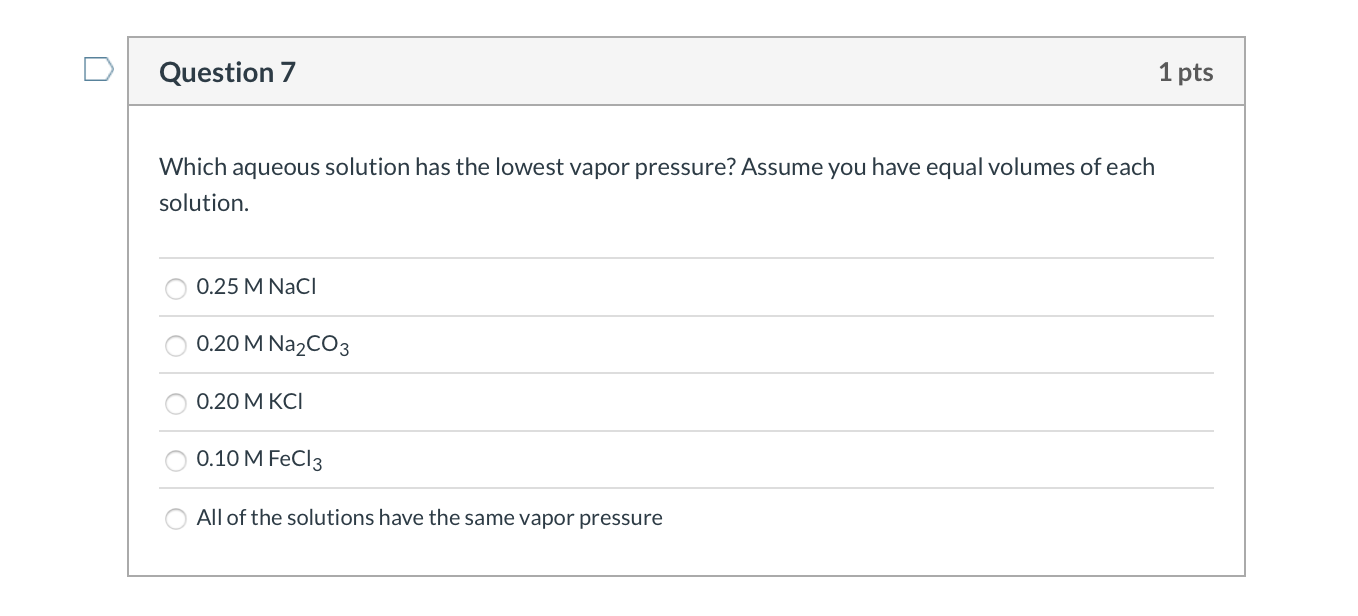
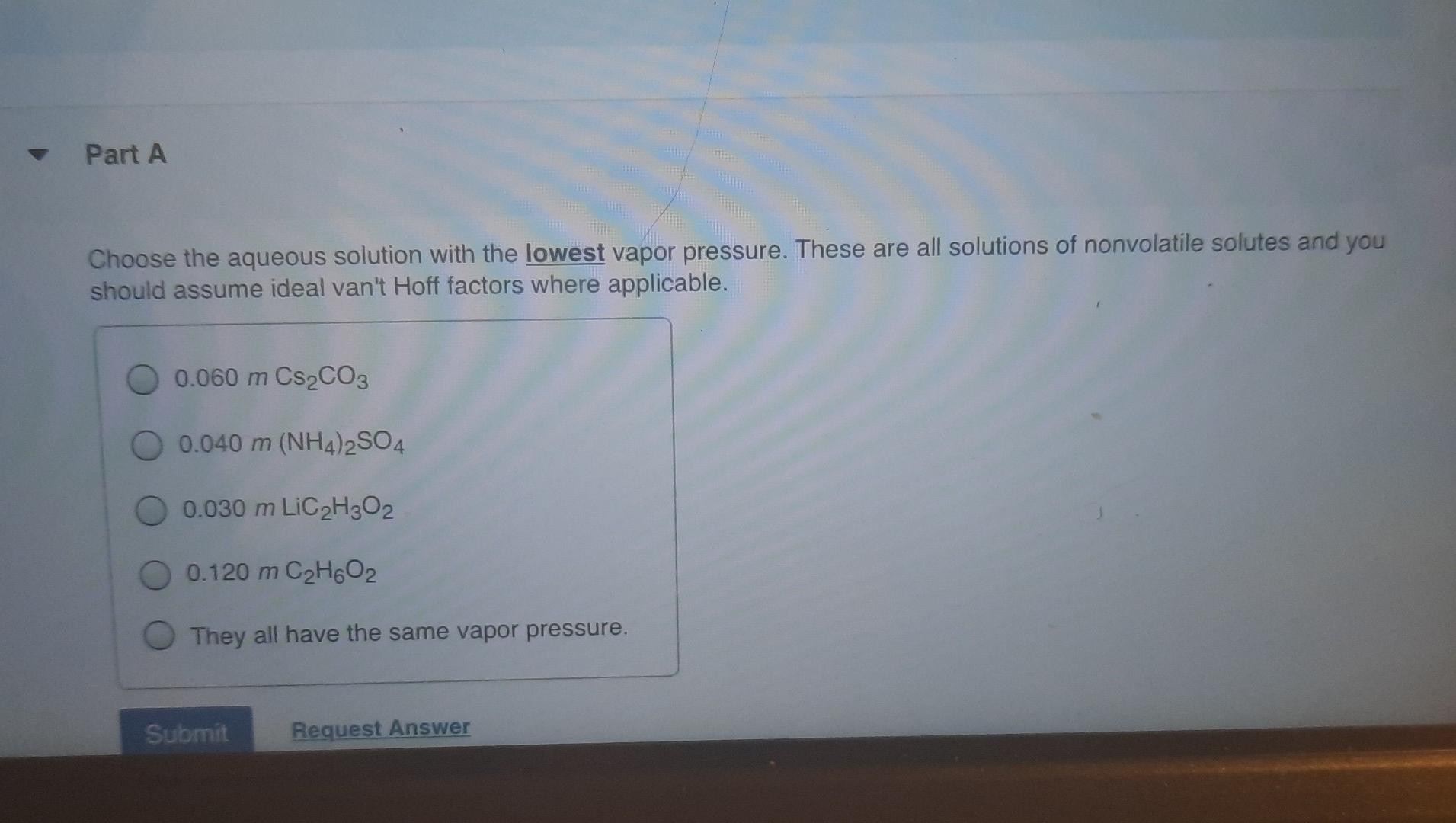
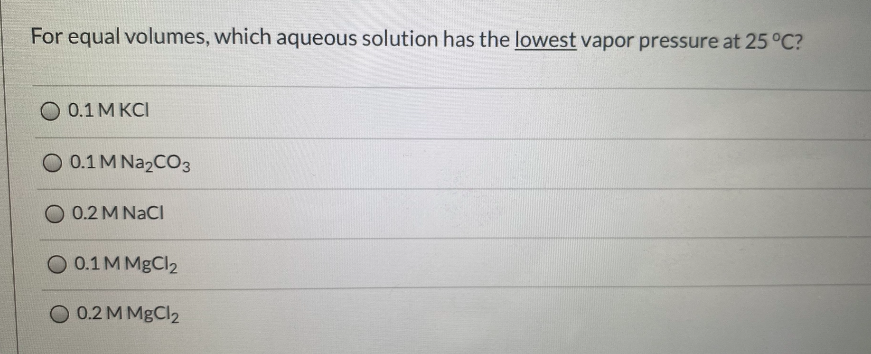
Which Aqueous Solution Has The Lowest Vapor Pressure
Let's talk about something that sounds super science-y but is actually kind of fun. We're diving into the mysterious world of aqueous solutions. Now, don't let that fancy term scare you. It just means things dissolved in water. Think of it as water that's having a little party with something else.
We're on a quest, a noble quest, to find out which of these water parties has the lowest vapor pressure. Sounds like something a detective would ponder, right? "The case of the vanishing water molecules!"
