Which Chemical Bond Most Likely Stores The Most Energy

Hey there, science curious folks! Ever wondered what holds everything together? Like, what’s the secret sauce that makes your toys, your food, even YOU, exist? It all comes down to tiny, invisible connections called chemical bonds. Think of them like super-strong, microscopic glue that atoms use to stick to each other. Pretty neat, right?
Now, these bonds aren't all created equal. Some are like a gentle handshake, while others are more like a super-tight hug. And guess what? The tighter the hug, the more energy they're holding onto! It's like a secret energy piggy bank. We're going to dive into the world of these atomic friendships and see which one is the ultimate energy champion. Get ready to have your mind blown, just a little bit!
The Contenders: A Bonding Bonanza!
We've got a few main types of chemical bonds duking it out for the title of "Most Energetic." First up, we have the ionic bond. Imagine one atom is a bit of a sharer, and another is a bit of a hoarder. The sharer gives away an electron (a tiny particle that orbits the atom's center), and the hoarder snatches it up. This creates two charged-up atoms, one positive and one negative, and opposites attract! So, they stick together like magnets. It's a strong connection, for sure, but is it the strongest?
Next in the ring is the covalent bond. This is where things get really interesting. Instead of one atom giving away an electron and the other grabbing it, they decide to share! It's like two friends sharing a toy. They both get to play with it, and in doing so, they become linked. Some covalent bonds are like a casual sharing of a cookie, while others are more like two people holding hands really, really tightly. The strength of this sharing can vary, and that's where the energy game gets exciting.
And then there’s the metallic bond. Picture a whole bunch of metal atoms chilling together. They’re all so eager to share their outer electrons that they basically create a “sea” of electrons that all the atoms can swim in. This shared electron sea is what makes metals so good at conducting electricity and heat. It’s a bit like a giant, communal energy pool.
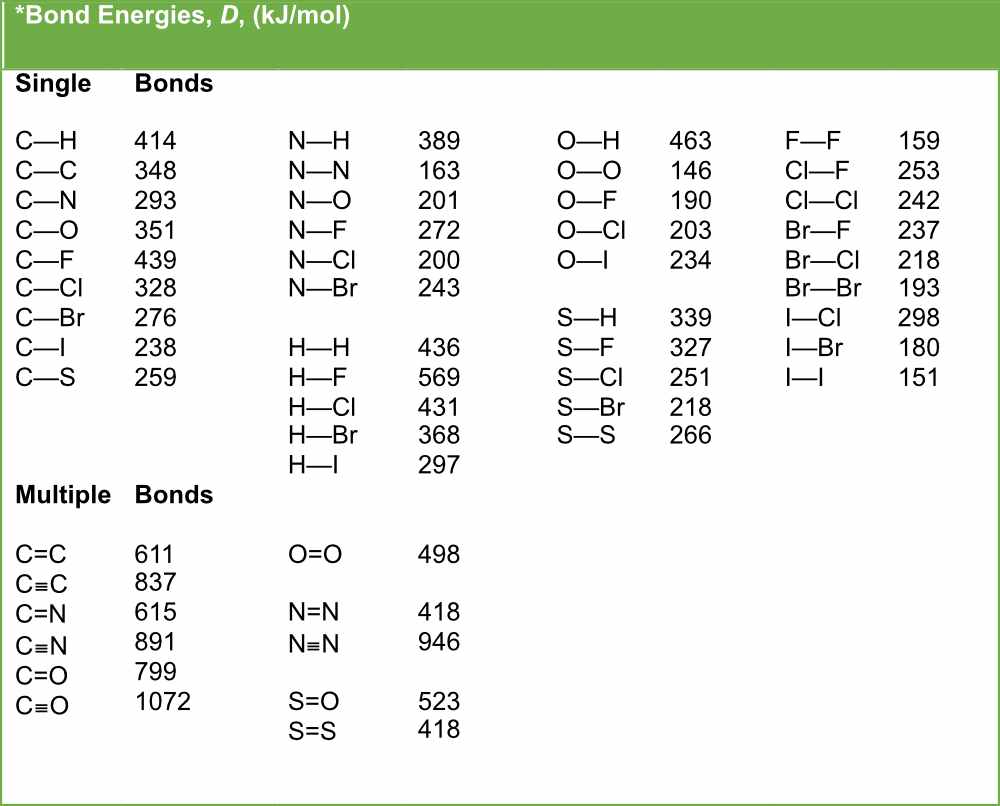
The Ultimate Energy Holder: Drumroll Please!
