Which Element Has The Fewest Valence Electrons
Ever wondered if there's a secret champion in the world of elements, a tiny titan with just a handful of electron buddies? Well, get ready to dive into the fascinating, and surprisingly fun, world of valence electrons! Think of them as the outermost social butterflies of an atom, the ones that get to mingle and make connections with other atoms. Understanding these little guys is like unlocking a secret code that explains why things react the way they do, why water is wet, and why your phone battery eventually dies (though that's a bit more complex!). It's a core concept in chemistry that’s not just for scientists in lab coats; it’s a fundamental piece of how the entire universe is put together. And today, we're going to zoom in on a special group: the elements that have the absolute fewest of these crucial valence electrons. It's like finding the minimalist superstars of the atomic world!
So, why should you care about elements with minimal valence electrons? Well, these elements are the foundation of so much around us. They dictate how elements bond together to form everything we see, from the air we breathe to the clothes we wear. For instance, elements with very few valence electrons are often super stable, meaning they're perfectly happy on their own and don't tend to react much. This makes them incredibly useful in applications where you need something that won't easily break down or change. Think of the protective coatings on your eyeglasses or the inert gases used in lighting. They're the quiet achievers, the dependable workhorses of the chemical world.
The Reigning Champions of "Less is More"
When we talk about having the fewest valence electrons, we're often looking at the very beginning of the periodic table, specifically, elements in Group 1, also known as the alkali metals, and the noble gases, though their situation is a little different. Let's start with the undisputed kings of having a minimal, yet incredibly reactive, set of outer electrons: the alkali metals. These are elements like Lithium (Li), Sodium (Na), Potassium (K), and so on, all the way down to Francium (Fr).
Imagine an atom is like a house, and electrons are people living in it. Valence electrons are the people who live on the top floor, the ones who can easily step out onto the porch and interact with neighbors. Elements with very few valence electrons have a very small, exclusive top floor – perhaps just one or two residents!
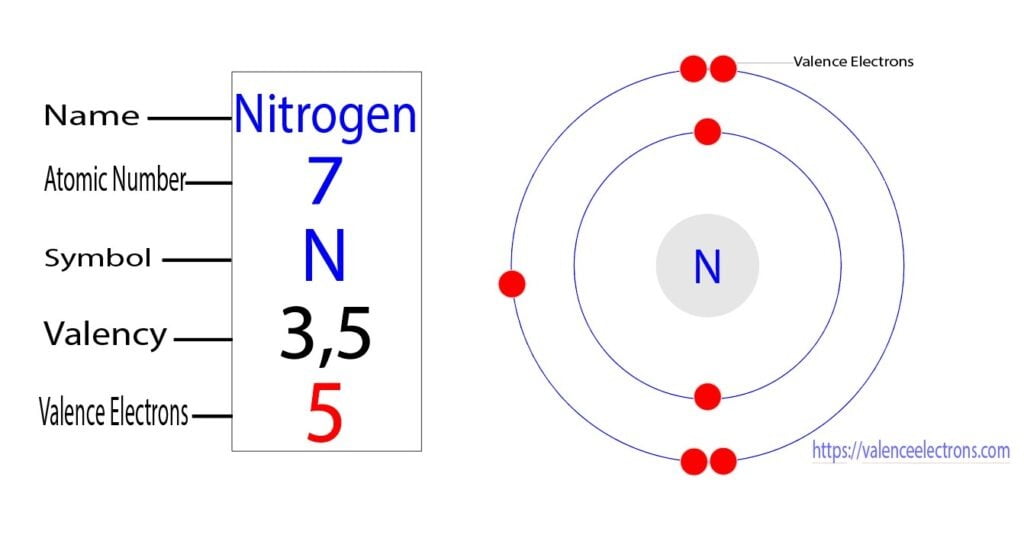
Valence Electrons - Chemistry Steps
These alkali metals famously possess just one single valence electron. That's it! One lonely electron out there, just itching to make a friend. This single valence electron is held very loosely by the atom's nucleus. It's like a child with one favorite toy they're eager to share. Because this electron is so easy to lose, alkali metals are incredibly reactive. They desperately want to give away that one extra electron to achieve a more stable, "full" outer shell, even if that means becoming a positively charged ion. This eagerness to react is why sodium (Na), a component of table salt, would cause a dramatic explosion if you tried to plop a chunk of pure sodium into water – it's reacting with the water molecules to get rid of that single, precious valence electron!
But what about the other end of the spectrum? We also have elements that are characterized by having a full outer electron shell, which makes them incredibly stable and, therefore, they have very few reactive valence electrons. These are the noble gases, found in Group 18 of the periodic table. Elements like Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and Radon (Rn). Now, Helium is a bit of a special case. Its outermost shell, which is its only shell, is completely full with 2 electrons. For all the other noble gases, their outermost shell is complete with 8 electrons. This full shell is the atomic equivalent of being perfectly content and self-sufficient. They have no need to gain, lose, or share electrons because their electron configuration is already as stable as can be. So, in a way, they have very few available valence electrons for bonding. They're the introverts of the periodic table, happy in their own company!
So, when we're asking "Which element has the fewest valence electrons?", we're really looking at two fascinating groups with different reasons for their low electron count. The alkali metals have the fewest in terms of being available to lose (just one!), making them highly reactive. The noble gases, on the other hand, have a full and stable outer shell, meaning they have effectively zero valence electrons to share or transfer, making them incredibly unreactive. Both scenarios, in their own way, showcase the elegant simplicity and powerful functionality that comes with having a minimal number of these crucial outer electrons.
Understanding this concept is a gateway to appreciating the intricate dance of atoms that forms our reality. It's a peek behind the curtain of chemical reactions and a reminder that sometimes, having less can lead to incredibly powerful and stable outcomes. So next time you see something made of metal or filled with glowing gas, remember the humble, yet mighty, elements with the fewest valence electrons – they're the silent architects of our world!

