Which Best Describes Why Gel Electrophoresis Works

Hey there! So, have you ever wondered how scientists, you know, actually sort out those teeny-tiny bits of DNA or protein? Like, how do they tell them apart? It’s not like they have a microscopic sorting hat, right? Well, one of the coolest tricks up their sleeve is something called gel electrophoresis. And honestly, it’s not as scary as it sounds. Think of it like a really, really fancy sieve. But way more exciting, obviously.
So, what’s the big idea behind it? Basically, it’s all about size and charge. Those are the two main players in this whole science party. Imagine you’ve got a bunch of different-sized marbles, all mixed up. You want to sort them, right? Gel electrophoresis is kind of like that, but for molecules. Molecules are like the microscopic equivalent of those marbles, but with their own little electric personalities.
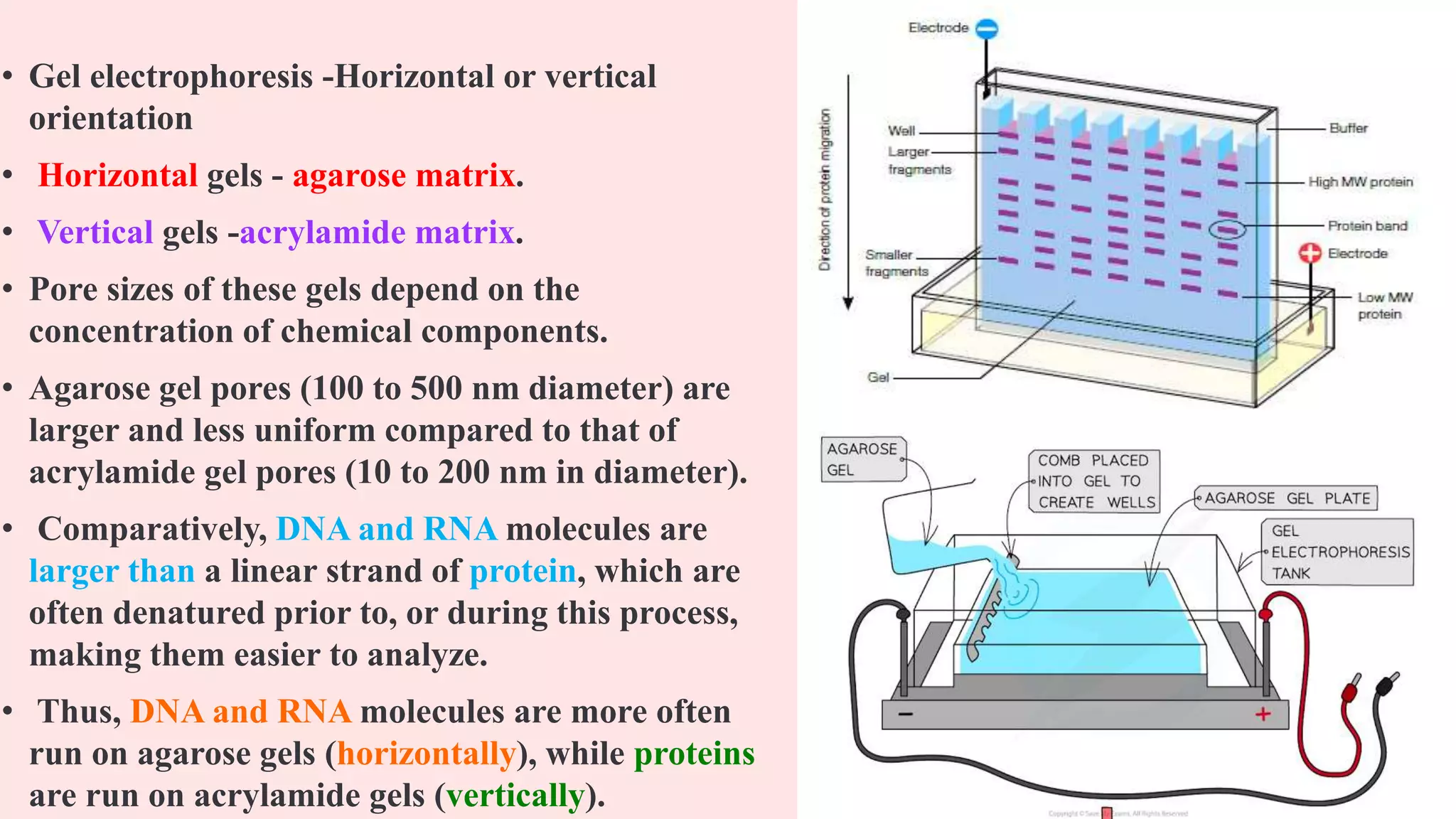
The "gel" part? That’s usually made from something called agarose. Think of it like a really, really weak jelly. It’s not for eating, sadly. This jelly has tons of little microscopic holes in it. Like, seriously, microscopic. So small, you can’t even see them with your naked eye. It’s like a maze designed by ants. And these holes are key. They’re the obstacles in our molecular race.