So, which of these atomic friendships is holding the most energy? While all bonds store energy, and breaking them often releases a burst of it (think of fireworks!), the real superstar for storing a significant amount of energy in a stable way often comes down to those sneaky, energetic electrons involved in covalent bonds.
Specifically, when atoms form really strong covalent bonds, they’re essentially locking up a lot of potential energy. Think about how much energy is packed into a tiny bit of fuel, like gasoline. That fuel is made of molecules with lots of these strong covalent bonds. When those bonds are broken in a controlled way, POOF! Lots of energy is released, powering your car or heating your home.
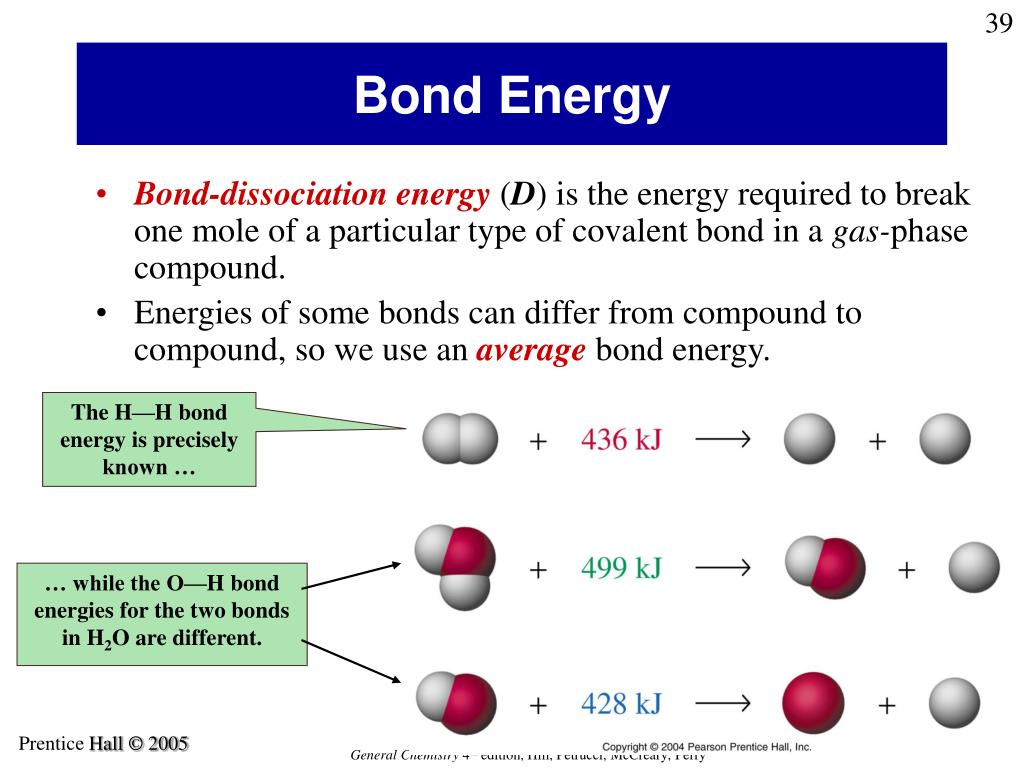
It's not just about the number of bonds, but also the type of atoms involved and how they arrange themselves. Some arrangements create particularly stable, energy-rich molecules. It’s like having a perfectly balanced, highly tensioned rubber band. It might not look like much, but stretch it, and it’s ready to snap back with gusto!
Why Is This So Cool?
Why should you care about which bond stores the most energy? Because it’s the engine behind so much of our world! The energy stored in covalent bonds is what powers life itself. When you eat food, your body breaks down the covalent bonds in that food to get the energy it needs to run, jump, and even think. Photosynthesis, the magic plants use to make their own food and the oxygen we breathe, is all about capturing and storing energy in covalent bonds.
