Which Element Does The Electron Configuration 1s22s22p2 Represent

Ever wondered what makes up the world around you? It's all about tiny building blocks called elements, and figuring them out can be a surprisingly fun and useful adventure! Think of it like solving a cosmic puzzle, and today we're going to tackle one specific piece: Which element does the electron configuration 1s²2s²2p² represent? This might sound a bit technical, but stick with us – it's more accessible and intriguing than you might imagine, making it a great topic for budding scientists, curious families, or anyone who enjoys a good mental challenge.
For beginners, understanding electron configurations is like learning the ABCs of chemistry. It's the fundamental way we describe how electrons are arranged within an atom, and this arrangement dictates the element's behavior. Knowing this can unlock a deeper appreciation for everyday objects. For families, it's a fantastic way to spark curiosity in children. Imagine explaining that the air you breathe and the materials you touch all have these intricate electron blueprints! For hobbyists, whether you're into gardening, crafting, or even brewing, a basic grasp of elements can lead to fascinating insights into how things work and interact.
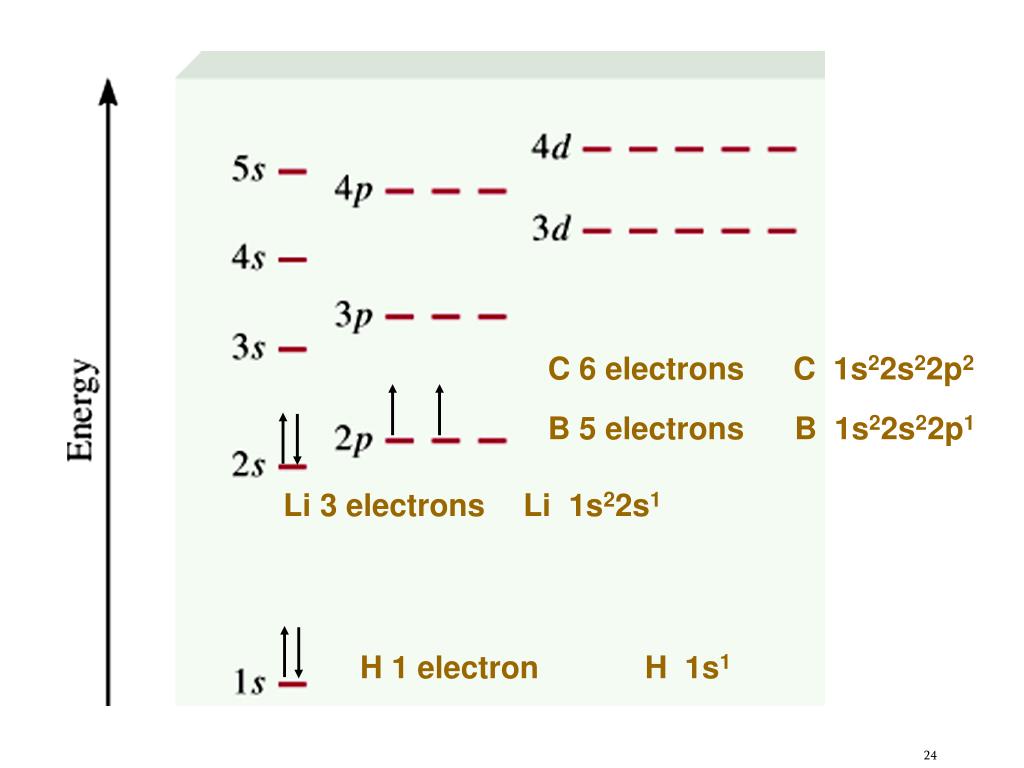
Let's break down our mystery configuration: 1s²2s²2p². The numbers and letters tell us about the "shells" and "subshells" where electrons hang out. The first number (1, 2) indicates the energy level or shell. The letters (s, p) denote the type of subshell, and the superscripts (², ², ²) tell us how many electrons are in each. So, we have 2 electrons in the first shell's 's' subshell, 2 electrons in the second shell's 's' subshell, and 2 electrons in the second shell's 'p' subshell.
To find our element, we simply add up the total number of electrons. In this case, 2 + 2 + 2 = 6 electrons. In a neutral atom, the number of electrons equals the number of protons, which is also known as the atomic number. So, we're looking for the element with atomic number 6.

Drumroll, please… the element represented by 1s²2s²2p² is Carbon! Yes, the very element that forms the backbone of all life on Earth, from the diamonds in jewelry to the graphite in your pencil. Pretty cool, right?
Variations of this topic are everywhere! For example, the electron configuration for Oxygen is 1s²2s²2p⁴, meaning it has 8 electrons (and thus an atomic number of 8). Helium, a simple but fascinating element, has the configuration 1s², showing just 2 electrons.
Getting started is easier than you think. Grab a periodic table – it’s your best friend! You can find a free one online or at a local bookstore. Look for the atomic numbers. Then, you can start practicing by identifying elements based on their number of electrons. For a fun family activity, try drawing simple Bohr models (even though electron configurations are more accurate, Bohr models are visually simpler) for the first few elements and associating them with their electron configurations.
Exploring electron configurations, even at this basic level, offers a wonderfully accessible glimpse into the intricate world of atoms. It's a journey that connects the abstract world of quantum mechanics to the tangible reality we experience every day. So, the next time you see something made of carbon, you'll know a little bit more about its fundamental makeup – and that’s a truly rewarding feeling!
