Which Characteristics Describe Most Nonmetals In The Solid Phase

Hey there, science explorers! Ever peeked at the periodic table and wondered what makes those guys on the right side so… different? I’m talking about the nonmetals! Specifically, when they’re all chilled out and solid. It’s way more interesting than it sounds, I promise. Think of it as getting the lowdown on some of the coolest, and sometimes weirdest, elements around.
So, what's the deal with solid nonmetals? Are they all grumpy and brittle? Do they sparkle like jewels? Let's dive in!
They're Not Exactly "Shiny" Guys
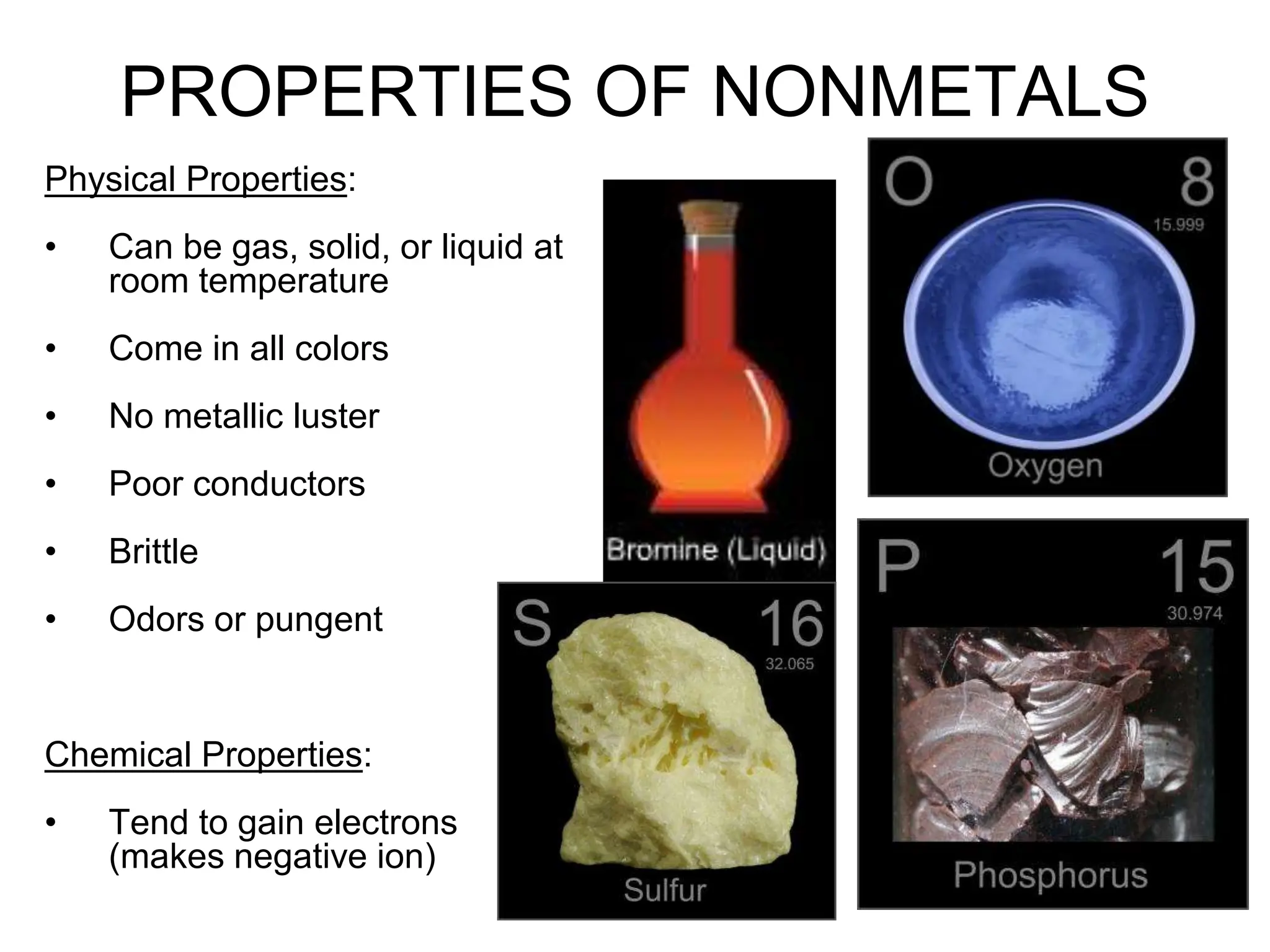
First things first, most solid nonmetals are like the wallflowers of the element world when it comes to shine. You won't find them reflecting light like a polished mirror. Think of sulfur, that bright yellow stuff. It's pretty, sure, but it's not exactly a disco ball. Or what about iodine? It's a deep purple solid, and if you heat it up a bit, it even turns into a funky purple gas, which is pretty neat! But shiny? Not its main gig.
Compare this to metals like gold or silver. They’re all about that gleam! Nonmetals, on the other hand, are usually more… matte. It’s like they’re saying, “Yeah, I exist, but I’m not trying too hard to impress you visually.” And honestly, that’s kind of cool in its own way. There’s a quiet confidence there, you know?
Brittle? Oh Yeah.
Here’s where things get a little dramatic. If you try to hammer a solid nonmetal, don't expect it to bend into a pretty shape. Nope. Most of them will just shatter. Imagine trying to shape a piece of charcoal. It just crumbles, right? That's the nonmetal vibe. They’re generally quite brittle.
This brittleness is a big clue about their atomic structure. Their atoms aren't linked up in that nice, orderly, flexible way that metals are. It’s more like a bunch of LEGO bricks that are stuck together, and if you push too hard, some of them just snap off. It makes them a bit delicate, but also means they behave very differently.
They're Not Great at Conducting Stuff
Okay, this is a huge one. Metals are famous for letting electricity and heat zip through them. They’re like the highways of the element world. Nonmetals? They’re more like the quiet country roads. They’re generally pretty poor conductors of both electricity and heat. Think about a wooden spoon versus a metal spoon in hot soup. The metal spoon gets scorching hot, right? The wooden one? Not so much.
This is why you see things like plastic (which is made of nonmetals) used for handles on pots and pans, and rubber (another nonmetal marvel) for electrical insulation. They’re keeping us safe and comfortable by not letting all that energy run wild. Pretty important job, wouldn't you say?
A Little Spark of Exception: Carbon!
Now, before you get too comfortable with the "poor conductor" rule, let's talk about carbon. Specifically, diamond. Yep, the sparkly gem! Diamond is pure carbon, and while it’s technically a nonmetal, it's actually a pretty decent conductor of heat. Not as good as a metal, but way better than, say, sulfur. And graphite? That’s also carbon, and it’s a fantastic conductor of electricity! Mind. Blown.
So, while the general rule is poor conductivity, carbon throws a fun little curveball. It just goes to show you, there are always exceptions to the rule, and those exceptions are often the most interesting part!
What About Their Colors? A Rainbow of Fun!
While they might not be shiny, solid nonmetals can boast some pretty wild colors. We already mentioned iodine's deep purple. Then there's that vibrant yellow sulfur. And let's not forget about phosphorus! White phosphorus is so reactive it can ignite just by touching air (which is why it's stored underwater – talk about a diva!). It's a waxy, white solid. Red phosphorus is more stable and is used in things like match heads. It’s a reddish-brown powder.

