Which Compound Has The Longest Carbon-carbon Bond Length
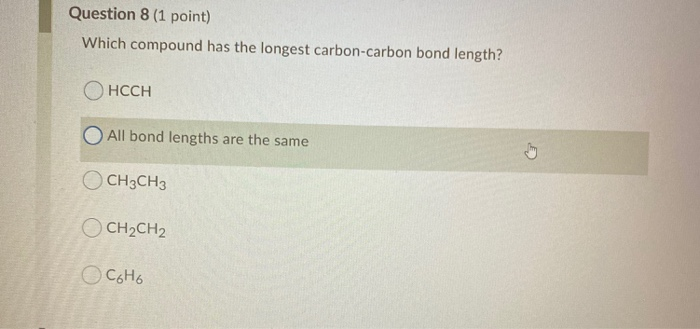
Hey there, curious minds! Ever wondered about the tiny, invisible building blocks that make up… well, everything? Today, we're diving into the wonderfully weird world of chemistry, and not in a stuffy, textbook kind of way. We're talking about something super cool: the longest carbon-carbon bond length! Yep, you heard that right. We're going to explore how atoms can link up and how, sometimes, those links can get a little… stretchy.
Now, I know what you might be thinking. "Bonds? Carbon? Is this going to be… boring?" Absolutely not! Think of it like this: imagine a bunch of Lego bricks. Some click together nice and snug, right? Others… well, they might have a little wiggle room. Carbon-carbon bonds are kind of like that, but on a molecular level. And understanding these tiny differences can actually be surprisingly fascinating. Who knew atoms could have personality, right?
So, what exactly is a carbon-carbon bond? It’s the way two carbon atoms decide to hold hands. Carbon is the superstar of organic chemistry, the element that forms the backbone of life as we know it. From the DNA in your cells to the pizza you might have had for dinner, carbon is everywhere. And its ability to form these sturdy, yet adaptable, bonds is what makes it so special.
Typically, carbon-carbon bonds are pretty consistent. They're like your reliable best friend, always there, always doing their job. But, like any relationship, sometimes things can get a little… strained. Or, in this case, a little longer. And that's where our quest for the longest bond comes in.
The Usual Suspects: Single, Double, and Triple Trouble!
Before we get to the record-holder, let’s quickly chat about the common types of carbon-carbon bonds. You've got your single bond, where two carbon atoms share one pair of electrons. This is your everyday, reliable bond. Think of it as a firm handshake. It’s strong, stable, and allows for a lot of flexibility.
Then there's the double bond. Here, two carbon atoms share two pairs of electrons. This is a bit more intense, like a strong hug. Double bonds are shorter and stronger than single bonds, and they restrict how the molecule can twist and turn. They add a certain rigidity, a determined stiffness.
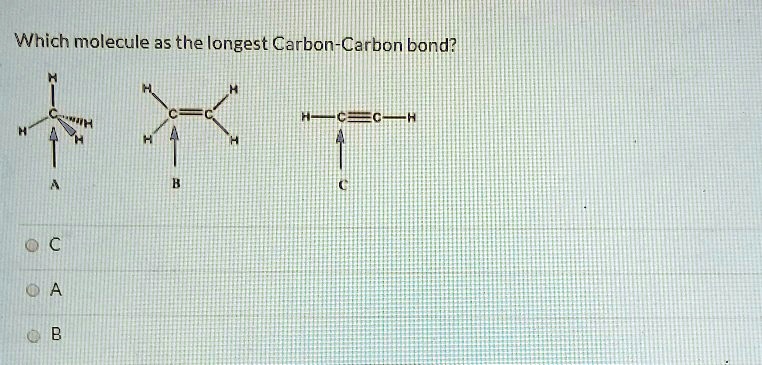
And finally, the most enthusiastic of the bunch, the triple bond. Two carbon atoms sharing three pairs of electrons! This is like a triple-threat handshake, super compact and powerful. Triple bonds are the shortest and strongest of the common types. They’re like tiny, taut strings, holding things together with all their might.
