Which Diagram Shows Electrons Violating The Pauli

Hey there, science explorers! Ever wonder what all those tiny, zippy things called electrons are up to? They’re basically the lifeblood of everything – how your phone works, why the sun shines, and even why you have a favorite comfy chair. Today, we're going to chat about a pretty cool rule these electrons have to follow, and what happens when they don't. Think of it as the ultimate "no cutting in line" policy for the atomic world!
So, the big question is: Which diagram shows electrons violating the Pauli Exclusion Principle? Now, that might sound a bit like a secret handshake from a sci-fi movie, but it's actually a fundamental idea that keeps our universe humming along nicely. Let’s break it down, no fancy lab coats required.
Imagine a Super-Popular Coffee Shop
Let’s picture an atom like a really popular coffee shop. This coffee shop has different seating areas, right? Some are right by the window with a great view, others are a bit cozier in the corner. In the atomic world, these seating areas are called "orbitals." They’re like little neighborhoods where electrons like to hang out.
Now, each of these orbitals can hold a maximum of two electrons. Think of them as tiny little booths that can fit a couple of friends. But here’s the super important part, the rulebook for our electron coffee shop: each of these two electrons in a booth has to be a little bit different from the other.
How are they different? Well, electrons have this property called "spin." You can imagine it like they’re spinning in two different directions – clockwise or counterclockwise. So, in our coffee shop booth, one electron might be spinning "up," and the other might be spinning "down." They’re sharing the same space (the orbital), but they have this one key difference. This, my friends, is the essence of the Pauli Exclusion Principle.

What If the Rule Gets Broken?
Now, what if, in our imaginary coffee shop, we tried to cram three people into a booth meant for two? Or, even worse, what if we tried to squeeze two people into that booth, but they were both spinning the same way, like two identical twins trying to wear the exact same outfit? That’s where things get a little… chaotic. And that’s exactly what happens when electrons break the Pauli Exclusion Principle.
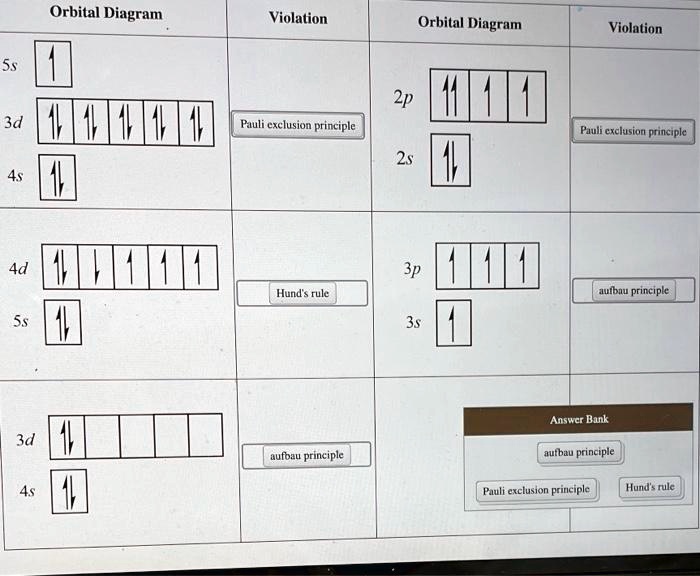
So, when you see a diagram showing electrons, you're looking for a violation of this rule. A diagram showing electrons violating the Pauli Exclusion Principle would be one where:
- More than two electrons are trying to squeeze into the same orbital. This is like trying to fit your entire extended family into a single booth. Impossible and messy!
- Two electrons are in the same orbital, but they have the exact same spin. This is like having two identical twins in the booth, both wearing the same "spin up" shirt. They’re identical in every way, and that’s not allowed.
Why Should We Even Care About This Electron Rule?
You might be thinking, "Okay, so electrons follow a rule. Big deal. How does that affect my Tuesday?" Well, it affects everything about your Tuesday, and your entire life! This seemingly simple rule is the bedrock of chemistry. It dictates how atoms bond together to form molecules.
Think about it: If electrons could just pile into any orbital they wanted, with any spin they liked, atoms wouldn't have their distinct properties. Water wouldn't be H2O. The metal in your cutlery wouldn't be shiny. The pigment in your favorite colorful shirt wouldn't exist.
The Pauli Exclusion Principle is what gives the periodic table its structure! Each element’s unique behavior is a direct consequence of how its electrons fill these orbitals according to this rule. It's the reason why some elements are noble gases and don't like to react, while others are super eager to form bonds.

A Little Story About Our Friend, Nitrogen
Let’s take Nitrogen, for instance. It has seven electrons. When we arrange these electrons in their orbitals, following the Pauli rule, we get a specific configuration. This configuration makes Nitrogen behave in a certain way, making it a key component of the air we breathe and essential for life.
Now, imagine if Nitrogen's electrons could just ignore the Pauli principle. Maybe two electrons would try to share the same spin in the same orbital. Suddenly, Nitrogen might not be the stable gas we know. It could be some unpredictable, reactive blob! Not so good for breathing, right?
It’s like having a carefully organized library. Each shelf has a certain number of books, and each book has a unique place. If you just started shoving books anywhere, with no order, you wouldn’t be able to find anything! The library (our universe) would be in utter chaos. The Pauli Exclusion Principle is the organizational principle that keeps the atomic library tidy and functional.

Spotting the "No-No" Diagram
So, when you look at diagrams of electron configurations, keep an eye out for these "no-no" situations. You'll usually see them represented by arrows in boxes. Each box is an orbital, and each arrow is an electron. An arrow pointing up and an arrow pointing down in the same box? Perfectly fine. Two arrows pointing up (or two pointing down) in the same box? Violation! Or a box with three or more arrows? Double violation!
These diagrams are like little snapshots of atomic behavior. And understanding which ones are "wrong" according to the Pauli Exclusion Principle helps us understand why atoms behave the way they do. It’s the secret sauce that makes everything around us possible.
So, the next time you hear about the Pauli Exclusion Principle, don't let it intimidate you. Think of it as the universe's way of saying, "Everyone gets their own space, and a little bit of uniqueness." It’s a simple rule, but its impact is absolutely profound. It’s the quiet, unseen force that ensures the stability and diversity of the matter that makes up our amazing world!
