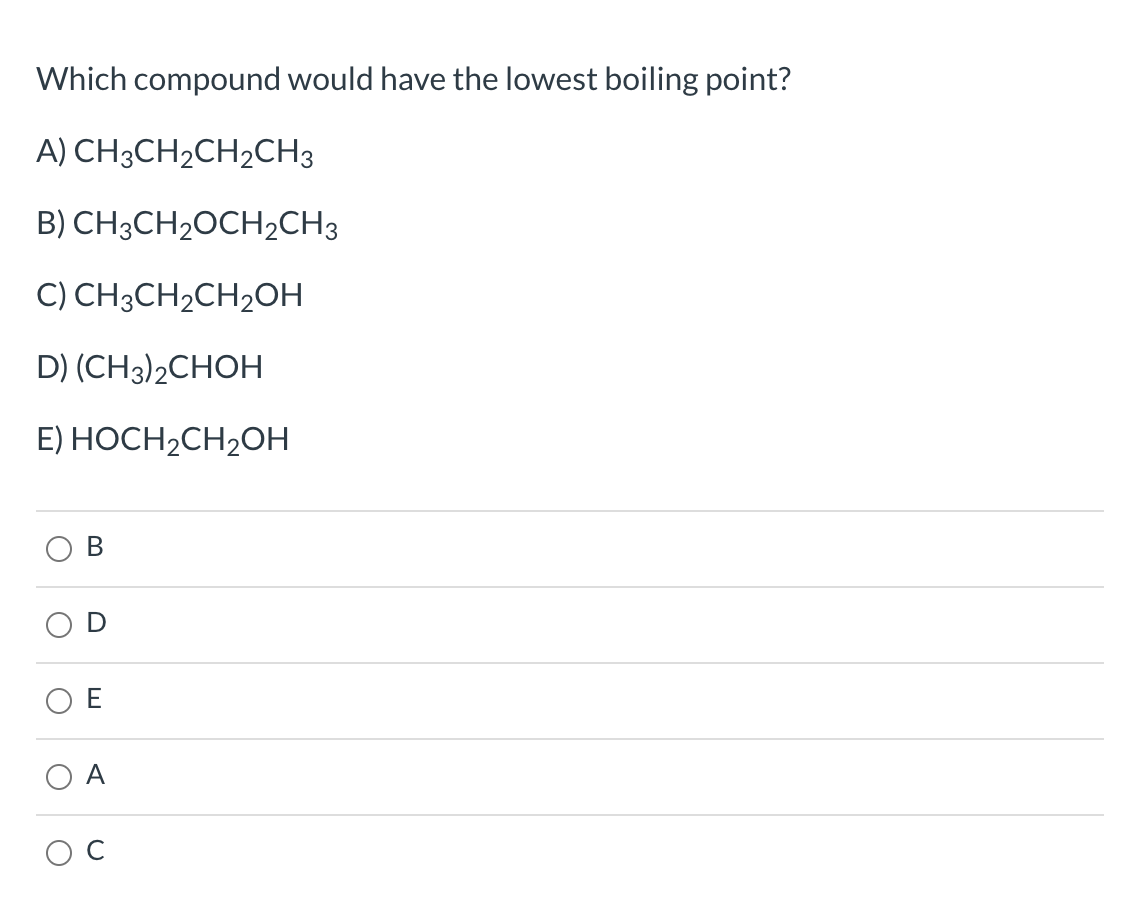
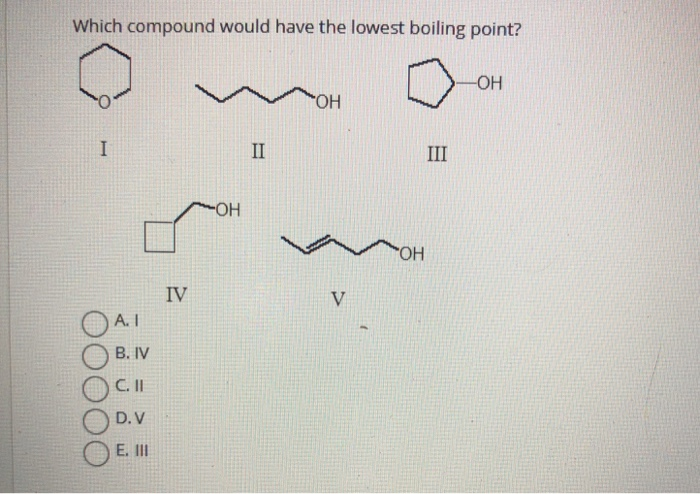
Which Compound Would Have The Lowest Boiling Point
Ever stood in the kitchen, staring at your spice rack, and wondered which of those little jars holds the least enthusiastic molecule? Or maybe you’ve been at a picnic, watching water boil for that crucial cup of tea, and thought, “Man, some things just seem to give up easier than others when it comes to heating up.” Well, my friends, we’re diving headfirst into the wonderfully wacky world of boiling points, and figuring out which compound is the ultimate slacker, the undisputed champion of “Nope, not doing that heat thing.”
