Which Characteristics Correctly Describe A Proton

Okay, so picture this: I’m in my dimly lit dorm room, probably late at night (shocking, I know), fueled by questionable instant coffee and the sheer terror of an upcoming chemistry exam. I’m staring at this textbook, and the word "proton" keeps popping up. Protons, protons, protons. It felt like I was in some kind of atomic sci-fi movie where everyone was whispering about these mysterious little guys. What ARE they, really? Are they like tiny, angry bumper cars of matter? Or are they more like… quiet, fundamental citizens of the atomic world? My brain felt like it was trying to juggle flaming bowling pins trying to grasp the abstract. You know that feeling, right? When you’re learning something totally new, and it’s like a foreign language, but with even less practical application for ordering pizza?
Anyway, after a good hour of flipping pages and muttering to myself (don't judge, it’s how I learn!), something finally clicked. It wasn't some grand epiphany under a starry sky, but more of a quiet "aha!" moment amidst the scent of old paper and desperation. Protons, it turns out, are pretty darn important. And, thankfully, not nearly as complicated as I was making them out to be. They’re the bedrock of so much of what makes up everything around us. Pretty cool, huh?
So, What's the Deal with Protons Anyway?
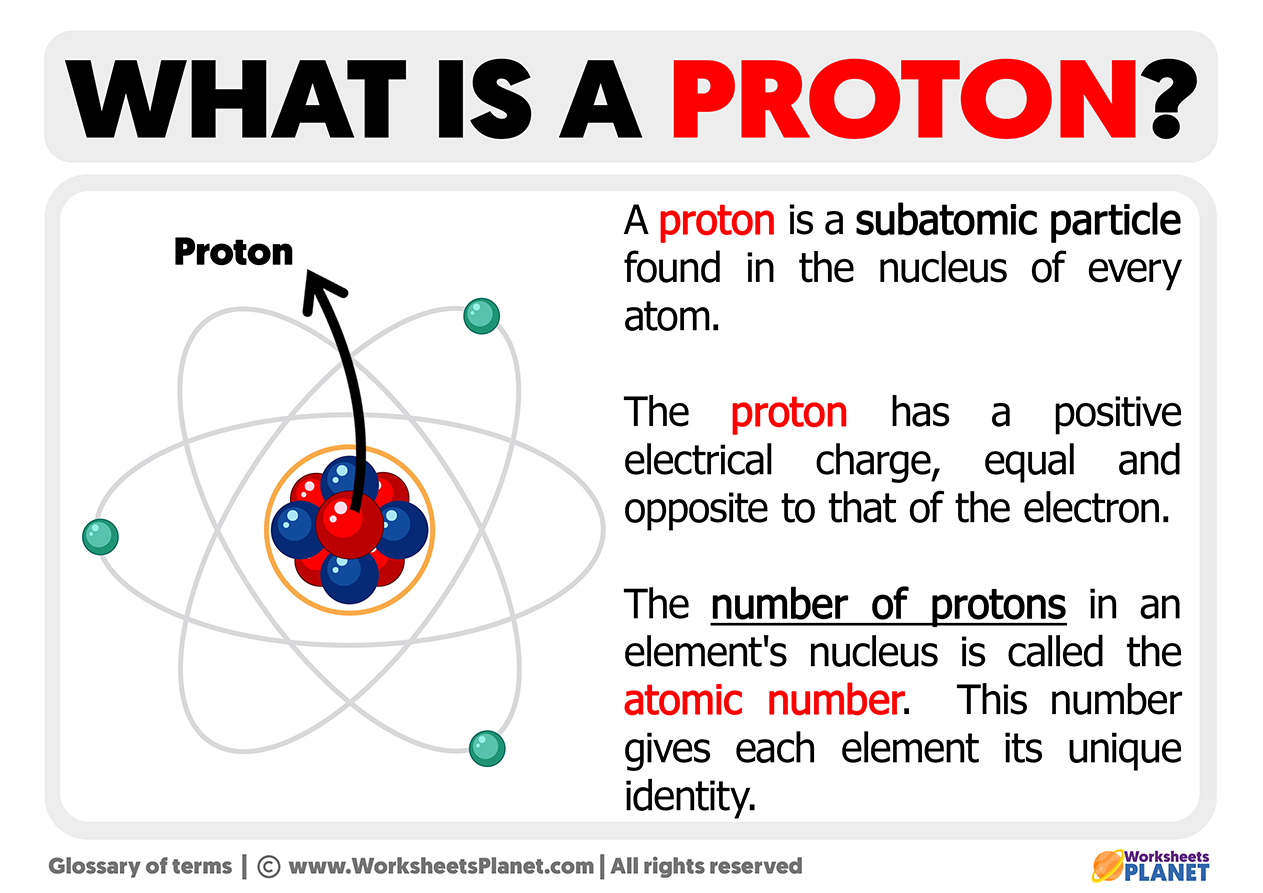
Let's break it down, shall we? Forget the sci-fi movie vibes for a sec. In the real, tangible, yet also incredibly microscopic world of atoms, a proton is basically one of the fundamental building blocks. Think of them as the VIPs of the atomic nucleus. You can't just have a random atom floating around without its protons. They’re kind of the identity card for an element.
