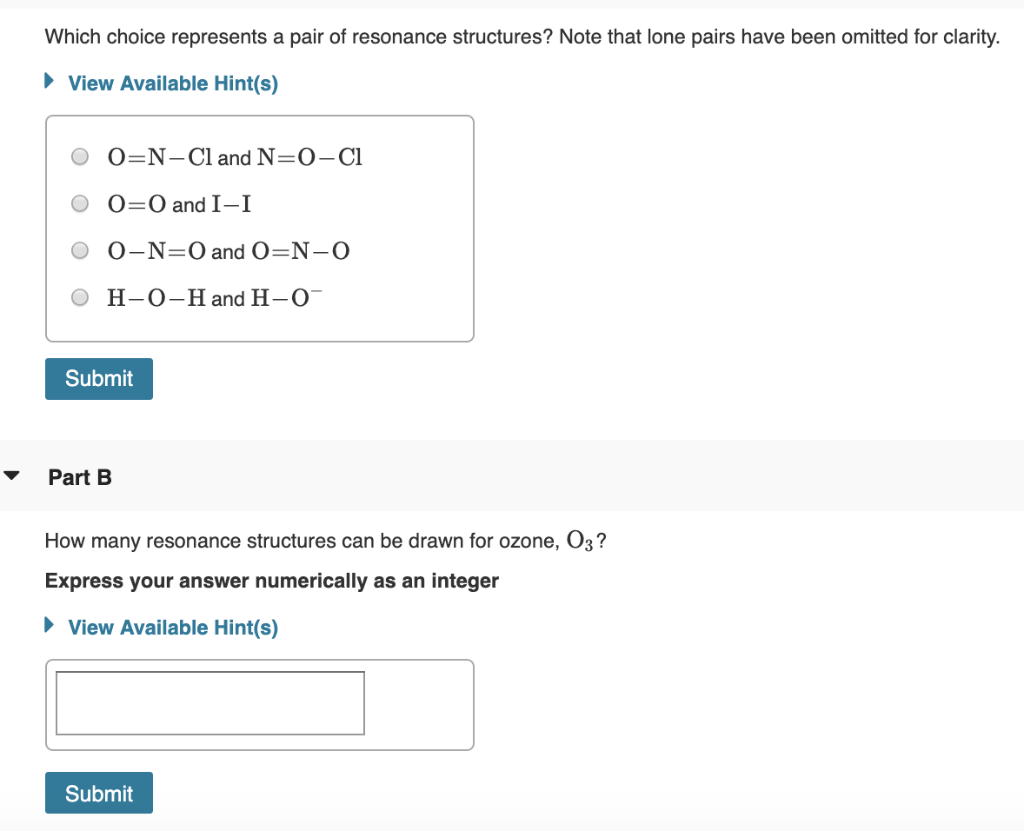
Which Choice Represents A Pair Of Resonance Structures

Hey there, fellow chemistry adventurers! Grab your mug, settle in, because we're about to dive headfirst into something super cool. You know those moments in chemistry class, when they throw around words like "resonance" and you're like, "Wait, what now?" It's okay, we've all been there. It's kind of like trying to explain a really good plot twist without giving away the ending – a little tricky, right?
So, let's chat about resonance structures. Think of it like this: sometimes, a molecule is just so extra with its electrons that it can't quite make up its mind where they should be. It's like having too many toys and not enough shelves! So, what does it do? It shares them around, moving them from one spot to another, creating what we call resonance structures. These aren't like separate molecules, nope. They're more like different pictures of the same molecule, trying to show you all the possibilities. It’s like a flipbook, but for atoms and electrons! How neat is that?
When you're staring at a bunch of chemical structures, and someone asks you to pick out the pair that are resonance structures, it can feel a bit like a scavenger hunt. But don't sweat it! There are some super handy clues to look for. It's all about spotting those delocalized electrons. They're the little nomads of the molecular world, zipping around. They're the real MVPs here, honestly.
So, what's the big deal with resonance? Why do we even care? Well, it turns out that molecules with resonance are often way more stable. Imagine trying to balance on one foot versus two. Two feet are just… more stable, right? Molecules with electrons spread out are like that. They're less likely to get into chemical kerfuffles. Plus, it explains a bunch of weird stuff we see in real life, like why certain bonds are exactly halfway between a single and a double bond. Magic? Nope, just resonance!
Alright, let's get down to the nitty-gritty. How do you actually spot a pair of resonance structures? It's not just about drawing squiggly lines willy-nilly. There are rules, of course. Think of them as the etiquette of electron movement. You can't just go around moving atoms; that would be chaos! We're only allowed to move electrons. Specifically, those pesky pi electrons, and sometimes lone pairs. Those are our main players in the resonance game.
