals, where we have 4 electrons.
The Role of Noble Gas Shorthand
Now, sometimes, you’ll see electron configurations written in a shorter, more elegant way, using noble gas shorthand. This is super handy when you’re dealing with elements that have a lot of electrons. Why write out every single orbital if it’s just the same as the previous element’s configuration?
The idea is to start with the electron configuration of the preceding noble gas and then add the remaining electrons. Noble gases (Group 18) are special because they have completely filled outer electron shells, making them very stable and unreactive. Their electron configurations are like the end of a chapter, a nice, complete set.
For example, the element Sodium (Na), which we identified earlier with the configuration 1s² 2s² 2p⁶ 3s¹, has 11 electrons. The noble gas before it is Neon (Ne), which has the electron configuration 1s² 2s² 2p⁶. See that overlap? So, we can write Sodium’s configuration as [Ne] 3s¹.
This tells us that the first 10 electrons of Sodium are arranged exactly like Neon’s, and then there’s one extra electron in the 3s orbital. It’s a time-saver and makes the configurations much easier to read, especially for larger atoms. It’s like saying, “Everything up to here is the same as [Noble Gas Name], and then this is what’s different.”

Let’s take Sulfur (S) again. Its full configuration is 1s² 2s² 2p⁶ 3s² 3p⁴. The preceding noble gas is Neon (Ne), with 1s² 2s² 2p⁶. So, Sulfur’s shorthand configuration is [Ne] 3s² 3p⁴. Much cleaner, right?
The Importance of Ground State
We’ve been talking about ground state electron configuration. What does that mean, exactly? It simply means the electrons are in their lowest possible energy levels and orbitals. Atoms, like us, prefer to be as relaxed and energy-efficient as possible. They don’t jump to higher energy levels unless they’re forced to (like when they absorb energy from light or heat).
When an atom does absorb energy, an electron can get excited and jump to a higher energy level. This is called an excited state configuration. But once the energy source is removed, the electron will quickly fall back down to its ground state, releasing the energy it absorbed. This is why things glow or emit light!
So, when a question asks for the ground state electron configuration, it’s specifically asking for the stable, unexcited arrangement of electrons. This is the configuration you’ll find on the Periodic Table, and it’s the one that defines the element’s fundamental properties.
Putting It All Together: Your Elemental Detective Kit
So, to recap your elemental detective kit:
- Count the electrons: Add up all the superscripts in the given electron configuration.
- Assume neutrality: For ground state configurations of unknown elements, assume it's a neutral atom. This means the number of electrons equals the number of protons.
- Find the atomic number: The number of protons is the atomic number.
- Consult the Periodic Table: Locate the element with that atomic number. Voilà! You've identified your element.
- If shorthand is used: Recognize the noble gas part and add the remaining electrons to determine the total.
It's a systematic process, and once you get the hang of it, you can practically read an element's identity just by looking at its electron arrangement. It’s like having a secret superpower in chemistry class!
Remember Archimedes, my cat? He might not understand quantum mechanics, but in his own way, he’s a master of finding the most stable, lowest-energy spot to be. And that, my friends, is the essence of the ground state electron configuration. It's nature's way of finding the most comfortable, stable arrangement for its tiniest building blocks. So next time you see a seemingly random string of numbers and letters, remember: it's a story, a fingerprint, and a direct clue to the identity of an element. Happy sleuthing!