This is where things get interesting. The number of protons in an atom’s nucleus is what defines what that atom is. Seriously. A hydrogen atom has one proton. A helium atom has two. A carbon atom, the backbone of life as we know it (and my keyboard, and my coffee mug, and probably that questionable stain on my rug), has six protons. If you change the number of protons, you don't just get a slightly different atom; you get a completely different element. It's like changing your name and suddenly you're a completely different person. Mind. Blown.
This is called the atomic number, and it's usually that little number you see floating above the element symbol on the periodic table. You know, the one you probably memorized a few of for that aforementioned chemistry exam? Yeah, that number. It’s literally just the proton count. Simple, right? Yet, utterly profound.
Charge It Up! The Positive Side of Things
Now, let's talk about one of the most defining characteristics of a proton: its electric charge. Protons are positively charged. Like, undeniably, unequivocally positive. Imagine tiny little cheerleaders for the universe, always radiating good vibes (and positive charge). This positive charge is super important because it’s what attracts the negatively charged electrons that zip around the nucleus. It's the cosmic dance of attraction that holds atoms together.
It's actually a very specific amount of positive charge, and it's the exact opposite of the charge of an electron. This is crucial for the atom to be electrically neutral overall. If an atom has, say, six protons, it needs six electrons to balance out that positive charge and be a happy, neutral atom. Think of it like a balanced scale. Too many protons, and it’s positively skewed. Too many electrons, and it’s negatively skewed. It’s all about that equilibrium, people!
The charge of a single proton is often represented by the symbol ‘+e’. It’s a fundamental unit of electric charge. So, when we talk about a helium atom having two protons, it means it has twice the positive charge of a hydrogen atom. This might seem like a minor detail, but it has massive implications for how atoms interact with each other, forming molecules and, well, everything else.
And here’s a fun little tidbit for you: if you ever hear about particles having a "fundamental charge," they’re usually talking about the charge of a proton (or the equal and opposite charge of an electron). It’s the smallest, indivisible unit of electric charge that we know of in this context. Kind of like the smallest LEGO brick for electrical interactions. Pretty neat, wouldn't you say?
Mass Appeal: They’re Not Exactly Featherweights
So, we’ve established that protons are positive identity markers. What else? Well, they also have mass. Big surprise, right? Everything that exists takes up space and has mass. But the interesting part about a proton's mass is how it compares to other subatomic particles.
Protons are significantly heavier than electrons. Like, way heavier. An electron is practically a fleck of dust compared to a proton. If an electron were a ping-pong ball, a proton would be a bowling ball. This difference in mass is one of the reasons why the vast majority of an atom’s mass is concentrated in its nucleus. That nucleus, packed with protons and neutrons (which are also quite massive), is the dense core of the atom, while the electrons are just… flitting around.
The mass of a proton is approximately 1 atomic mass unit (amu). This is a handy unit for atomic masses, and it’s defined relative to carbon-12. It's not a huge amount of mass in our everyday experience, obviously. You'd need a ridiculously, astronomically large number of protons to even register on a kitchen scale. But at the atomic level, that mass is substantial. It’s the stuff that contributes to the weight of, well, everything.
