Which Diagram Represents A Gas That Has Been Ionized
Ever looked up at the dazzling spectacle of the aurora borealis, or wondered how those neon signs actually glow? The magic behind these captivating displays, and so much more, boils down to a fascinating scientific concept: the ionization of gases. It might sound a bit technical, but understanding ionized gases is like unlocking a secret code to some of the most visually stunning and technologically important phenomena around us. Think of it as a backstage pass to the universe's light show and a peek into the engines of modern tech!
Unlocking the Glow: What Happens When Gases Get Energized?
So, what exactly is an "ionized gas"? In our everyday world, gases are made up of tiny particles called atoms. These atoms are usually electrically neutral, meaning they have an equal number of positive and negative charges. But, when you zap a gas with enough energy – think of a jolt of electricity, intense heat, or even powerful radiation – things get interesting. This energy can knock one or more of the negatively charged electrons right out of an atom.
When an electron leaves, the atom is no longer neutral. It now has more positive charges than negative ones, making it a positively charged ion. At the same time, the ejected electron is a negatively charged particle on its own. Together, these charged particles – the ions and the free electrons – make up what we call an ionized gas, or more scientifically, a plasma. Plasma is often called the "fourth state of matter," distinct from solids, liquids, and gases.
Why Should We Care About Ionized Gases?
The applications and occurrences of ionized gases are incredibly diverse and impactful. For starters, they are the stars of the show in many natural wonders. That breathtaking aurora borealis? It's caused by charged particles from the sun (which is essentially a giant ball of plasma!) colliding with gases in Earth's atmosphere, ionizing them and causing them to emit light. Lightning, too, is a dramatic, fleeting example of a natural ionized gas channel.
Beyond the natural world, ionized gases are workhorses in technology. Those vibrant neon signs you see advertising everything from diners to disco clubs? They work by passing an electric current through a gas in a tube. This current ionizes the gas, and when the charged particles recombine or are excited, they release energy in the form of visible light. Different gases produce different colors, making them incredibly versatile for signage.

But the influence of ionized gases goes much deeper. They are crucial in various industrial processes. For example, plasma torches are used for cutting and welding metals with incredible precision due to the high temperatures they can generate. In the realm of semiconductors, plasma etching is a vital technique used to create the intricate circuits found in your smartphones, computers, and pretty much all modern electronics. Without plasma, our digital world would grind to a halt!
The scientific exploration of ionized gases is also a hotbed of research. Scientists are investigating using plasmas in fusion energy research, aiming to harness the same power source that fuels the sun to provide clean, abundant energy here on Earth. They are also explored for advanced medical treatments, sterilization, and even for propelling spacecraft.

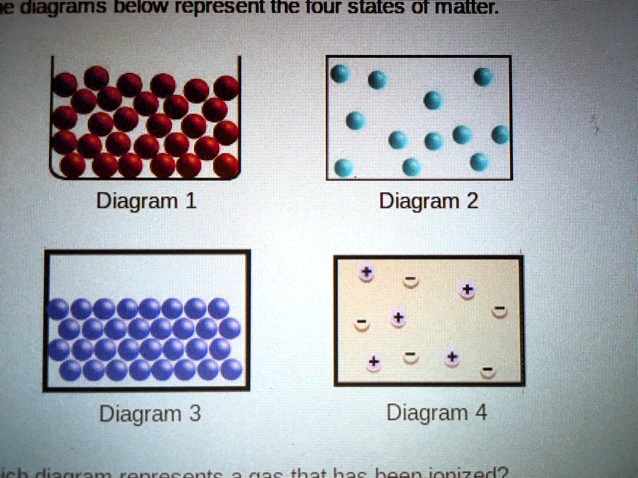
Visualizing the Difference: What an Ionized Gas Diagram Looks Like
Now, let's get to the fun part: how do we represent this ionized state visually? When we talk about diagrams representing gases, we're often looking at models that show the particles and their arrangement. In a regular, non-ionized gas, you'd typically see representations of neutral atoms or molecules moving around freely, with plenty of space between them.
When a gas becomes ionized, the picture changes dramatically. Instead of just neutral atoms, you'll see a mix of positively charged ions and negatively charged electrons, often depicted as separate entities. These charged particles are highly reactive and interact with each other and with any external fields. A diagram representing an ionized gas will likely show:
- Positive Ions: These might be represented by circles with a "+" sign inside, indicating they've lost an electron.
- Free Electrons: These would be shown as small dots or circles, often with a "-" sign, representing the ejected electrons.
- Neutral Atoms/Molecules (possibly): Depending on the degree of ionization, there might still be some neutral particles present.
- Increased Motion and Interaction: The charged particles in plasma are very energetic and interact strongly, so diagrams might imply more chaotic or directed motion compared to a simple gas.
The key visual cue for an ionized gas is the presence of these distinct, separated charges. It's the breaking apart of the neutral atom that defines this energetic, glowing state. So, the next time you see a neon sign flicker to life or marvel at the dancing auroras, you're witnessing the captivating power of ionized gases!
