Which Combination Of Compounds Will Create A Buffer Solution
Ever wondered what keeps your favorite shampoo from feeling like acid rain on your scalp, or how your body manages to stay at a stable internal temperature and pH? It's a bit like a chemical magic trick, and the secret ingredient is often something called a buffer solution. Learning about these amazing concoctions isn't just for science whizzes; it's a surprisingly fun and relevant peek into the world of chemistry that explains a lot of what goes on around us, and even inside us!
So, what exactly is a buffer solution, and why should we care? In simple terms, a buffer solution is a special mixture that can resist changes in pH when small amounts of acid or base are added to it. Think of it like a chemical shock absorber. Your body, for instance, has a sophisticated buffering system to keep your blood at a very specific, slightly alkaline pH. This is crucial for all sorts of biological processes to function correctly. Without buffers, even small fluctuations in acidity could cause serious problems.
The benefits are pretty clear: stability and control. In a lab, buffers are essential for experiments where precise pH conditions are needed for reactions to occur as expected. In industry, they are used in everything from manufacturing pharmaceuticals and dyes to keeping food products fresh and maintaining the proper conditions for growing cultures. Even in your own home, the gentle pH of skincare products and laundry detergents is often maintained by buffering agents. It’s this ability to maintain a consistent environment that makes them so valuable.
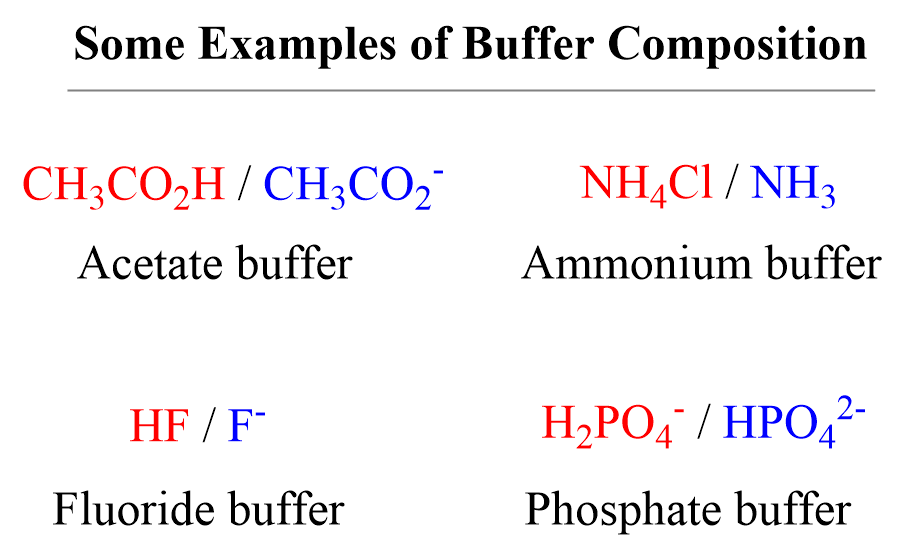
When it comes to the "which combination of compounds" question, the general rule of thumb is that a buffer is typically made from a weak acid and its conjugate base, or a weak base and its conjugate acid. For example, a common buffer system uses acetic acid (a weak acid) and sodium acetate (its conjugate base). Another example is ammonia (a weak base) and ammonium chloride (its conjugate acid). The key is that these two components are able to neutralize small additions of both acids and bases, effectively “buffering” the solution. It's like having a chemical team ready to tackle any incoming acid or base intruders!
Curious to explore this further? You don't need a full laboratory to get a feel for it. Sometimes, you can find everyday examples. Think about how some foods and drinks are formulated to have a certain tang or smoothness – that’s often related to pH and buffering. For a more hands-on (but still simple!) exploration, you could look into how different household liquids react to pH strips. While you won't be making your own buffers at home without proper guidance, understanding the principle behind them can make you a more informed consumer and a more curious observer of the chemical world.