This mass is also a consequence of the subatomic particles within the proton. Yep, protons aren’t even truly fundamental in the deepest sense! They are made up of even smaller particles called quarks. Protons are made of two 'up' quarks and one 'down' quark. These quarks have their own masses and charges, and it’s the combination of them that gives the proton its overall characteristics. It’s like an onion, or those Russian nesting dolls, but with quarks. Still with me? Don’t worry, we’re not going to dive too deep into quark theory today. My brain is still recovering from the proton realization!
Location, Location, Location: The Atomic Heart
Where do you find these important little guys? They hang out in the nucleus of an atom. The nucleus is the central, dense core. Think of it as the atom’s brain or its heart. It’s where the action is, where the majority of the atom's mass resides, and where the protons (and neutrons, their equally massive, neutral buddies) are tightly packed together.
This nucleus is tiny! Compared to the overall size of the atom (which is mostly empty space occupied by the electron cloud), the nucleus is like a pea in a football stadium. It’s incredibly dense. And it’s positively charged, thanks to all those protons. This positive charge is what holds the negatively charged electrons in orbit around it. Without that central positive pull, the electrons would just… zoom off into the void. It's a delicate balance, a fundamental force at play.

The strong nuclear force is another key player here. It's an incredibly powerful force that binds the protons and neutrons together in the nucleus, overcoming the natural repulsion between the positively charged protons. Without the strong nuclear force, the nucleus would just fly apart. So, protons are not only charged and massive, but they’re also part of a super tight-knit group in the atomic center, held together by some serious forces.
Stability and Reactivity: The Backbone of Matter
So, are protons stable? Like, can they just hang around forever? For the most part, yes. Protons are considered to be extremely stable particles. Scientists have conducted experiments looking for evidence that protons might decay over vast timescales, but so far, no decay has been observed. This means that, for all practical purposes and for the age of the universe as we understand it, protons are here to stay. They are the dependable, enduring foundation of matter.
This stability is what allows elements to exist as they are. If protons were constantly breaking down, we wouldn't have the stable elements that form stars, planets, and us. It’s this longevity that makes them such a fundamental characteristic. They are the eternal residents of the atomic nucleus.
Now, while protons themselves are stable, the atoms they are a part of can be reactive. The reactivity of an atom is primarily determined by its electrons, specifically how many valence electrons it has and how they are arranged. However, the number of protons (the atomic number) dictates the element, and therefore the electron configuration, which in turn dictates chemical behavior. So, in a roundabout way, the proton's identity is intrinsically linked to the reactivity of the atom.

Think of it this way: a proton is like the architect's blueprint for a house. The blueprint itself (the proton) doesn't change, but the house built from it (the atom) can be designed in countless ways, leading to different structural strengths and appearances (reactivity). It’s a fascinating interplay between the core identity and the outward expression.
Quantum Quirks: Not Just Tiny Balls
And finally, because we’re talking about subatomic particles, we have to touch on the fact that protons are governed by the weird and wonderful rules of quantum mechanics. This means they don’t behave like little tiny solid balls in the way we might intuitively imagine. They exhibit wave-particle duality, meaning they can behave like both waves and particles depending on how you observe them.
This is where it gets mind-bending. It's not just that they're small; it's that their very nature is probabilistic and fuzzy. You can't pinpoint a proton's exact location and momentum simultaneously, thanks to the Heisenberg Uncertainty Principle. It's like trying to catch a ghost that's also a ripple in a pond. Sounds confusing? Yeah, it is! That’s quantum mechanics for you.
They also exist in specific energy states, and their interactions are governed by probabilities rather than deterministic paths. So, while we describe them as having charge, mass, and location in the nucleus, these are macroscopic descriptions of fundamentally quantum entities. They are fundamental constituents, but their behavior is anything but simple or classical. They are the enigmatic dancers of the quantum realm, fundamental to the structure of everything we see.
So, there you have it! Protons: positively charged, relatively massive, residing in the atomic nucleus, incredibly stable, and governed by the strange laws of quantum mechanics. They are the silent, powerful architects of the elements, and without them, the universe as we know it simply wouldn't exist. Pretty wild when you think about it, and a lot less terrifying than tiny, angry bumper cars. Though, sometimes, when I'm wrestling with a tough problem, I like to imagine them as tiny, determined cheerleaders for understanding. Keeps me going!
