Which Change Of State Has The Wrong Energy Change Listed

Hey there, science explorers! Ever find yourself staring at a science textbook, maybe during a late-night study session or just out of sheer, you know, curiosity, and wonder about the little quirks of the universe? Today, we're diving into something super neat, something that might make you do a double-take. We're talking about changes of state, those everyday transformations like ice melting into water or water boiling into steam. But here's the twist: we're going to sniff out a change of state where the energy change listed might just be a little bit… off. Let's get our mental magnifying glasses out!
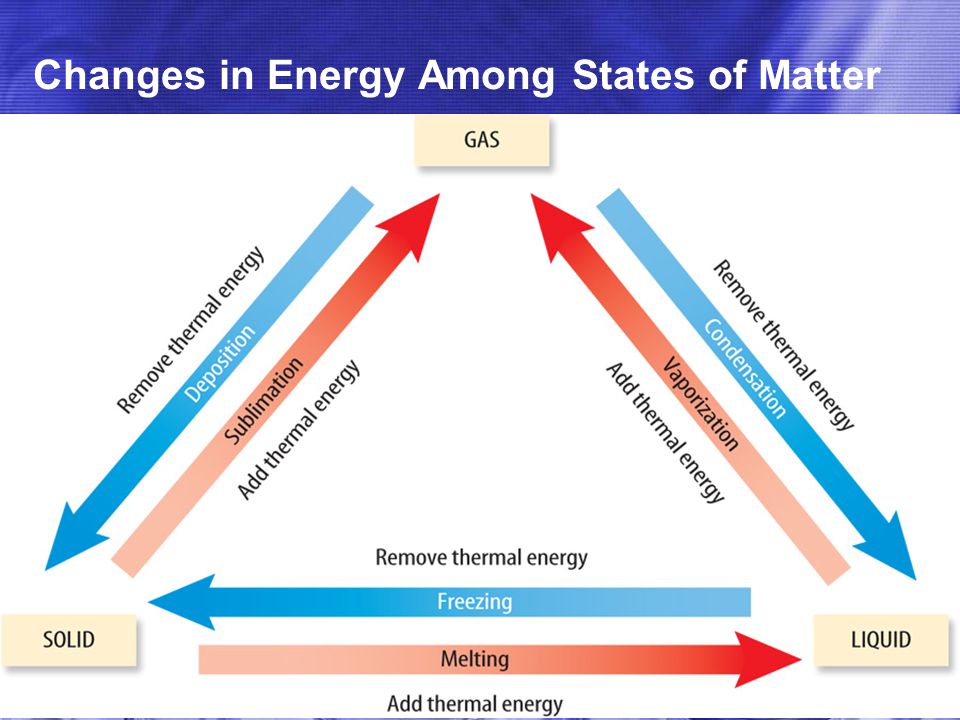
So, what exactly is a change of state? Think about it. It's when matter decides to switch its outfit, going from solid to liquid, liquid to gas, or even directly from solid to gas. These are the classic trio: melting, boiling, and sublimation. And their opposites exist too: freezing, condensation, and deposition. It's like water playing dress-up!
Now, the energy part. Every single one of these transitions requires energy to happen. Either you have to put energy in (like turning on the stove to boil water) or energy gets released (like when steam turns back into water on a cold windowpane). We call these endothermic (energy in) and exothermic (energy out) processes. Easy peasy, right? It’s like adding fuel to a car to make it go, or the car releasing heat as it runs.
But sometimes, things can get a little… confusing. Textbooks are usually spot on, of course, but imagine you’re looking at a list, and one of them just feels a bit… wrong. Like seeing a picture of a cat wearing a top hat and thinking, "Well, that's unexpected, but also kind of adorable!"
Let's consider our options. Melting, for instance. You take an ice cube, and it needs energy from its surroundings to become liquid water. It absorbs heat. So, melting is definitely endothermic. Your ice cream cone melting on a hot day? Yep, it's soaking up that sunshine energy. No surprises there.
Boiling is similar. Water needs a good amount of energy, a serious kick, to transform into steam. That's why your kettle takes a while to heat up. Boiling is also endothermic. It’s all about pumping that energy in to break those bonds and let the molecules spread out.