Now, you might be thinking, "Why on Earth would I care about vapor pressure?" Well, imagine your morning coffee. That lovely steam rising? That's vapor pressure at work. It's how much of a liquid likes to ditch its liquid form and become a gas, floating away into the air.
The lower the vapor pressure, the less the liquid wants to become a gas. It's like a stubborn teenager refusing to leave their comfy bed. It's just happier staying put.
So, we've got all sorts of things we can dissolve in water. We could have salty water, like the ocean on a really bad day. Or maybe sugary water, like when you make lemonade and forget to stir properly, and end up with a syrupy surprise at the bottom.
Then there are more exotic things. Maybe some fancy chemicals from a lab. But we're keeping it light and fun, remember? So let's stick to things we know. Like, what happens when you add salt to water? Does it make the water more or less likely to evaporate?

And what about sugar? That sweet stuff we love in our tea or cookies. Does that have an effect? My personal, totally unscientific, and probably wrong opinion? I suspect both salt and sugar are a bit of a drag on water's party spirit.
They kind of weigh down the water molecules. Like adding extra guests to a small dance floor. Suddenly, there's less room to boogie, and the whole vibe gets a bit more… contained.
So, our contenders for the lowest vapor pressure are likely to be solutions where we've added something that really likes to hang out with water. Not in a way that makes it go poof into the air, but in a way that makes it say, "Nah, I'm good right here."

Imagine a glass of pure water. It's pretty eager to become vapor. Think of it as a water molecule with wanderlust. It sees the sky and thinks, "Ooh, adventure!"
Now, add some salt. Those little salt ions are like bodyguards for the water molecules. They're hugging them close, saying, "Don't you go anywhere, buddy!" This makes it harder for the water molecules to break free and become vapor. So, the vapor pressure goes down. It’s like putting a damper on the water’s enthusiastic escape plans.
And then there's sugar. Sugar molecules are also pretty good at mingling with water. They’re like friendly conversationalists. They keep the water molecules busy chatting, which also makes it harder for them to decide it's time to become a gas. Again, the vapor pressure dips.
Now, what if we mixed both? A salty, sugary water concoction? Would that be the ultimate vapor pressure party pooper? My gut tells me yes. It's like having double the bodyguarding, double the conversation. The water molecules are practically glued to their dissolved buddies.
But here's where my unpopular opinion might kick in. I'm going to go out on a limb and say that pure water actually has the highest vapor pressure among the common solutions. It's the free spirit, the one who wants to explore. All these dissolved things are like anchors, holding it back.
So, if we're looking for the lowest vapor pressure, we need to pick a solution that's really good at keeping those water molecules grounded. And in my humble, non-expert opinion, the more stuff you dissolve in water, the lower that vapor pressure gets.
Think about a really, really concentrated sugar solution. Like, almost candy-making thick. That stuff barely evaporates. It’s like a liquid fortress. Or imagine the Dead Sea, which is super salty. That water is famously resistant to evaporation. It’s like the ocean’s grumpy uncle who just sits there and doesn’t budge.
So, my grand, possibly flawed, conclusion is this: The aqueous solution with the lowest vapor pressure is probably the one that has the most stuff dissolved in it, making it really hard for water molecules to escape.
We're talking about something like a super-concentrated salt solution or a ridiculously thick sugar solution. They've invited so many guests to their water party that there’s barely any room for the original host (water) to go have a solo adventure. They are the ultimate chillers, the masters of staying put.
It’s not the most exciting revelation, I know. There’s no dramatic explosion or sudden change of state. It’s just… less evaporation. But in the quiet world of liquids and gases, sometimes the biggest story is about who's not going anywhere.
And if you ask me, a solution that's packed with dissolved solutes, making it a reluctant evaporator, is the real champion of low vapor pressure. It's the introvert of the liquid world, and I kind of admire its commitment to staying put. So next time you see a puddle that's taking forever to dry up, you'll know it's probably got some dissolved buddies making it stay grounded. And that, my friends, is the low-down on low vapor pressure.
