Where Does Cleavage Of The Peptide Bond By Chymotrypsin Occur
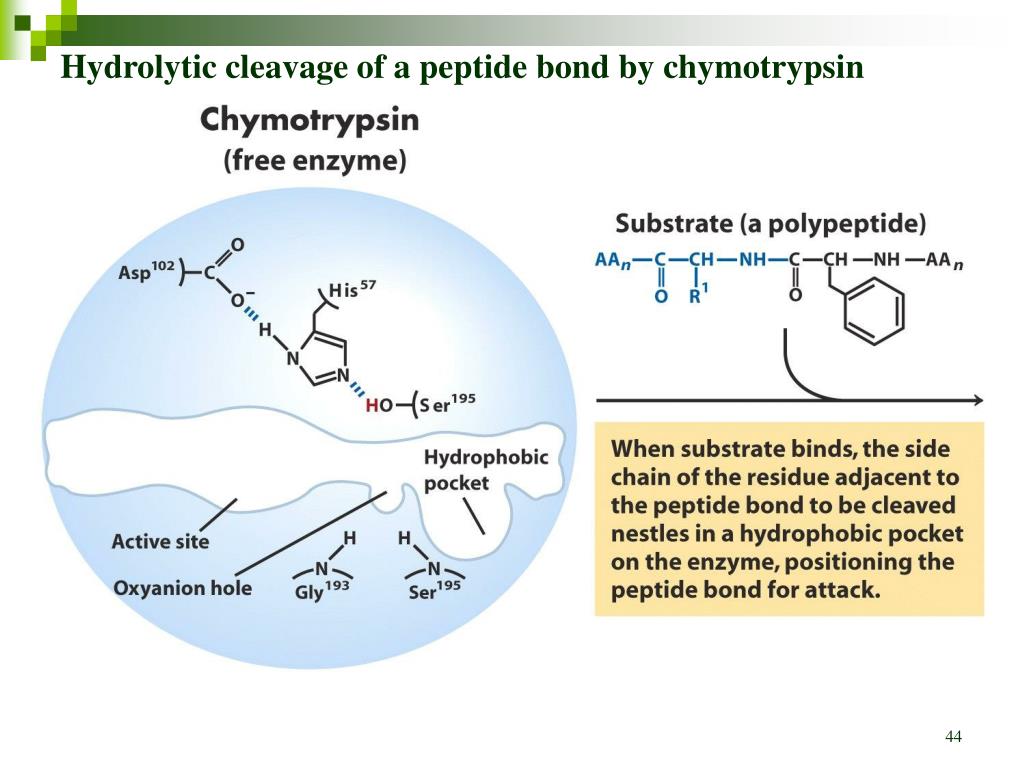
Alright, gather 'round, folks! Imagine this: you're at a swanky dinner party, a real shindig, and the star of the show isn't the tiny quiches or the suspiciously glittery champagne. Nope, it's a protein. Proteins, those magnificent molecular machines that build our muscles, carry oxygen, and probably gossip about us in our cells. Now, sometimes, these proteins need a little… trimming. Like a fancy haircut, but with enzymes. And today, we're talking about a particularly posh stylist: Chymotrypsin.
Think of chymotrypsin as the VIP of protein chopping. It’s not just slicing and dicing willy-nilly. Oh no, this enzyme has standards. It’s like a sommelier for peptide bonds, only instead of wine, it’s sniffing out specific amino acids. And where does this discerning enzyme like to set up its shears? Well, it’s all about the aromatics, my friends!
So, what's a peptide bond, you ask? Imagine proteins as long necklaces of beads. Each bead is an amino acid. The peptide bond is the strong string holding those beads together. Chymotrypsin’s job is to find that string and snip it, but only under very specific circumstances. It’s not just any old string it’s after.
Picture this: our protein necklace has some beads that are a bit… fancy. They're the ones with rings! Like a little, aromatic halo around them. These are our key players: tyrosine, tryptophan, and phenylalanine. If you see one of these ring-wearing beauties on your protein necklace, chymotrypsin’s ears perk up. It’s like it smells a five-star meal from a mile away.
But here’s the kicker, and this is where chymotrypsin gets really pick-y. It doesn’t just cut any peptide bond. It’s only interested in the bond immediately following one of these aromatic amino acids. It’s like saying, "Okay, I see that fancy bead? I’m going to cut the string right after it." It’s a very precise operation, not a chaotic protein massacre. We’re talking surgical precision, folks, with a side of molecular snobbery.

Think of it as a bouncer at an exclusive club. The bouncer (chymotrypsin) is standing at the door (the peptide bond). And only people wearing a certain type of accessory (tyrosine, tryptophan, or phenylalanine) are allowed to pass through that specific exit. Anyone else? Tough luck, you gotta stay in line!