Now, for the "electrophoresis" part. "Electro" means electricity, obviously. And "phoresis" is a fancy Greek word meaning "to carry." So, it’s literally carrying things with electricity. Mind-blowing, right? We’re talking about making molecules move, using the power of a battery. Who knew batteries could be so useful for separating stuff? Besides, you know, turning on your TV remote.
So, picture this: we have our jiggly gel, right? We stick it in a special box. This box has two ends, and we connect it to a power source. One end is the positive pole (think of it as the "go here!" sign for certain molecules), and the other is the negative pole (the opposite direction, obviously). It’s like a tiny, scientific highway system.
Now, what are we trying to move? Usually, it’s DNA or RNA. These guys are negatively charged. Like, they’ve got a little electric frown on them. And why are they negative? Because they have these phosphate groups attached. Those phosphate groups are like tiny little negative magnets, always ready to repel anything positive. It’s a whole molecular drama happening!
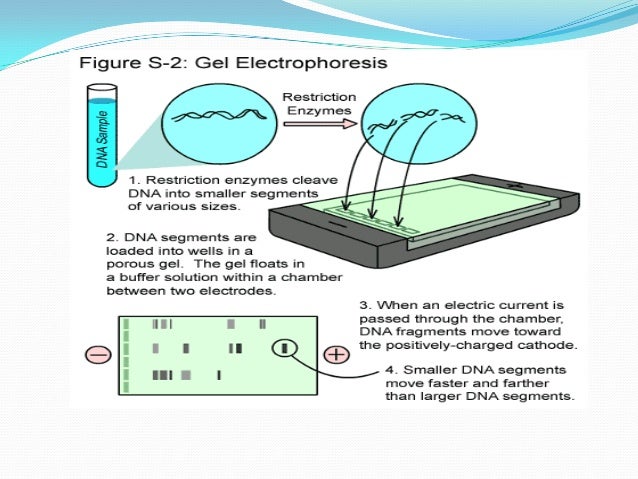
So, when we hook up the electricity, guess what happens? All those negatively charged DNA or RNA molecules, they get pushed away from the negative pole and pulled towards the positive pole. It’s like they’re suddenly being told, "Okay, time to move!" And they start to shuffle along through that agarose gel maze. They’re on a mission, folks. A mission to reach the positive side.
