Which Compound Forms An Acidic Solution When Dissolved In Water
Alright folks, gather ‘round, grab your lattes, and prepare yourselves for a tale of chemical intrigue, a story so juicy, so downright… sour… it’ll make your taste buds do a little jig. We’re diving deep into the bubbling cauldron of water, that H₂O stuff we all chug like it’s going out of fashion. But have you ever stopped to think about what happens when we toss other things into this aquatic party? Today, we’re talking about a special kind of guest, one that crashes the water’s chill-out session and turns it into a full-blown acidic extravaganza.
Now, when I say "acidic," don't go picturing some mad scientist cackling in a lab with bubbling beakers of doom. Think more like… a really tart lemon. Or that time you accidentally bit into a cranberry thinking it was a cherry. Yeah, that kind of acidic. We’re talking about a specific type of compound, a molecular mischief-maker, that, when it meets its watery match, decides to crank up the proton party. And the star of our show, the compound that throws the most epic acidic bash in water, is none other than… drumroll, please… a compound containing hydrogen that can readily donate a proton.
The Humble Hydrogen: Our Proton-Dropping Hero (or Villain?)
Let’s break this down, shall we? It all comes down to our old pal, hydrogen. This little guy, with just one proton and one electron, is like the energetic toddler of the periodic table. It’s always itching to get involved, to share, and sometimes, to just flat-out give away its electron. When hydrogen is part of a larger molecule, and it’s feeling particularly generous (or perhaps a bit misunderstood), it can decide to ditch its electron and become a positively charged ion, a lone ranger known as a proton (H⁺). Now, this is where the magic, or the sourness, happens.
Imagine water molecules (H₂O) are like a group of really friendly, slightly clingy friends at a party. They’re all holding hands, so to speak. When our proton-donating compound shows up, it’s like that one friend who’s always a bit too loud, a bit too… energetic. This hydrogen proton is looking for a new home, a new hand to hold. And wouldn't you know it, water is just full of opportunities!
The Watery Embrace: A Proton's New Best Friend
The oxygen atom in a water molecule is like the ultimate hugger. It’s got these extra electrons just waiting to be shared. So, when our generous hydrogen proton (H⁺) comes along, the oxygen in a nearby water molecule basically says, "Hey there, little fella! Come on in, the water’s fine!" And the proton, with its positive charge, finds the negatively charged parts of the water molecule incredibly attractive. It hops on board, forming a new, slightly more complex structure called the hydronium ion (H₃O⁺).
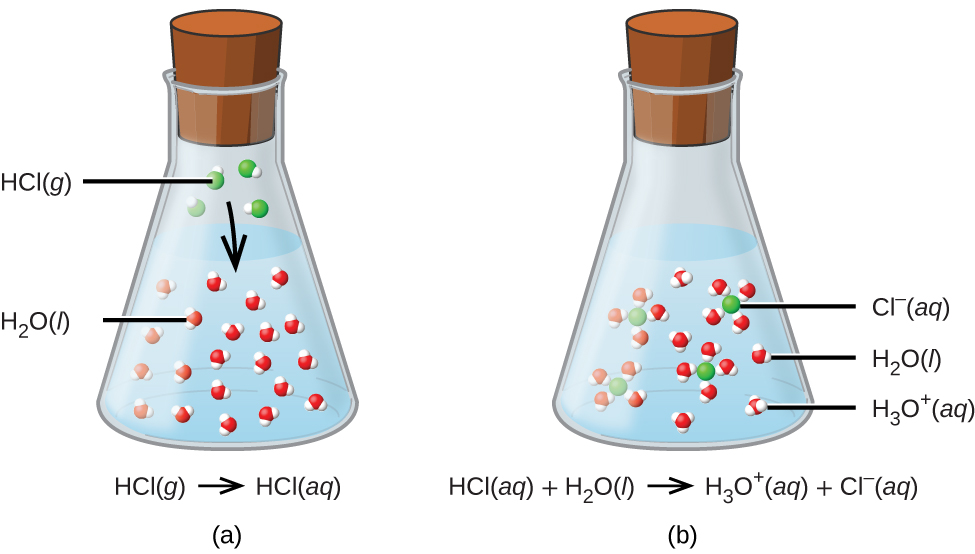
This formation of hydronium ions is the smoking gun, the tell-tale sign of an acidic solution. It’s like the party got so wild, there are now more of these hydronium characters milling about than your average water molecule. And these hydronium ions? They’re the ones responsible for that characteristic sour taste, that ability to make metal rust (slowly, don't worry, your spoon isn't going to vanish overnight), and that general "I mean business" vibe that acids give off.
It's All About the "Donation" Game
So, to be crystal clear, the key ingredient for forming an acidic solution when dissolved in water is a compound that has a hydrogen atom attached in such a way that it can easily split off as a proton. Think of it like a very precarious handshake. Some handshakes are firm and stable. Others? Well, they’re a bit wobbly, and one person might just let go unexpectedly. Our acidic compounds have those "wobbly handshake" hydrogens.
A classic example, and one you’ve probably encountered more times than you can count, is hydrochloric acid (HCl). When HCl hits the water, that hydrogen atom, feeling a bit lonely and extra positive, just poof! It becomes a proton. The chlorine, being a bit of a drama queen itself, happily takes the electron and becomes a negative chloride ion (Cl⁻). And that free-roaming proton? It’s off to find a water molecule to hug. Voilà! Instant acidity.

Not All Hydrogens Are Created Equal
Now, here’s a curveball: not every compound with hydrogen in it is an acid-maker. Take, for instance, good old methane (CH₄). It’s got hydrogen galore, right? But those hydrogens are happily shackled to the carbon in a very strong, very stable bond. They’re not going anywhere. They’re like the people at the party who are perfectly content just standing in the corner, observing. They’re not looking to donate their protons. So, methane in water? Nothing exciting happens. No acidic explosion, just… methane floating around, probably complaining about the music.
The trick is the type of bond. The hydrogen needs to be bonded to something electronegative, meaning something that’s really good at hogging electrons. Think of things like oxygen, nitrogen, or halogens (like chlorine, bromine, iodine). These elements pull the electrons in the bond closer to themselves, leaving the hydrogen a bit electron-deprived and, thus, more willing to bail and become a proton.

The pH Scale: Where the Acidic Party Gets Rated
And how do we know if we’ve achieved peak acidity? We consult the legendary pH scale! This is like the bouncer at the acidic club, telling you how wild the party is. A pH of 7 is neutral – think pure water, perfectly balanced, probably sipping on a non-alcoholic beverage. Below 7? You’re entering acidic territory. The lower you go, the more intense the proton party. A pH of 0? That’s like the mosh pit at an acid concert – extremely intense!
So, when you’re adding a compound that’s a pro at donating its hydrogen protons to water, you’re effectively increasing the concentration of those H₃O⁺ ions. And that, my friends, is what makes the solution acidic. It’s all about that proton donation, that willingness of a hydrogen to go solo and find a cozy spot with a water molecule. It’s a beautiful, albeit sometimes sour, dance of molecules.
So, next time you’re squeezing a lemon, or pouring vinegar (another acidic hero!), remember the silent, invisible drama unfolding. It’s a compound with a hydrogen ready to give up its electron, a water molecule ready to embrace it, and a whole lot of hydronium ions making things a little bit… zesty. And that, my friends, is the wonderfully acidic truth!