Think of boiling point like a party. Some molecules are total party animals, needing a lot of energy (heat!) to get them jumping around and turning into a gas. Others? They’re the ones who just want to chill on the couch, maybe with a Netflix binge, and don't need much convincing to ditch the liquid life and float around the room like a particularly unmotivated ghost. We're on a quest to find that couch potato molecule.
So, how do we pick the winner of the “Least Likely to Boil” award? It’s all about how much those little molecular buddies like to hang out together. Imagine them as a group of friends at a chilly bus stop. If they’re all bundled up, holding hands, and telling each other jokes to stay warm, it’s going to take a lot of persuasion (heat) to get them to scatter and go their separate ways. But if they’re a bunch of introverts, barely acknowledging each other, they’ll be happy to drift apart with just a gentle nudge.
The main culprit behind these molecular huddles is something called intermolecular forces. Don’t let the fancy name scare you! It’s just a way of saying how strongly the molecules are attracted to each other. Think of it like static cling, but on a much, much tinier scale. The stronger these clingy forces are, the more energy you need to rip them apart and get them to boil.
We're talking about a few different types of these sticky situations. You’ve got the really strong ones, like hydrogen bonding. This is where a hydrogen atom in one molecule gets super attracted to an oxygen or nitrogen atom in another. It's like that one friend who always needs a hug. Water, bless its little heart, is a prime example of a molecule that’s big on hydrogen bonding. That’s why water has a surprisingly high boiling point – it takes a lot of energy to get those hug-happy water molecules to break free!
Then there are dipole-dipole interactions. Imagine molecules that are a bit like a tiny magnet, with a positive end and a negative end. They’ll naturally stick to each other, positive to negative. It’s not quite as intense as a full-on hug, but it’s still a definite connection. These guys need a decent amount of heat to get going.
And finally, we have the weakest of the bunch: London dispersion forces (sometimes called van der Waals forces, which sounds way cooler, like a secret agent name). These are like those awkward acquaintances you bump into at a party. They’re not actively seeking each other out, but sometimes, just by chance, they get close enough to have a fleeting attraction. It’s all about temporary imbalances in electron distribution. Think of it like a tiny, accidental nudge. These forces are way weaker, and they’re present in all molecules, but they become the dominant force when there aren't any of the stronger attractions at play.

So, who’s the ultimate champion of low boiling points? Generally, it’s going to be molecules that are small and nonpolar. Why nonpolar? Because nonpolar molecules don't have those distinct positive and negative ends, meaning they can't do the dipole-dipole dance, and they definitely can’t do the hydrogen bonding hug. They're the molecules that are just kind of… floating through life, barely interacting.
Let’s break down some contenders. Imagine you’ve got a lineup of potential boiling point slackers. We’ve got the big, clunky molecules, and the tiny, sleek ones. We’ve got the ones that are all charged up, and the ones that are just… neutral.
Consider something like methane (CH₄). This little guy is super simple. It's got one carbon atom and four hydrogen atoms, all arranged in a nice, symmetrical tetrahedron. Because of its symmetry and the fact that carbon and hydrogen have very similar electronegativities (their "pull" on electrons), methane is pretty much nonpolar. It’s like a perfectly round, smooth ball – it doesn’t have any little nooks or crannies for other molecules to grab onto. The only forces at play here are those weak London dispersion forces.
Now, contrast that with something like water (H₂O). As we mentioned, water has those powerful hydrogen bonds. Those bonds are like superglue holding water molecules together. It needs a whopping 100°C (212°F) to finally get all those molecules to break free and become steam. Methane, on the other hand, boils at a chilly -161.5°C (-258.7°F)! That’s colder than a penguin’s picnic in Antarctica.

