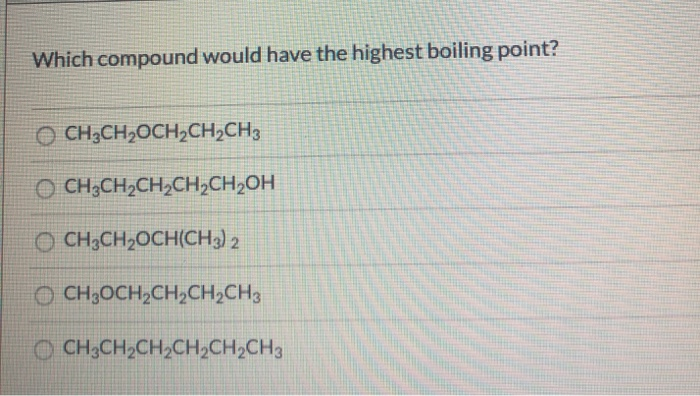
Which Compound Would Have The Highest Boiling Point
So, I was at my friend Sarah’s place the other day, helping her move some boxes. You know how it is – a delightful mix of dusty memories and the inevitable existential dread of unpacking. Anyway, we stumbled upon this old chemistry textbook from her high school days. It was dog-eared, had doodles of terrible stick figures in the margins, and smelled faintly of stale pizza. I, of course, being the nerd I am, immediately flipped it open.
And there it was, a chapter on intermolecular forces. Not exactly beach reading, but surprisingly fascinating. We got talking, and the conversation took a weird turn, as it often does with me. We started wondering, amongst all the different chemicals out there, which one would just refuse to boil? Like, which one would put up the biggest fight against becoming a gas? Which compound would have the absolute highest boiling point? It’s a silly question, I know, but it got me thinking.
It’s easy to take boiling for granted, isn’t it? We see water turn into steam, we boil pasta, we evaporate solvents in the lab. It’s just a thing that happens. But why it happens, and why some things are way harder to get to that point than others… that’s where the magic (and a bit of science) lies. So, settle in, grab a cuppa, and let’s dive into this somewhat nerdy, but hopefully fun, exploration of why some molecules are just built for a good, long, liquid stay.
The Battle of the Molecules: What Makes Something Boil?
Okay, so before we can even think about who wins the boiling point marathon, we need to understand what boiling actually is. Think of it like a party. Molecules are chilling together, having a good old time in the liquid phase, bumping into each other, holding hands, whatever it is they do. When you add heat, it's like turning up the music and handing out energy drinks. The molecules start dancing faster, getting more excited. Eventually, some of them get so excited, they break free from the crowd and float off on their own as a gas. That’s boiling!
The key to this whole shindig is the forces that hold these molecules together in the first place. In the liquid state, they’re attracted to each other. These are called intermolecular forces. The stronger these attractions are, the more energy you need to pump into the system to get those molecules to break free and become a gas. It’s like trying to pull apart a group of really tight-knit friends versus a bunch of acquaintances who barely know each other. You’re gonna need way more effort for the friends.
The Usual Suspects: Van der Waals and Dipole-Dipole
Let’s start with the basics, the more common types of intermolecular forces you’ll encounter in chemistry class. You've got your London dispersion forces (sometimes called Van der Waals forces). These are the weakest of the bunch. They’re temporary attractions that arise from the random movement of electrons in a molecule, creating a tiny, fleeting imbalance of charge. Think of it like a little electrical hiccup. The more electrons a molecule has, and the bigger and more spread out it is, the stronger these dispersion forces can be. So, bigger molecules often have higher boiling points just because of this.
Then you have dipole-dipole forces. These happen in molecules that are polar. Polar molecules have a slightly positive end and a slightly negative end, like tiny magnets. These opposite charges attract each other, holding the molecules together. Water is a classic example – it’s super polar, which is why it has a relatively high boiling point for its size (100°C, which is pretty decent!).

So, if we were just comparing simple, non-polar molecules, the bigger ones would generally have higher boiling points. Think of methane (a small gas) versus octane (a liquid fuel). Octane is much bigger, has more electrons, and therefore stronger London dispersion forces, making it harder to vaporize. Simple enough, right?
The Superstar: Hydrogen Bonding
But wait, there’s a superstar in the intermolecular force world. It’s called hydrogen bonding. This isn't a true chemical bond in the sense of sharing electrons, but it’s a super strong type of dipole-dipole attraction. It happens when a hydrogen atom is bonded to a highly electronegative atom like oxygen (O), nitrogen (N), or fluorine (F). This makes the hydrogen atom really, really positive, and it gets strongly attracted to a lone pair of electrons on an oxygen, nitrogen, or fluorine atom of another molecule. It’s like a really strong, reliable handshake.
Water, as I mentioned, is the poster child for hydrogen bonding. The H-O bonds are very polar, so the hydrogen atoms are quite positive, and the oxygen atoms have those handy lone pairs. This is why water stays liquid up to a relatively high temperature. If water only had London dispersion and regular dipole-dipole forces, it would probably be a gas at room temperature, just like methane or ammonia (though ammonia also has hydrogen bonding).
Alcohol molecules (like ethanol, the stuff in your drink) also have hydrogen bonding because of the -OH group. That’s why they tend to have higher boiling points than similar-sized molecules without that functional group. Ever noticed how rubbing alcohol evaporates pretty quickly? It’s still got some of those other forces at play, but the hydrogen bonding is a significant player keeping it in liquid form longer than, say, propane.
When Molecules Get Really, Really Big and Really, Really Sticky
So, we've got London dispersion, dipole-dipole, and hydrogen bonding. These can get pretty strong, especially in large molecules. Imagine a very long chain of hydrocarbons, like the ones found in waxes or oils. They have tons of electrons, so their London dispersion forces are enormous. The longer the chain, the more surface area for interaction, and the higher the boiling point. Think about paraffin wax – you have to heat that up quite a bit to melt it, let alone boil it.