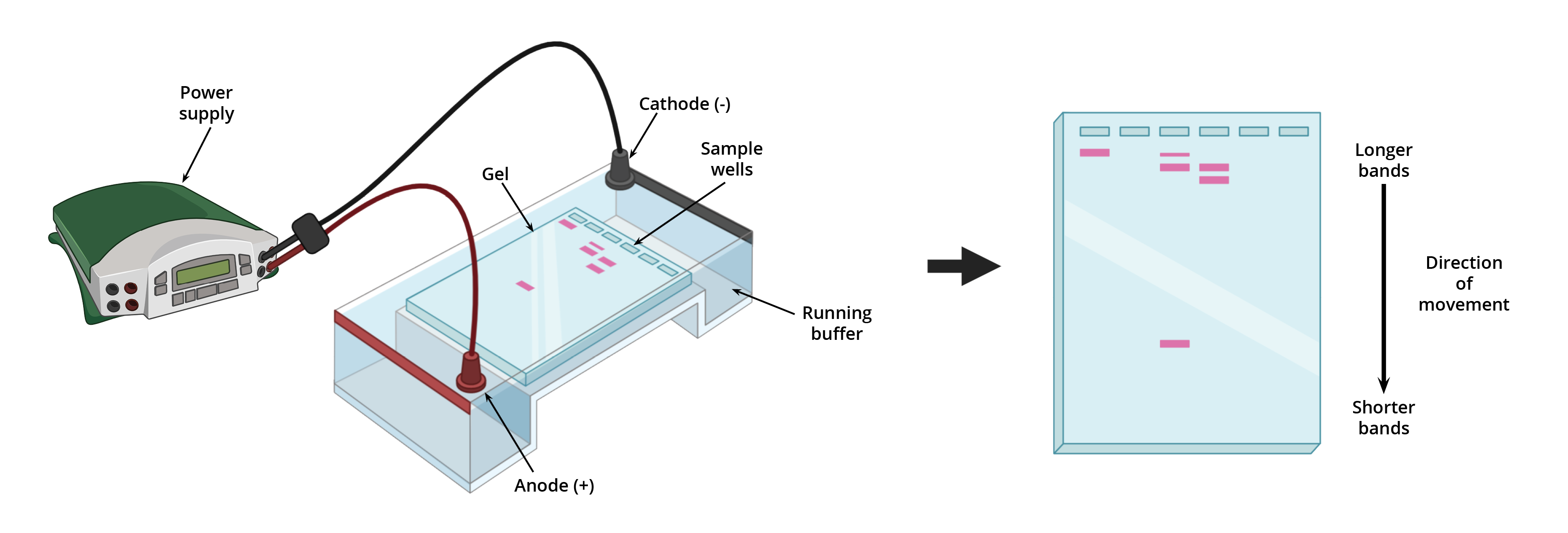
But here’s where the "size" thing comes in. Remember those microscopic holes in the gel? They’re not just for decoration. The bigger molecules? They’re going to have a really hard time navigating that maze. They’re like a bus trying to get through a tiny alleyway. They’ll get stuck, they’ll bump into things, and they’ll move really slowly. Think of them as the super-slow-motion runners in a race.
The smaller molecules, though? They’re like little speedy ninjas. They can zip through those holes with relative ease. They’re agile, they’re quick, and they’re going to get to the finish line (the positive pole) much, much faster. So, the gel acts as a sort of molecular sieve. It separates things based on how easily they can pass through its intricate network of holes.
So, at the end of our little electrophoresis experiment, what do we see? We don’t see individual molecules, of course. That would be way too easy, and frankly, a bit boring. Instead, we see bands. These bands are like little crowds of molecules that are all roughly the same size. All the fast-moving, small molecules end up clustered together at the bottom (closer to the positive pole). And all the slower-moving, bigger molecules are further up the gel (closer to the negative pole).
It’s like if you threw a bunch of different-sized glitter into a sieve. The tiny glitter would fall right through, and the bigger chunks would get stuck. Except, you know, on a molecular level. And with electricity. And science. It’s pretty neat, if you ask me. It’s like a tiny, silent concert where each instrument plays at a different speed, and we can see the different sections of the orchestra by their arrival time.

What about proteins, though? Can we do this with proteins too? Absolutely! Proteins are a bit more complicated. Some proteins are positively charged, some are negatively charged, and some are neutral. So, we can’t just throw them all in and expect them to go to the positive pole. That would be chaos. Pure protein pandemonium.
