Which Compound Is Not A Constitutional Isomer Of The Others
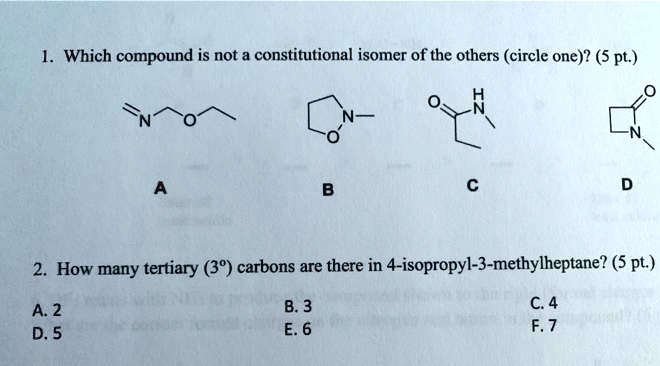
Hey there, my fellow molecule enthusiasts! Ever find yourself staring at a bunch of chemical structures and feeling a little bit… overwhelmed? Like, they all look kind of similar, but then suddenly one just pops out and says, "Nope, not part of this club!" That’s exactly what we’re diving into today. We’re on a mini-adventure to spot the odd one out, the compound that just isn't playing by the same constitutional isomer rules as its buddies. Think of it as a chemical scavenger hunt, but way cooler and with fewer mosquitoes.
So, what in the name of Avogadro’s number are we even talking about? Constitutional isomers! Don't let the fancy name scare you. It basically means molecules that have the same molecular formula – they’ve got the same number of each type of atom, like the same number of carbons, hydrogens, oxygens, and so on. BUT! And this is the big, juicy "but" – they have their atoms connected in a different order. It's like having the same set of LEGO bricks, but building two completely different things. One might be a car, the other a spaceship, and they’re definitely not interchangeable, right?
Imagine you have a bag with 4 carbon atoms and 10 hydrogen atoms. That molecular formula is C4H10. Easy peasy. Now, with those exact same atoms, you could put them together in a few different ways. You could have a straight chain, like a little molecule highway: CH3-CH2-CH2-CH3. That’s called butane. Nice and simple. Or, you could take one of those middle carbons and make it a branch. So, you’d have a chain of three carbons, and then one carbon sticking out from the middle one. Like a T shape, but with hydrogens chilling everywhere. That’s isobutane (or 2-methylpropane, if you want to sound extra fancy). See? Same atoms, different arrangement. Totally different molecules. They’ll have different boiling points, different melting points, and they’ll probably react in slightly different ways. It’s like comparing a perfectly straight road to one with a few quirky bends – both get you there, but the journey is different!
Today, though, we're not just looking at two isomers. We're looking at a group of compounds, and one of them is going to be the black sheep. The one that, despite looking like it should fit in, is actually totally different. It's not a constitutional isomer of the others. This usually happens when two (or more) compounds are constitutional isomers of each other, but then there’s one that has a different molecular formula altogether. Or, sometimes, they have the same molecular formula, but one of them is a stereoisomer or some other type of isomer that doesn't qualify as a constitutional isomer. But let's keep it simple for now – the most common way to be the odd one out is to have a different molecular formula. It’s like having a group of apples, and then one sneaky orange. They’re both fruit, sure, but they’re not the same kind of fruit, if you catch my drift.
So, how do we play this game? The first, and arguably most important, step is to determine the molecular formula for each compound. This is where your trusty counting skills come in. Go atom by atom, molecule by molecule. Don't rush it! It’s like checking your homework before you hand it in – a little bit of careful checking can save you from a world of confusion. For each molecule, count up all the carbons, all the hydrogens, all the oxygens, and anything else that might be lurking in there. Write down the molecular formula for each one. Think of it as giving each molecule a secret ID card.
Once you have those ID cards, you just look for the one that doesn't match the others. If you have, say, three molecules with the formula C3H8O and one with C3H6O, then the C3H6O is your outlier. It’s not a constitutional isomer because it’s literally made of a different set of building blocks. It’s like trying to build a LEGO car with only three wheels – it's just not going to work out the same way as the four-wheeled ones, no matter how you try to arrange them.