Think of it this way: getting water to boil is like trying to get a group of very enthusiastic dancers in a conga line to stop and go their separate ways. They’re all linked up and having a blast. Getting methane to boil is like trying to get a few solitary individuals standing at a bus stop to wander off. They're not really connected in the first place.
Another way to think about it is with molecular size. Generally, as molecules get bigger, they have more electrons, and these electrons can create more temporary dipoles, leading to stronger London dispersion forces. So, a smaller molecule, all other things being equal, will have weaker London dispersion forces than a larger one.
Let’s take helium (He). This is the simplest element, just a single atom. It doesn’t even form molecules in the typical sense unless you force it to. Helium atoms are tiny and have very, very weak attractions between them. The forces are almost exclusively London dispersion forces, and because it's just a single atom and so small, these forces are incredibly weak. Helium boils at an astounding -268.9°C (-452°F). That’s practically absolute zero, the coldest theoretically possible temperature! It’s like the ultimate hermit, barely interacting with anyone, needing the absolute least amount of coaxing to go from liquid to gas.
So, if we're looking for the lowest boiling point, we’re generally hunting for the smallest, most nonpolar molecules. And often, those are the noble gases, like helium and neon. They’re so unreactive, they’re like the aloof celebrities of the periodic table – they just do their own thing, barely mingling.

Let’s compare helium to its slightly larger, but still very aloof cousin, neon (Ne). Neon is also a noble gas, meaning it’s super nonreactive. It boils at -246°C (-410.8°F). Still incredibly cold, but a bit higher than helium. Why? Neon atoms are slightly larger than helium atoms, so they have a few more electrons, which means slightly stronger London dispersion forces. Imagine two tiny pebbles versus two slightly bigger pebbles. The bigger pebbles will have a tiny bit more friction when you try to slide them past each other.
Now, let’s throw in another common gas, nitrogen (N₂). Nitrogen gas, the stuff that makes up most of our atmosphere, is a diatomic molecule – two nitrogen atoms bonded together. While the N-N bond itself is strong, the molecule as a whole is nonpolar. So, the only intermolecular forces are London dispersion forces. Nitrogen boils at -195.8°C (-320.4°F). Still very cold, but higher than helium and neon. This is because nitrogen molecules are larger than individual helium or neon atoms.
What about oxygen (O₂)? Similar to nitrogen, it’s a diatomic molecule and nonpolar. It boils at -183°C (-297.4°F). Again, a bit higher than nitrogen, likely due to slightly larger size and more electrons leading to slightly stronger London dispersion forces. These guys are like the slightly more social introverts. They’re not hugging, but they’re definitely aware of each other’s presence.
So, to summarize our low-boiling point champions: we’re looking for molecules that are small and nonpolar. This means they have minimal intermolecular forces, primarily just the weak London dispersion forces. The less "sticky" they are, the less energy (heat) they need to break free from their liquid buddies and float off into the gaseous world.
When you think about it, it makes perfect sense. Imagine trying to get a bunch of tiny, lightweight ping pong balls to fly apart. They’ll scatter with a gentle puff of air. Now imagine trying to get a bunch of heavy bowling balls to do the same. You’ll need a serious gust of wind! The ping pong balls are our low-boiling point compounds, and the bowling balls are our high-boiling point ones.
The compounds with the absolute lowest boiling points are usually the noble gases, especially the lighter ones like helium. They are monatomic (single atoms), perfectly nonpolar, and incredibly small. They are the ultimate embodiments of molecules that just don't want to be bothered with strong interactions. They’re the ones who show up to the molecular party, take one look around, and immediately head for the exit, needing only the faintest nudge to get there.
So, the next time you’re boiling water, or perhaps marveling at how quickly a balloon filled with helium seems to lose its bounce (because the helium is escaping!), remember the molecular dance. It’s all about how much those tiny particles want to hold hands. And in the quest for the lowest boiling point, we’re rooting for the loners, the introverts, the molecules that just can’t be bothered to get too close.
It’s a simple principle, really: less attraction equals less heat needed. And who doesn’t love a good shortcut? These low-boiling point compounds are the masters of taking the easy route, the molecular equivalent of hitting the snooze button one too many times. They’re the undisputed champions of chilling out, and their boiling points are a testament to their laid-back molecular lifestyle.