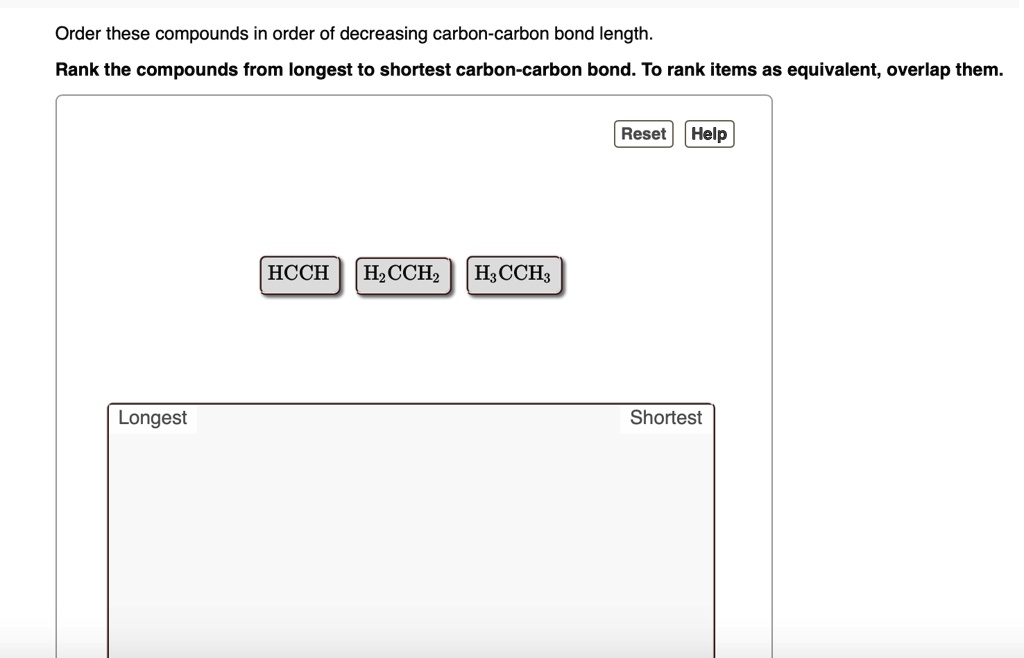
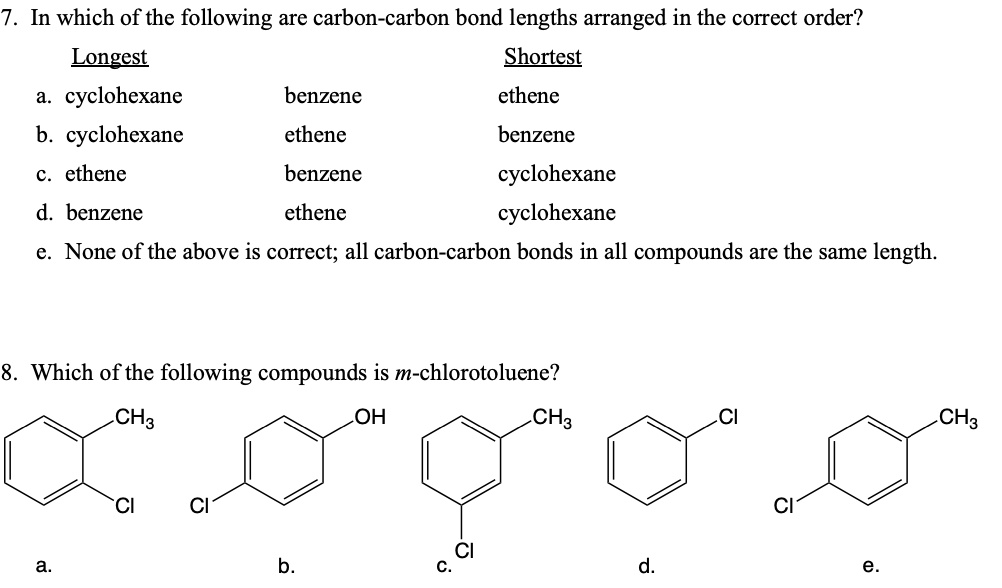
So, if double and triple bonds are shorter and stronger, you'd think the longest bond would definitely be a single bond, right? You’re on the right track! Generally speaking, single bonds are indeed the longest among these common types. But the world of chemistry is full of surprises, and nature loves to push the boundaries.
