Which Compound Represents The Compound Aluminum Iodide
Alright, gather 'round, folks, and let me tell you a tale. It’s a tale of atoms, of ions, and of a compound that sounds a bit like a superhero's sidekick, or maybe a particularly fancy brand of artisanal cheese. We're talking about Aluminum Iodide, people! Now, you might be thinking, "Whoa there, chemistry nerd! I just wanted my morning latte, not a lecture on chemical bonding." But trust me, this isn't your grandma's dusty textbook. This is the behind-the-scenes gossip of the periodic table, the drama you won't find on reality TV. So, settle in, grab your biscotti, and let’s unravel the mystery of our friend, Aluminum Iodide.
First off, let's set the scene. Imagine two very different characters meeting at a cosmic mixer. On one side, we have Aluminum. Think of Aluminum as the cool, slightly aloof teenager. He’s got three electrons he’s just itching to get rid of. He’s like, "Ugh, these things are so last season," and he’s ready to make a clean break. He’s got that metallic sheen, you know? Shiny, useful, can be molded into everything from airplane wings to tinfoil. A real go-getter.
On the other side, we have Iodine. Now, Iodine is a bit of a different vibe. She’s more of the “I need just one more thing to be complete” type. She’s got seven valence electrons, and she’s got this longing, this desire, for just one more electron to finally feel whole. Imagine her at a party, looking around with a sigh, "If only someone had one more chip to share..." She’s a bit of a drama queen, but in a fascinating, glowy way. You’ve probably seen her in those fancy antiseptic solutions, giving cuts that slightly alarming brownish tint. She’s got personality, that’s for sure.
So, what happens when these two meet? It’s a classic case of opposites attract, or rather, opposites transfer. Aluminum, with his abundance of easily-let-go electrons, sees Iodine’s electron-craving gaze. It’s like a match made in molecular heaven. Aluminum is like, "You know what? I’ve got spares. Let’s make a deal." And Iodine is practically doing a little jig, thinking, "Yes! Finally! My electron destiny!"
This electron-swapping, or more technically, electron-transferring, is what we call an ionic bond. It’s not like they’re holding hands and singing Kumbaya. It’s more like Aluminum is a generous, albeit slightly impulsive, donor, and Iodine is an eager, grateful recipient. Aluminum loses an electron and becomes a positively charged ion, which chemists creatively call a cation. And Iodine gains an electron and becomes a negatively charged ion, a anion. Get it? Positively charged Aluminum Ion (Al+) and negatively charged Iodide Ion (I-).

Now, here’s where it gets a little tricky, and where you might need to adjust your latte-sipping posture. Aluminum actually likes to lose three electrons to achieve that super-stable electron configuration that all atoms secretly crave. It’s like finally getting that perfect outfit that just makes everything click. So, Aluminum is ready to shed three electrons, one, two, three! But remember Iodine? She’s only built to accept one electron at a time. She’s got her limits, bless her electron-hungry heart.
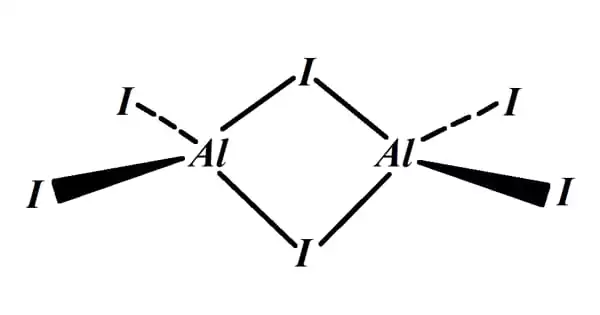
So, for every Aluminum atom that wants to get rid of three electrons, you’re going to need three Iodine atoms to come along and take one each. It's a carefully orchestrated dance of electron distribution. Think of it like a really popular celebrity (Aluminum) with a limited number of autographs (electrons) to give, and a whole crowd of adoring fans (Iodine atoms) clamoring for one. They’ll need to line up! One, two, three! And thus, the recipe for Aluminum Iodide is born.
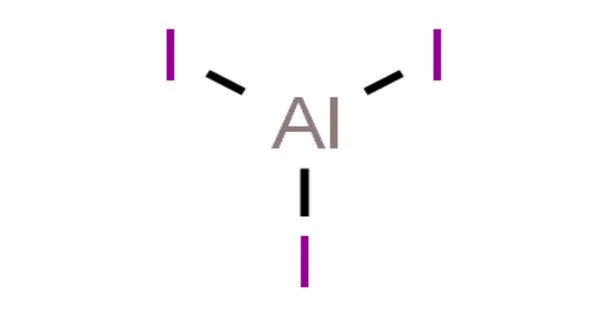
The chemical formula, the secret handshake of this compound, is AlI₃. See? One Aluminum (Al) and three Iodines (I). It's like the chemical world's way of saying, "This is the winning combination!" It's not just a random assortment of letters and numbers; it’s a precise representation of how these elements have decided to buddy up and share their electron responsibilities.
Now, what kind of compound is this Aluminum Iodide? Is it a solid? A liquid? Does it sparkle? Well, it’s a bit of a shapeshifter, which adds to its mystique. In its pure, solid form, it’s typically a crystalline solid. Think of tiny, perfect geometric shapes, like microscopic diamonds, but probably not as sparkly, and definitely not something you’d wear to a gala. It tends to be a white or off-white color.

But here’s the kicker, and this is where things get really interesting, and maybe a little dramatic. Aluminum Iodide is notoriously hygroscopic. What does that mean, you ask? It means it’s an absolute sponge for water. Like, it’ll actively suck moisture right out of the air. Imagine leaving a perfectly dried raisin on your counter, and then bringing over a tiny bit of Aluminum Iodide. Within minutes, that raisin would be plump and rehydrated, and the Aluminum Iodide would be… well, a bit of a mess. It reacts with water, forming complex hydrated structures, and can even release hydrogen iodide, which is a nasty acidic gas. So, it’s basically saying, "Give me all the water! And maybe a bit of acid too, why not?" It’s like that friend who’s always a little too enthusiastic when the snacks come out.
This makes it a bit of a diva to handle in the lab. You have to keep it in a dry environment, like a hermetically sealed container. It’s the chemical equivalent of wearing a fancy silk dress to a water balloon fight – you just don’t do it! Chemists who work with it have to be super careful, wearing gloves and working in fume hoods. It’s a good reminder that even seemingly simple compounds can have their own quirky personalities and demands.
So, to recap our adventure: we’ve got Aluminum, the electron-rich teen, and Iodine, the electron-coveting diva. They meet, they bond (ionically, of course), and they form Aluminum Iodide with the formula AlI₃. It's a white, crystalline solid that’s incredibly thirsty for water and can be a bit of a handful. It’s not just a chemical formula; it’s a story of electron exchange, a testament to the dynamic relationships that exist at the atomic level. Pretty cool, right? Now, who needs a refill?