To make proteins behave, scientists often do a little trick. They use something called SDS-PAGE. Don't let the acronym scare you! SDS (sodium dodecyl sulfate) is like a detergent. It coats the proteins and gives them a uniform negative charge. So, even if a protein was naturally a bit positive or neutral, SDS makes it negative. Now, all the proteins are playing by the same charged rulebook.
And then, just like with DNA, the size of the protein becomes the main factor. The bigger proteins get bogged down in the gel maze, and the smaller ones zoom ahead. So, SDS-PAGE is another way of using gel electrophoresis to separate molecules based on their size. It’s a versatile technique, this gel electrophoresis thing. It’s not just a one-trick pony.
Why is this so important, you ask? Well, think about it. Scientists need to analyze DNA. Maybe they’re trying to identify a suspect in a crime scene, or figure out if a plant has a specific gene. Or maybe they’re studying diseases. They need to be able to see and sort these molecular components. Gel electrophoresis is like their trusty magnifying glass and sorting bin all rolled into one.
It’s also how they can tell if a gene editing experiment worked. Did they actually change the DNA? Gel electrophoresis can show them! It’s a fundamental tool in molecular biology. Without it, a lot of the amazing scientific breakthroughs we’ve seen just wouldn’t have been possible. It’s the unsung hero of the lab coat. The quiet, but essential, piece of equipment.
So, to recap, the main reason gel electrophoresis works is because it leverages two fundamental properties of molecules: their electric charge and their size. We use an electric field to make charged molecules move through a porous gel matrix. The gel acts like a sieve, with smaller molecules navigating it more easily and therefore traveling further, while larger molecules are impeded and travel shorter distances.
It's a beautifully simple concept, really. You have a matrix with pores, you apply an electric field, and things move. The key is that different sized things move at different speeds through that matrix. And for molecules like DNA and RNA, their inherent negative charge makes them perfect candidates for this electric "push." For proteins, we might need a little help from a detergent to give them that uniform charge, but the principle remains the same: size matters when you're navigating a microscopic obstacle course powered by electricity.
Think of it like a footrace. All the runners (molecules) start at the same line (the well in the gel). They're all trying to get to the finish line (the positive electrode). But the track is littered with tiny, uneven patches of mud (the pores in the gel). Some runners are super light and agile and can just skip over the mud patches. They’re the small molecules. Others are heavier and less coordinated. They get bogged down in the mud, tripping and stumbling. Those are the big molecules.
So, by the time they reach the finish line, they're not all there at once. The speedy ones are way ahead, looking smug. The slower ones are still trudging along, looking a bit dejected. And by looking at where the runners (or, in our case, the bands of molecules) are clustered, scientists can figure out which ones are the sprinters and which ones are the marathoners. It’s all about their speed, which is directly related to their size and how well they can navigate the gel.
It's like a science detective story, where the bands on the gel are the clues. Scientists can compare the bands from different samples, and if they look the same, it's a good sign they're dealing with the same type of molecule. If they look different, well, then something else is going on. It’s a way to get a molecular fingerprint, if you will. A way to identify and differentiate between these tiny building blocks of life.
And the cool part is that this isn't just theoretical mumbo jumbo. This is happening in labs all over the world, every single day. It’s what allows us to understand genetics, develop new medicines, and even trace our ancestry. It’s a cornerstone of modern biology. So, the next time you hear about DNA analysis or protein studies, remember the humble gel and the power of electricity. It’s a simple idea, but it has revolutionized our understanding of the microscopic world.
It really boils down to this: you’re using an electrical field to drive charged molecules through a porous matrix. Molecules of different sizes will move at different speeds through that matrix. The smaller ones go further, the bigger ones don't. And voilà, you have separation! It’s elegant, it’s effective, and it’s a fundamental tool for anyone trying to understand the intricate dance of molecules. Pretty awesome, right? Who knew a bit of jelly and a battery could do so much?