Enter the Contenders: When Things Get Stretched!
Now, to find the absolute longest carbon-carbon bond, we need to look beyond the everyday molecules we encounter. We need to delve into some pretty specialized chemical structures. Think of it like looking for the world’s longest diving board – it’s not going to be at your local community pool!
What makes a carbon-carbon bond stretch? It’s often about the surrounding environment and the electron distribution. Sometimes, molecules are put under extreme pressure, either physically or electronically. Imagine trying to stretch a rubber band. If you just give it a gentle tug, it stays pretty much its original size. But if you keep pulling, and pulling, and pulling… well, you get the idea. Certain chemical situations can cause those electron pairs to spread out, making the bond longer.
One of the main players in the realm of longer carbon-carbon bonds are compounds that are highly strained. Think of molecules that are forced into unnatural shapes, like tiny rings that are too small. The atoms are all crammed together, and to ease that tension, some of the bonds might stretch out a bit. It’s like trying to fit a large suitcase into a tiny locker – something has to give!
The Champion of Length: A Truly Stretched-Out Molecule!
So, who holds the crown for the longest carbon-carbon bond length? Drumroll, please… it’s often found in highly strained molecules, particularly those involving cyclic structures with very small ring sizes or with bulky groups attached that push the carbons apart. Scientists have synthesized compounds where the carbon-carbon bond is significantly longer than your average single bond. We're talking about lengths that are truly exceptional!
One remarkable example is found in certain highly strained cyclopropane derivatives. Cyclopropane itself is a three-membered ring of carbon atoms. It's already quite tense because the ideal angles for carbon bonding are much larger. When you add other atoms or groups to this already strained ring, you can further distort the geometry and, consequently, stretch out one of the carbon-carbon bonds. It’s like putting a tiny trampoline under those already stressed-out atoms, making them bounce a little further apart!

These exceptionally long bonds are not just a curiosity for chemists. They tell us a lot about how atoms interact under unusual conditions. They push the limits of what we thought was possible and help us understand the fundamental forces at play in molecules. It's a testament to the incredible diversity and adaptability of the chemical world.
Why Should You Care About a Stretchy Bond?
Okay, so a long carbon-carbon bond might not directly impact your daily commute or your grocery shopping. But understanding these microscopic marvels can actually make life a whole lot more fun and inspiring! It’s about appreciating the ingenuity of nature and the cleverness of scientists who figure this stuff out.
Think about it: the fact that chemists can design and create molecules with these extreme bond lengths is a testament to human curiosity and our drive to explore the unknown. It’s like discovering a new species of bird with an incredibly long beak – it’s fascinating and makes you wonder why and how!
This knowledge can spark your imagination. It can make you see the world around you with a renewed sense of wonder. That plastic bottle, that piece of wood, even the air you breathe – it’s all made of atoms and molecules, each with its own unique story. And sometimes, those stories involve incredibly stretched-out bonds!
It’s a reminder that even in the smallest, seemingly insignificant things, there’s a universe of complexity and beauty waiting to be discovered. It’s about embracing the fact that there’s always something new to learn, something more to understand. The world is a vast laboratory, and every atom is a potential discovery.
So, the next time you look at something made of organic material, or even just ponder the elements, remember the incredible flexibility of carbon. Remember that even the strongest-seeming connections can sometimes stretch to surprising lengths. It’s a beautiful metaphor for resilience, for pushing boundaries, and for the endless possibilities that exist when we dare to explore.
Keep asking questions. Keep exploring. The universe, down to its tiniest bonds, is an endless source of wonder. Who knows what incredible discoveries you’ll make when you start looking a little closer?