It's also the reason we can have electricity. When we harness energy from burning fuels or even from breaking down materials in batteries, we're tapping into the energy stored in those chemical bonds. It’s like unlocking a treasure chest of power.
Imagine the molecules in a battery. They're holding onto energy in a very specific way through their covalent bonds. When you connect the battery to a device, you create a pathway for those bonds to change, releasing the stored energy as electricity. It’s a controlled release, not a sudden explosion, which is perfect for powering your phone or your flashlight.
The Magic of Molecules
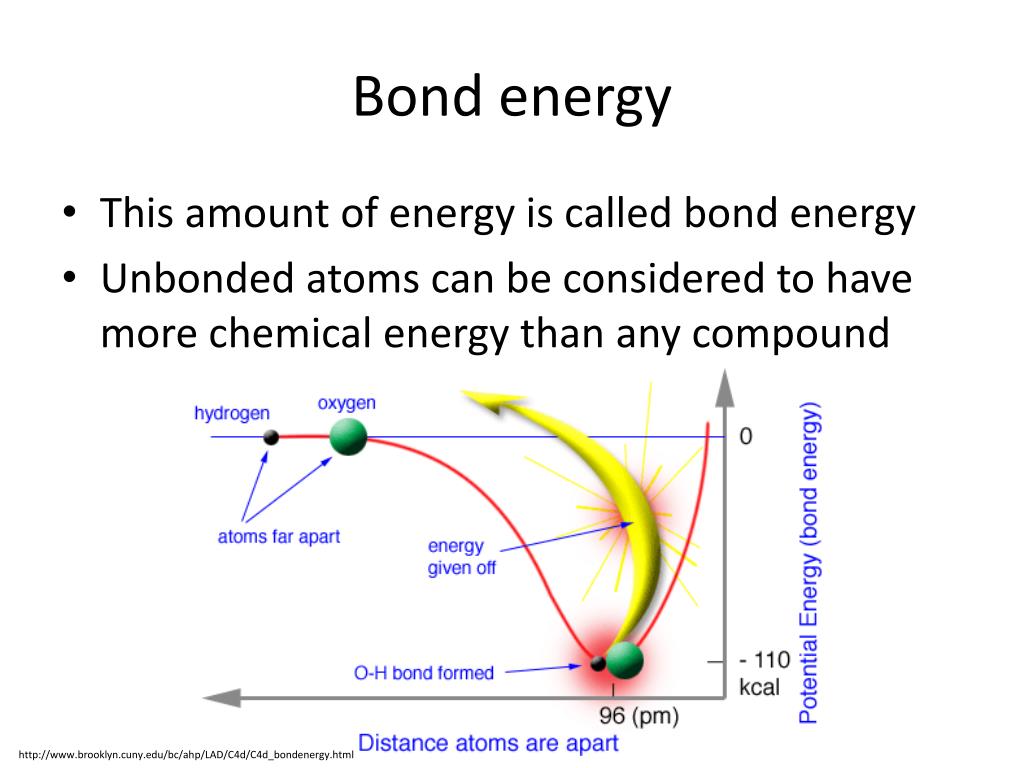
What makes covalent bonds so special in this energy-storage game is the precise way electrons are shared. It's a delicate dance. When atoms share electrons to form a covalent bond, they reach a state of lower energy than they were in as individual atoms. But the way they share and the specific atoms doing the sharing can lead to a very stable, yet energy-rich, arrangement. It's a bit like packing a very tightly wound spring. It's stable, but there’s a huge amount of potential energy just waiting to be unleashed.
Think about it: a single spark can ignite a whole log. That log is packed with energy thanks to the covalent bonds in its wood. That spark is just enough to get the reaction going, breaking those bonds and releasing all that stored energy as heat and light. It’s a dramatic demonstration of the power held within these seemingly simple connections.
So, the next time you see a flame, or feel the warmth from your body, or even just look at a piece of wood, remember the incredible power locked away inside. It's all thanks to the amazing, energetic world of chemical bonds, and especially the champions of energy storage: those versatile and powerful covalent bonds. They’re the unsung heroes that keep our world humming and buzzing with life and energy!
