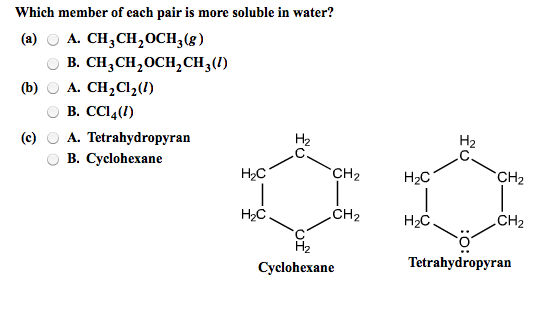
Which Compound In Each Pair Is More Soluble In Water
Ever stared at a glass of water, perhaps with a squirt of lemon or a dissolving tablet, and wondered about the invisible dance happening within? That seemingly simple act of mixing is actually a fascinating world of chemistry at play, and today, we're diving into one of its most popular and useful aspects: solubility! Understanding how and why things dissolve in water isn't just for scientists in lab coats; it's a superpower that explains everything from cooking your favorite meals to how our bodies function. Think about it: why does salt disappear in soup but oil floats stubbornly on top? Why do some medicines dissolve quickly to get to work, while others need special formulations? It’s all about which compound is more eager to buddy up with those friendly water molecules. Get ready to have some fun as we explore the intriguing question: Which compound in each pair is more soluble in water?
The Magic of Dissolving
So, why is this topic so cool and, dare we say, useful? Well, solubility is the backbone of countless everyday phenomena. When you bake a cake, the sugar and leavening agents dissolve in the liquid ingredients, allowing them to react and create that fluffy goodness. In your body, essential nutrients and medications dissolve in your bloodstream, allowing them to travel where they're needed. Even the way we clean our homes relies on solubility; soaps and detergents are designed to dissolve grease and grime in water so it can be washed away. By understanding solubility, we gain a deeper appreciation for the world around us and can even make smarter choices, from choosing the right cleaning products to understanding how to store our food. It’s like unlocking a secret code to how the physical world operates.
Our mission today is to demystify the concept of solubility by comparing pairs of compounds. We'll be playing the role of detectives, figuring out which partner in each duo is more likely to be welcomed into the cozy embrace of water molecules. This isn't about memorizing complex formulas, but rather about recognizing some key principles that make one substance more 'water-friendly' than another. Think of water as a very social molecule, always looking for friends it can easily connect with. The compounds that are best at making these connections are the ones that dissolve best.
Water's Best Friends: Polar Pals
The secret to water's dissolving power lies in its unique structure. Water molecules (H₂O) are polar. This means they have a slightly positive end (where the hydrogen atoms are) and a slightly negative end (where the oxygen atom is). This polarity makes water an excellent solvent for other polar or ionic compounds, which also have charged regions or full charges. Water molecules can surround these charged particles, effectively pulling them apart and dispersing them throughout the solution. It's like a big group hug where water molecules grab onto the positive and negative parts of other substances, making them dissolve.
On the other hand, nonpolar compounds, like oils and fats, don't have these distinct positive and negative ends. Water molecules, being polar, find it difficult to interact with and separate nonpolar molecules. This is why oil and water don't mix – they're like ships passing in the night, each preferring their own kind. So, when we look at our pairs, we'll often be asking: which one is more like water – polar and charged, or nonpolar and neutral?

Let the Solubility Showdown Begin!
Get ready for our exciting comparisons! We'll be looking at common substances, and with a little chemical intuition, we can predict which one is more likely to disappear into your glass of water.
Salt vs. Sugar
Our first contestants are sodium chloride (NaCl), also known as table salt, and sucrose, which is plain old table sugar. Both are white crystalline solids commonly found in kitchens. Salt is an ionic compound, made of positively charged sodium ions (Na⁺) and negatively charged chloride ions (Cl⁻). Sugar, while it contains polar bonds due to the oxygen and hydrogen atoms, is a molecular compound and is not ionic. Water's polar nature is fantastic at surrounding and separating these charged ions in salt. While water can also dissolve sugar by forming hydrogen bonds with its polar groups, the strong ionic attraction in salt makes it generally more soluble in water than sugar, especially at room temperature. So, when it comes to dissolving speed and quantity, salt often takes the lead!
Which compound in each pair is more soluble in water? (a) Strontium
Alcohol vs. Oil
Next up, we have ethanol, the alcohol found in beverages, and vegetable oil. Ethanol has a unique structure: it has a polar 'head' (the -OH group) that likes water, and a nonpolar 'tail' (the carbon chain) that dislikes water. However, the polar 'head' is quite influential in ethanol. Vegetable oil, on the other hand, is primarily made up of long, nonpolar fatty acid chains. Because ethanol has a significant polar component, it can readily interact with water molecules. Vegetable oil, being predominantly nonpolar, cannot. This is why you can mix ethanol and water to create alcoholic drinks, but oil and water remain stubbornly separate. Therefore, ethanol is significantly more soluble in water.
Vinegar vs. Cooking Oil
Let's consider acetic acid (the main component of vinegar) and cooking oil. Acetic acid has a polar carboxyl group (-COOH) that allows it to form hydrogen bonds with water, making it quite soluble. Cooking oil, as we've established, is largely nonpolar. The polar nature of acetic acid allows it to integrate with water molecules, while the nonpolar oil will float on top. You can see this in salad dressings where oil and vinegar separate unless an emulsifier is used. For this pair, vinegar (acetic acid) is the clear winner in water solubility.
Solved Which member of each pair is more soluble in water? | Chegg.com
Lemon Juice vs. Milk
Our final pair features lemon juice and milk. Lemon juice is essentially a solution of citric acid and other compounds in water, all of which are quite polar or ionic. Milk, while it appears uniform, is a complex mixture containing water, fats, proteins, and sugars. The fats in milk are nonpolar. However, milk also contains proteins (like casein) which are polar and act as emulsifiers, helping to disperse the fats throughout the water. When comparing the overall interaction with water, the primarily polar and ionic components of lemon juice, along with the water already present, make it readily miscible. Milk's composition, while having some water-soluble components, also contains a significant amount of dispersed nonpolar fats. In many contexts, the acidic and polar nature of lemon juice makes it more fundamentally 'dissolved' in water's terms than the emulsion of milk. However, it's worth noting milk doesn't 'dissolve' in the same way as salt; it forms a suspension or emulsion. For the sake of comparing which components are more water-friendly, lemon juice’s solution-based nature leans towards greater overall water solubility.
And there you have it! A quick tour through the fascinating world of solubility. Remember, it’s all about the polarity and ionic character of a compound. Substances that have a similar 'personality' to water – meaning they are polar or ionic – tend to dissolve much better. This knowledge is not just fun trivia; it's a key to understanding so many aspects of our world. So next time you're mixing something in water, take a moment to appreciate the invisible, yet powerful, chemical interactions at play!