Now, there’s a tiny little caveat, a microscopic loophole in this aromatic regime. Sometimes, the ring-wearing amino acid might be followed by another amino acid with a small, aliphatic side chain – specifically, methionine. Chymotrypsin is still happy to cut after tyrosine, tryptophan, and phenylalanine in this case. But if it's lysine or arginine right after our aromatic friend? Uh-uh, no cutting allowed. Chymotrypsin slams the door shut. It’s like a strict dress code: "No velvet ropes for these guys!" It’s a bit of a mystery why it’s so picky about those two, but hey, who are we to question a molecular maestro?
So, where does all this dramatic snipping happen? Well, it happens in your small intestine. Yes, that's right! This sophisticated protein-chopping action is crucial for digestion. When you eat that juicy steak or that protein-rich lentil soup, the proteins are all bundled up, like tightly packed suitcases. They’re too big and complex to be absorbed into your bloodstream.
+Hydrolytic+cleavage+of+a+peptide+bond+by+chymotrypsin..jpg)
Enter chymotrypsin, our digestive superhero! It’s secreted by your pancreas in an inactive form called chymotrypsinogen. This is a smart move, because you don’t want your pancreas to start chopping up itself, do you? That would be a real downer. Once chymotrypsinogen gets into the small intestine, a little nudge from another enzyme (we call it trypsin, a distant cousin) wakes it up and turns it into the active, sharp-suited chymotrypsin we know and love.
And then, the feast begins! Chymotrypsin gets to work, snipping those long protein chains into smaller, more manageable peptide fragments. These smaller pieces can then be further broken down by other enzymes into individual amino acids, which your body can then absorb and use for all sorts of amazing things. Building muscles, making enzymes, you name it!
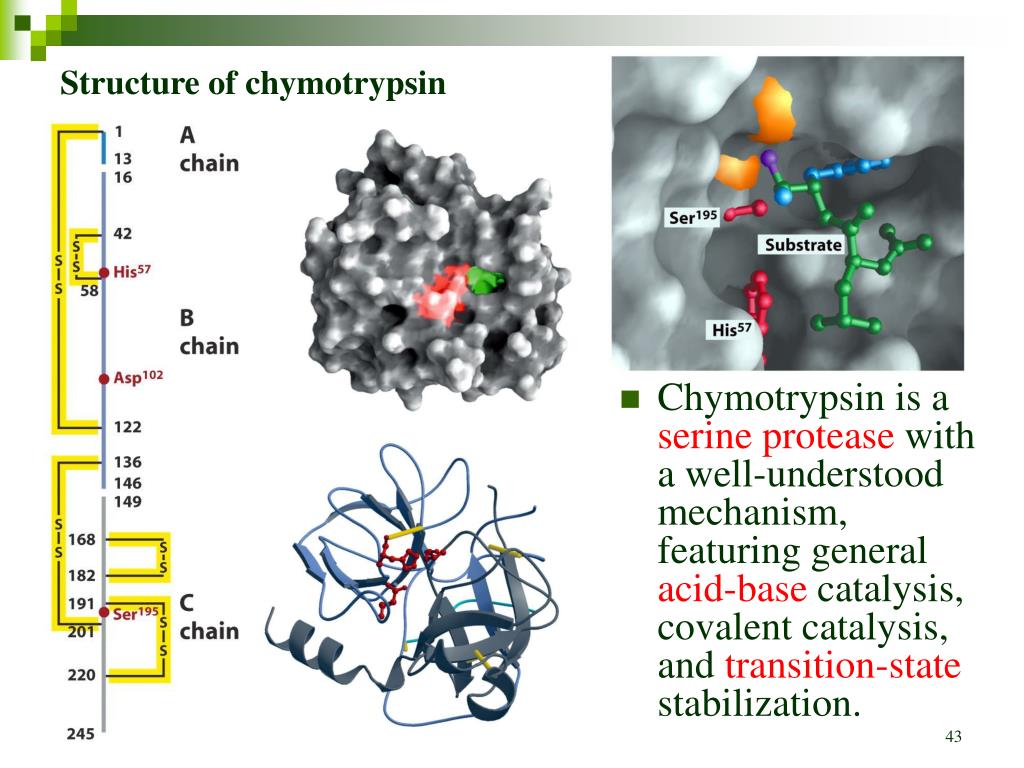
It’s a fascinating dance of molecules, isn't it? This whole process highlights the incredible specificity of enzymes. They don't just wander around randomly breaking things. They have highly evolved active sites, like molecular puzzle pieces, that are designed to fit and interact with specific substrates. Chymotrypsin’s active site is perfectly shaped to accommodate those aromatic side chains and guide the peptide bond to its doom.
Think about the sheer number of peptide bonds in a typical protein. Billions upon billions! Yet, chymotrypsin will only make its mark in a few very specific locations. It’s like a master locksmith, only unlocking certain doors on a massive, intricate building. And all because of a little bit of aromatic flair.
So, the next time you're enjoying a delicious meal, take a moment to appreciate the silent, invisible work of chymotrypsin. It’s out there in your gut, with its sophisticated tastes and precise scissors, making sure you get the most out of your protein. It’s a tiny enzyme with a big job, and it does it with an elegance that frankly, most of us could only dream of. Who knew digestion could be so… chic?