The most important rule, the one you absolutely, positively must remember, is that the arrangement of atoms stays exactly the same. This is crucial! You're not bending the molecule out of shape or swapping atoms around. It's like rearranging the furniture in a room. You're still in the same room, just with the sofa over here and the lamp over there. The room itself hasn't changed its walls or windows. The same goes for resonance structures. The skeleton of the molecule, the arrangement of the atoms, remains identical.
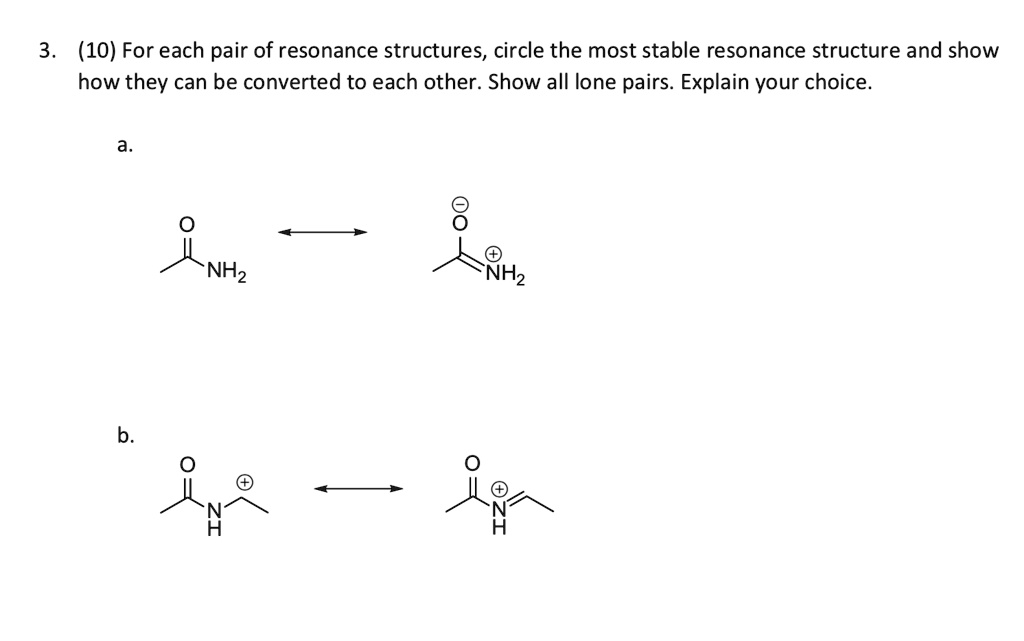
So, if you see two structures where the atoms are in different places, slam on the brakes! Those are definitely not resonance structures. They might be isomers, or something else entirely, but they're not part of the same resonance story. Think of it as a family photo. You want to see the same faces in the same general positions, maybe smiling a little differently, but the same people!
What else do we look for? Well, the total number of electrons must be the same in both structures. This is like counting the number of candies in two identical bags. If one bag suddenly has more candies, something's fishy. The electrons are conserved, my friends. They just move around, they don't magically appear or disappear. So, do a quick mental count, or even a literal one if you're feeling fancy, to make sure you've got the same number of little negatively charged buddies in each drawing.
And then there’s the fun part: the movement of electrons. Resonance structures differ only in the placement of double bonds and lone pairs. Imagine you have a double bond, like two friends holding hands. In a resonance structure, maybe one of those hands lets go, and a lone pair of electrons on a nearby atom decides to join the party, forming a new double bond. Or a double bond might break, and its electrons spread out as lone pairs. It's all about shifting those pi electrons and lone pairs. Never, ever, ever move a single bond to become a double bond without a corresponding change elsewhere, or vice versa, unless it’s part of a delocalization process. It’s a delicate dance, not a mosh pit!
You’ll often see a double-headed arrow (↔) between resonance structures. This is like the official stamp of approval. It’s the signal that says, "Hey, these two drawings are just different ways of looking at the same molecule!" If you see a single arrow (→) or something else, that's a different chemical reaction happening, not resonance. So, keep an eye out for that trusty double-headed arrow. It's your best friend in this quest.
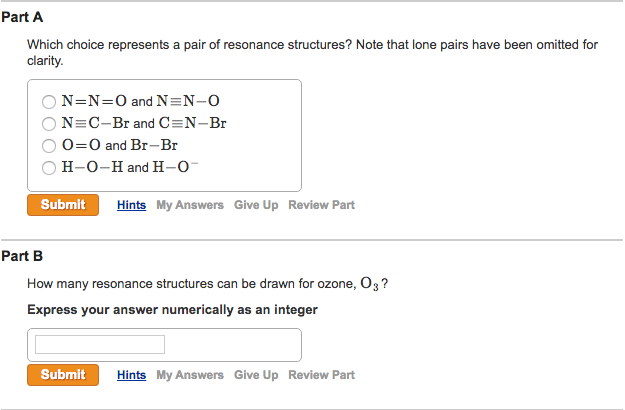
Let’s consider a classic example, shall we? The carbonate ion, CO₃²⁻. Now, this little guy is a prime candidate for resonance. If you draw it with one double bond and two single bonds to the oxygens, you might think one oxygen is special. But nope! The electrons are spread out evenly. So, you can draw the double bond between the carbon and the first oxygen, or the second, or the third. These three structures are resonance structures! They look different, sure, but the carbon-oxygen bonds are all identical in length and strength. They’re not quite a double bond, not quite a single bond. They're somewhere in between, thanks to resonance. The double bond is delocalized over all three oxygen atoms.