And then there’s the noble gases. They’re typically gases at room temperature, but when you zap them with electricity in a tube, they glow in all sorts of spectacular colors! Neon glows red, argon glows blue. While not technically solid at room temp, it’s a fun reminder of the colorful personalities within the nonmetal family.
It’s like a box of crayons, but way more scientifically significant. Each color tells a story about the element's electron arrangement and how it likes to interact with the world.
They're Not Exactly "Malleable" or "Ductile"
Remember how metals can be hammered into sheets (malleable) or drawn into wires (ductile)? Nonmetals? Not so much. We talked about brittleness, and that's pretty much the opposite of malleability and ductility. They just don't have that flexible, bendy quality.
This is why you won't find copper wires made of, say, selenium. Selenium is a reddish, brittle solid. Imagine trying to pull a selenium wire! It would just snap into pieces. Metals are great for building and conducting because they can be shaped. Nonmetals are great for other things because they can't be shaped in those ways.

They Often Form Molecules
Another quirky thing about nonmetals is how they tend to hang out together. They often bond with other nonmetals to form molecules. Think of water (H₂O). That’s two hydrogen atoms and one oxygen atom, all bonded together. Both hydrogen and oxygen are nonmetals!
Or how about the air you breathe? Nitrogen (N₂) and oxygen (O₂) are diatomic molecules – meaning they consist of two identical nonmetal atoms bonded together. This tendency to form molecules is a big part of why they behave so differently from metals, which often exist as giant, repeating structures of atoms.
The "Diatomic Dudes": A Special Crew
A fun little group within the nonmetals are the "diatomic dudes." These are elements that, in their natural state, exist as molecules of two atoms. We're talking hydrogen (H₂), nitrogen (N₂), oxygen (O₂), fluorine (F₂), chlorine (Cl₂), bromine (Br₂), and iodine (I₂). Even though some of these are gases at room temperature, in their solid forms (like solid iodine or solid bromine), they still exist as these paired-up molecules.
It’s like they’re best friends who never go anywhere alone. And it really affects their properties!

They're Often Found in Living Things!
Here’s a mind-blowing fact: many of the elements that make up you and me are nonmetals! Carbon, hydrogen, oxygen, nitrogen, phosphorus, sulfur – they’re the building blocks of life. We’re basically walking, talking bags of nonmetals, held together by some pretty amazing chemistry.
When you look at a piece of coal (mostly carbon) or think about the water you drink (H₂O), you’re seeing nonmetals in action. They’re not just abstract symbols on a chart; they’re fundamental to everything around us, and especially, to us!
So, Why Is This Fun to Talk About?
Because it’s all about understanding the world in a new way! It’s about noticing the differences and appreciating why they matter. The brittleness of sulfur, the non-shininess of carbon (in most forms), the way oxygen forms breathing molecules – these aren't just facts. They’re explanations for how things work. They’re the reasons why your electrical cords are safe, why your soup spoon gets hot, and why you’re even here!
Next time you see a lump of coal, or a bright yellow rock, or even just breathe in, remember the fascinating world of solid nonmetals. They’re not always the flashiest, but they are undeniably essential and endlessly interesting. Keep exploring!