Sometimes, the molecular formulas will match for most of the compounds, but then you’ll have one that has the same molecular formula but is a different type of isomer. This is where things get a little more nuanced, but still totally manageable. For instance, if we’re talking about compounds with double or triple bonds, or rings, the number of hydrogens can change. Let's say you have a bunch of C4H10 molecules (our trusty butane buddies). But then, one of them is actually C4H8. That C4H8 molecule is missing two hydrogens. Where did they go? Well, they probably got together and formed a double bond, or maybe they decided to form a ring structure. A molecule with a double bond or a ring will have fewer hydrogens than a similar molecule that’s all single bonds and no rings. So, if you see a formula with fewer hydrogens than expected for a saturated hydrocarbon (meaning all single bonds and no rings), and the other formulas are saturated, that’s a big clue that you’ve found your rebel.
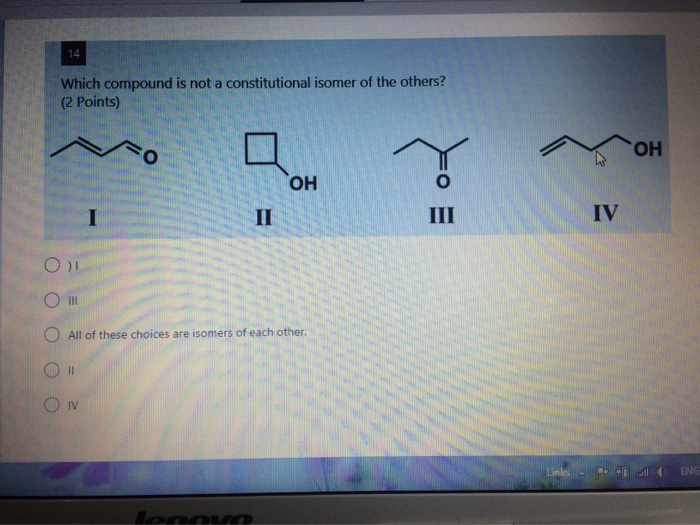
Let's try a little hypothetical scenario, shall we? Imagine you're presented with these three potential friends for our molecular party:
Compound A:
Let's say Compound A looks like this: CH3-CH2-OH. What's its molecular formula? We've got 2 carbons, so C2. Then we have 3 + 2 + 1 hydrogens, which is 6. And one oxygen. So, C2H6O. Easy!
Compound B:
Now, Compound B is a bit of a shape-shifter. It’s CH3-O-CH3. Let’s count again. Still 2 carbons (C2). Still 3 + 3 hydrogens (6). And one oxygen. So, C2H6O. Huh, same as Compound A!
Compound C:
Finally, Compound C comes along. It's a ring structure, like a little hexagonal pizza with carbons at each corner and hydrogens attached. This is cyclohexane. For cyclohexane, you have 6 carbons. Each carbon in a perfect ring of single bonds will have 2 hydrogens attached. So, C6H12. Whoops, I forgot to mention earlier that sometimes the question might be phrased in a way that implies all the isomers are related to a specific molecular formula. Let's rewind and make our example better.
Okay, let's stick with our C2H6O theme. We found Compound A (ethanol) and Compound B (dimethyl ether), both with C2H6O. They are constitutional isomers of each other. Now, let's introduce our third contender:
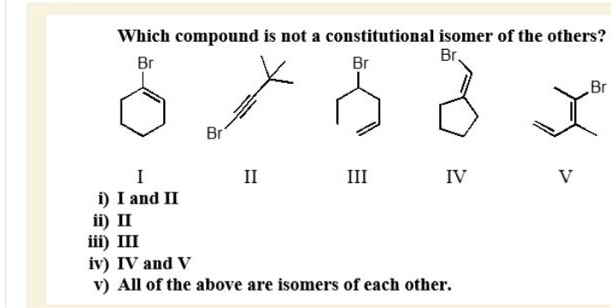
Compound C (Revised):
Imagine Compound C is something like CH3-CH2-CHO. Let's count: 3 carbons (C3). Hydrogen count: 3 + 2 + 1 = 6. And one oxygen. So, C3H6O. This molecule is called propanal.
Now, let's look at our ID cards again:
- Compound A: C2H6O
- Compound B: C2H6O
- Compound C: C3H6O
See it? Compound C has a different molecular formula! It has an extra carbon and two fewer hydrogens than Compounds A and B. Therefore, Compound C is not a constitutional isomer of Compounds A and B. It’s in a different molecular league, a different family of atoms altogether. It’s like you’re comparing two identical twins (A and B) and then suddenly a cousin shows up (C) who looks a little similar but is definitely not one of the twins.