How do you know when to look for resonance in the first place? Usually, it happens in molecules with conjugated systems. That's a fancy term for alternating single and double bonds, or double bonds next to lone pairs, or double bonds next to positive charges. Think of it like dominoes. Once one domino is pushed, it can trigger a chain reaction. These systems are prime real estate for electron delocalization. If you see a double bond next to an atom with a lone pair, ding ding ding! You’ve likely got resonance potential.
Another big clue? When you draw a Lewis structure and have more than one reasonable way to do it, especially if you have formal charges that could be distributed better. Resonance helps spread out those charges, making the molecule happier and more stable. It's like sharing the load, you know? Nobody likes to carry all the weight.

Let’s talk about a common mistake. Sometimes people confuse resonance with tautomerism. Big difference, my friends! Tautomerism involves the movement of an atom, usually a hydrogen atom, and a change in the position of a double bond. Resonance, remember, only involves the movement of electrons. The atoms themselves do not move. So, if you see an H atom hopping from one place to another, that's not resonance. That's a whole other kettle of fish, a tautomeric fish!
When you're given a multiple-choice question, and you have to pick the pair of resonance structures, here's your strategy: 1. Check the atom positions. Are they identical? If not, move on. This is your first and most important filter. If the skeletons don't match, you're in the wrong place. 2. Count the total electrons. Are they the same? If not, nope. This is your sanity check. 3. Look at the electron movement. Does it involve only pi bonds and lone pairs? Are double bonds and lone pairs shifting positions? Are you using double-headed arrows between them? This is where the magic happens, or rather, where the electrons do their groovy dance. 4. Consider formal charges. Are the formal charges the same in both structures? Do they make sense with resonance? Resonance often helps minimize formal charges or spread them out onto more electronegative atoms. This is like the bonus round, the tie-breaker.
Imagine you’re looking at a picture. One picture shows your friend with their arms crossed, looking a bit stern. Another picture shows them with their arms open, smiling. They are still the same person, right? Their nose, their eyes, their hair – all in the same place. It’s just their pose, their state that’s different. Resonance structures are like those different poses of the same molecule. They represent the distribution of electrons, not a change in the molecule's backbone.
So, when you’re faced with that dreaded question, just breathe. Remember the core principles: identical atom positions, conserved electrons, and the delightful dance of pi electrons and lone pairs. These are your bread and butter. They’re your compass in the often-confusing world of electron distribution. It’s not about memorizing every possible resonance structure out there; it’s about understanding the rules of the game. Once you get those, you can spot resonance pairs like a pro!
Think of resonance as the molecule's way of saying, "I'm not stuck in one configuration! I'm flexible!" And that flexibility, that ability to spread out electron density, is what gives these molecules their extra oomph of stability. It’s like having superpowers, but for chemistry. Who wouldn't want that?
Sometimes, you'll see structures where a lone pair is next to a positive charge. That's another classic resonance scenario! The lone pair can jump over to form a double bond, neutralizing the charge. Or a double bond can break, and its electrons can go on a lone pair vacation on a positive atom. It’s all about achieving a more even distribution of charge and electron density. A molecule with all its electrons neatly tucked away in single bonds is stable, sure. But a molecule with its electrons spread out, like a cozy blanket over several atoms? That’s super stable. Think of it as a distributed network versus a single, vulnerable point. The network is just harder to disrupt.
And don't forget about resonance in aromatic compounds like benzene! That famous hexagonal ring with the circle in the middle? That circle is the ultimate representation of resonance. It signifies that the six pi electrons are completely delocalized around the entire ring, making benzene incredibly stable. Those alternating double and single bonds we sometimes draw are just snapshots of one possible electron arrangement, but in reality, they're constantly shifting, and the electrons are shared equally by all six carbons. It’s a beautiful, harmonious electron symphony!
So, the next time you’re presented with a set of chemical structures and asked to identify resonance forms, just follow your instincts, armed with these trusty rules. Look for the subtle shifts, the electron migrations, the dance of the double bonds and lone pairs. It’s all about understanding that molecules are not always rigid, static entities. They can be fluid, dynamic, and incredibly versatile. And that, my friends, is the beauty of resonance. It's where chemistry gets a little bit magical, a little bit like a choose-your-own-adventure story for electrons. Happy hunting!