But what happens when we move beyond simple organic molecules and start looking at giant structures, like polymers or even solids held together by incredibly strong forces? This is where things get truly interesting, and the boiling points start to climb into the stratosphere.
Ionic Compounds: The Unbreakable Bonds?
Let’s talk about ionic compounds. These are formed when atoms transfer electrons, creating ions – positively charged cations and negatively charged anions. Think of table salt, sodium chloride (NaCl). Sodium loses an electron to become Na+, and chlorine gains it to become Cl-. These oppositely charged ions are attracted to each other incredibly strongly through electrostatic forces. It's not just a little handshake; it's like they're glued together by a powerful magnetic attraction.
In a solid ionic compound, these ions are arranged in a crystal lattice, a repeating 3D structure. To melt or boil an ionic compound, you’re not just trying to pull apart individual molecules; you’re trying to overcome these strong electrostatic attractions between all the ions in the lattice. That requires a huge amount of energy. Table salt melts at a scorching 801°C and boils at a blistering 1413°C! That’s way higher than water, or even most organic molecules.
So, simple ionic compounds like NaCl already have seriously impressive boiling points. But we can go even higher.

Covalent Networks: The Ultimate Strength
Now, let’s talk about the true titans of high boiling points: covalent network solids. These are compounds where atoms are linked together by a continuous network of covalent bonds, essentially forming one giant molecule. There aren’t distinct, separate molecules floating around. Instead, the entire solid is held together by these strong, shared electron bonds. Breaking these bonds takes an insane amount of energy. Imagine trying to break apart a diamond with your bare hands – it’s not happening!
The classic example, and the undisputed champion in many a high school chemistry quiz, is diamond. Diamond is made entirely of carbon atoms, with each carbon atom covalently bonded to four other carbon atoms in a tetrahedral structure. This creates an incredibly strong and rigid three-dimensional network. To boil diamond (if it were even possible without it just decomposing or turning into graphite first), you'd have to break all those incredibly strong covalent bonds.
Theoretically, diamond has an incredibly high boiling point, often cited as being around 4027°C (or 7280°F). That's hotter than the surface of many stars! (Okay, maybe not that hot, but definitely hotter than the sun's surface, which is around 5,500°C. My bad, stellar trivia is not my strong suit today!) The point is, it’s unimaginably hot.
Another example is silicon dioxide, also known as silica (SiO2), the main component of sand and glass. In its crystalline form (like quartz), it also forms a covalent network where silicon atoms are bonded to oxygen atoms, and oxygen atoms are bonded to silicon atoms, creating a huge, interconnected structure. Silicon dioxide doesn't have quite the same strength as diamond, but it still has a very, very high melting point (around 1710°C) and an even higher boiling point (estimated to be around 2950°C).
These materials are so stable and resistant to heat because you're not just overcoming weak attractions between molecules; you're fundamentally breaking the strong chemical bonds that hold the entire structure together. It’s like trying to un-bake a cake – it’s a whole different level of difficulty!

So, Who Wins?
Given what we've discussed, the compound with the highest boiling point would almost certainly be a substance that forms a very strong covalent network. The strength and density of these covalent bonds, extending throughout the entire material, make them incredibly resistant to breaking apart into a gaseous state.
While there are other substances with extremely high boiling points, like some refractory metals (think tungsten, which boils around 5930°C!), they are elements, not compounds. When we’re talking about compounds, the diamond structure (pure carbon) or similar robust covalent network structures are the clear front-runners for the title of "Highest Boiling Point Compound."
It's important to note that for many of these super-high boiling point materials, reaching their actual boiling point in a lab setting can be incredibly challenging. They might decompose or undergo other transformations before they even get close to becoming a true gas. But theoretically, based on the strength of the forces holding them together, they win the prize.
A Final Thought on Extreme Heat
It’s kind of wild to think about these substances just refusing to turn into vapor, even under conditions that would melt or vaporize almost everything else we know. It reminds you that even the most common-looking stuff, like sand, is made of incredibly resilient stuff at the molecular level. It's a good reminder that the universe is full of extremes, and sometimes the answers to seemingly simple questions lead us down paths of incredible complexity and astonishing feats of molecular tenacity.
So, next time you’re boiling water for tea, spare a thought for your friend diamond, chilling out at thousands of degrees Celsius, completely unbothered. It puts things in perspective, doesn’t it? And who knows, maybe this little dive into boiling points has sparked a new curiosity in you. That’s the best part about science, right? It’s always a journey, and sometimes, the most interesting destinations are found in the dustiest old textbooks.