Sublimation, the cool one where solids go straight to gas (think dry ice turning into that spooky fog), is also endothermic. It’s like skipping a step in the dance – no liquid phase needed, just a direct jump from solid to gas, requiring a good chunk of energy.
Okay, so those are our energy-in processes. What about energy-out? Freezing is the opposite of melting. Water gets cold enough, and it releases energy to become ice. So, freezing is exothermic. Condensation, where steam turns back into liquid water (like those water droplets on your cold glass of soda), also releases energy. Exothermic. And deposition, the reverse of sublimation, where a gas turns directly into a solid (frost forming on a window), is also exothermic.
So, if all these seem to line up, where’s the potential for a mix-up? It's often in how we describe or quantify these changes. Let's think about the amount of energy. Some changes need more "oomph" than others.
For example, the energy required to melt one gram of ice is quite different from the energy required to boil one gram of water. Boiling takes way more energy than melting. It's like the difference between gently nudging a ball versus giving it a full-on cannon shot to get it moving in the same way.
Now, imagine a list that says something like: * Melting: Energy Released (Exothermic) * Freezing: Energy Absorbed (Endothermic) * Boiling: Energy Absorbed (Endothermic) * Condensation: Energy Released (Exothermic) * Sublimation: Energy Absorbed (Endothermic) * Deposition: Energy Released (Exothermic)
See that second line? Freezing: Energy Released (Endothermic)? That’s our potential culprit for a bit of confusion! Freezing, as we established, is the opposite of melting. If melting absorbs energy, then freezing must release energy. It's giving back what melting took. So, listing freezing as endothermic (energy absorbed) would be the "wrong energy change listed."

Why does this happen? Well, sometimes in our heads, we focus on the result. We see ice forming, and we might associate it with "getting colder," which feels like things are absorbing heat from the surroundings. But it’s the other way around! The water molecules are losing energy, and that energy has to go somewhere. It’s released into the environment. Think about how sometimes the surface around something that's freezing can actually feel a little warmer, just for a moment, as that heat is being expelled.
It's like when you’re packing for a trip. If you're taking things out of your closet to put in your suitcase, you're absorbing items. That’s endothermic. But if you're unloading your suitcase after the trip and putting things back, you're releasing items back into the closet. That’s exothermic.
Another way this could be presented incorrectly is by misstating the magnitude of the energy change. Imagine a table showing:

Energy Changes for Water
| Change of State | Approximate Energy Change (J/g) |
| Melting (Solid to Liquid) | +334 |
| Boiling (Liquid to Gas) | +2260 |
| Freezing (Liquid to Solid) | +334 |
| Condensation (Gas to Liquid) | -2260 |
The signs are crucial here. A positive sign (+) usually means energy is absorbed (endothermic), and a negative sign (-) means energy is released (exothermic). So, in this hypothetical table, the energy change for freezing is listed as +334 J/g. That positive sign suggests it's absorbing energy, which is incorrect for freezing. It should be -334 J/g, indicating energy is released.
This is where it gets really interesting! It highlights how precise we need to be with scientific terms and symbols. It's not just about knowing that water freezes; it's about understanding the energy flow involved. And that energy flow has a direction, just like a river flows downhill.
Why is this stuff cool? Because it explains so much about our world! Think about clouds forming. That's condensation, and it releases heat, influencing weather patterns. Or think about why sweating cools us down: the evaporation of sweat (a phase change from liquid to gas) absorbs heat from our bodies. If the energy changes were listed incorrectly, our understanding of these fundamental natural processes would be skewed. It’s like having a compass that sometimes points north and sometimes points south – you’re going to get lost!
So, next time you see melting ice, boiling water, or even frost forming, take a moment. Think about the energy. Is it being absorbed, or is it being released? And remember that little quirk where freezing, the act of becoming solid, is actually releasing energy back into its surroundings. It’s a subtle but significant detail, a small mystery solved in the grand, ever-unfolding story of science. Keep that curiosity alive, and you’ll find these fascinating tidbits everywhere!