Another common way this plays out is with rings versus chains, or double bonds. Let's try another set. Suppose we have:
Molecule 1:
Butane: CH3-CH2-CH2-CH3. Formula: C4H10.
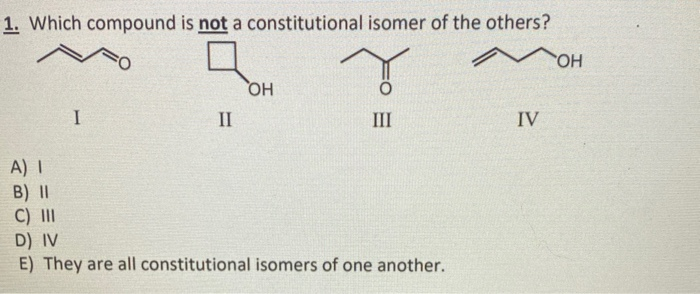
Molecule 2:
Isobutane (2-methylpropane): CH3-CH(CH3)-CH3. Formula: C4H10.
So far, so good! Butane and isobutane are constitutional isomers. They are both members of the C4H10 club.
Molecule 3:
Now, let's throw in Cyclobutane: a square with carbons at each corner and hydrogens. Formula: C4H8. This molecule is a ring with all single bonds. For a saturated hydrocarbon with 4 carbons, the formula should be C4H10. Since cyclobutane has C4H8, it’s missing two hydrogens. This is because in a ring, each carbon is bonded to two other carbons (in addition to its hydrogens), so it doesn't have as many free spots for hydrogens as a linear chain might.
In this case, our ID cards look like:
- Molecule 1: C4H10
- Molecule 2: C4H10
- Molecule 3: C4H8
Boom! Molecule 3 (cyclobutane) is the one that’s not a constitutional isomer of the others. It has a different molecular formula. It’s like comparing a delicious bread loaf (butane and isobutane are basically different shapes of the same loaf) to a fancy croissant (cyclobutane – still made of flour and butter, but a different structure and composition).
Sometimes, the tricky part comes when you have the same molecular formula, but one of them is a stereoisomer. Remember how constitutional isomers are about the order of atom connectivity? Stereoisomers have the same connectivity but differ in the spatial arrangement of their atoms. Think of it like two identical triplets who have the same name and same parents, but one is always wearing a blue shirt and the other a red shirt. They're still fundamentally the same person, just with a visual difference. Constitutional isomers are more like siblings – same general building blocks (parents), but arranged differently to create unique individuals.
For example, if you have 2-butene, there are two forms: cis-2-butene and trans-2-butene. They both have the molecular formula C4H8. They are stereoisomers. If the question was “Which of these is not a constitutional isomer of the others?” and you were given cis-2-butene, trans-2-butene, and, say, 1-butene, they would all be constitutional isomers because their connectivity is different (the double bond is in a different position). But if you were given cis-2-butene, trans-2-butene, and cyclopropane (C3H6), then cyclopropane would be the outlier because it has a different molecular formula. So, the key is always to check the molecular formula first.
The trick to acing these problems is to be systematic. Don't just glance at the pictures and guess. Take a deep breath, grab your imaginary molecular counting beads, and diligently count every single atom. If the molecular formulas are different, you’ve found your winner! If the molecular formulas are the same, then you need to look a little closer at the connectivity. Are the atoms linked in the same sequence? If not, they are constitutional isomers. If they have the same connectivity but differ in 3D space, they are stereoisomers. But usually, the "not a constitutional isomer" question is pointing towards a different molecular formula.
So, the next time you’re faced with a lineup of molecules and asked to find the odd one out, remember this simple mantra: Count your atoms! It’s your secret weapon, your superhero power for spotting that one molecule that just doesn't belong to the same molecular family. It’s not about being smarter, it’s about being thorough. And that, my friends, is a superpower anyone can develop!
And hey, even if you get one wrong now and then, that’s totally okay! Learning is a journey, and sometimes that journey has a few fun detours. The important thing is that you’re engaging with these amazing building blocks of the universe. Each molecule, whether it’s a perfect isomer or the quirky outlier, plays its part in the grand tapestry of chemistry. So, keep counting, keep exploring, and always remember to smile at the fascinating world of molecules. You’re doing great!
