When Two Monosaccharides Undergo A Dehydration Synthesis

Hey there, sugar lovers! Ever wondered what’s really going on when you enjoy that sweet scoop of ice cream, a piece of fruit, or even just a sprinkle of sugar in your coffee? It all boils down to some seriously cool chemistry happening at the tiniest level. Today, we're diving into a process that sounds a bit fancy but is actually super important and, dare I say, a little bit magical: dehydration synthesis when two monosaccharides get together.
Now, "monosaccharide" is just a fancy word for a simple sugar. Think of them as the individual LEGO bricks of the sugar world. The most famous ones you probably know are glucose (the fuel for your body's engine) and fructose (the sweet stuff found in fruits). They're like two best friends, just chilling, doing their own thing.
When Two Become One (and Lose a Little Water!)
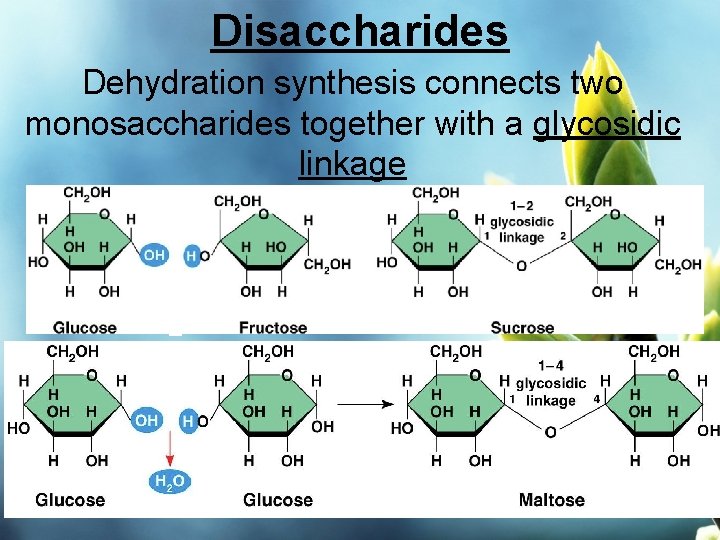
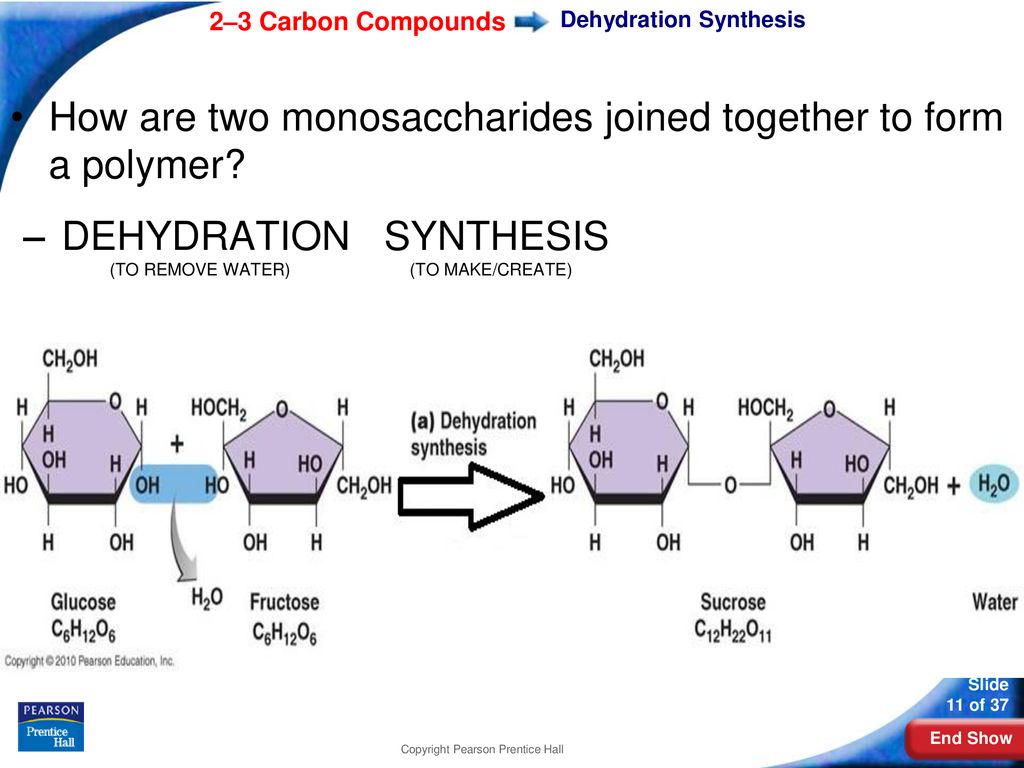
So, what happens when these two simple sugar friends decide to team up? Well, they can join forces to create something bigger and, frankly, more useful. This joining process is called dehydration synthesis. Don't let the big words scare you; it's actually quite intuitive. "Dehydration" means losing water, and "synthesis" means to make or build something. So, essentially, these two sugars build a new, bigger sugar molecule by shedding a water molecule in the process.
Imagine two kids, Timmy and Sally, each holding a delicious gummy bear. They decide they want to share and make one super-gummy-bear. To do this, they have to, you know, connect. As they press their gummy bears together to form a bigger, slightly less individual gummy bear, a tiny bit of imaginary "gummy dust" (which is like our water molecule) pops off. It's not exactly the same, but you get the idea! Two smaller things combine to make a bigger thing, and a little bit of something extra is released.
The Sweet Results: Disaccharides!
When two monosaccharides link up, they form a disaccharide. This is where things get really interesting for us, because disaccharides are the sugars we commonly consume and recognize. You've definitely met these guys before!
Let's talk about the superstars:
- Sucrose: This is your everyday table sugar! It’s what you put in your tea, bake with, and sprinkle on your cereal. Sucrose is made by joining a glucose molecule and a fructose molecule. So, that sweet crystalline stuff? It’s a marriage of glucose and fructose, with a little water waving goodbye.
- Lactose: This is the sugar found in milk. If you've ever enjoyed a glass of milk, yogurt, or cheese, you've encountered lactose. It’s formed from a glucose molecule and another simple sugar called galactose.
- Maltose: This one is less common in its pure form for us to eat directly, but it's super important in things like brewing beer and making malted milkshakes. Maltose is made of two glucose molecules linked together.
Isn't that neat? Your basic sweetness is often a result of these little sugar buddies holding hands and giving up a tiny droplet of water. It's like they're saying, "We're stronger, sweeter, and more useful together!"
Why Should You Care About This Sugar Hug?
Okay, okay, I hear you. "This is cool, but why should I, a busy person trying to navigate life, care about two sugars losing water?" Great question! Because this simple act of dehydration synthesis is fundamental to how we get energy and how our bodies function.
Think about your morning. You wake up, maybe you're a bit groggy. You might reach for a banana (fructose!) or a piece of toast (eventually breaks down into glucose!). This is your body getting the raw materials it needs. But your body doesn't just use big sugar molecules like sucrose or lactose directly. It needs to break them down again! And guess what the opposite of dehydration synthesis is? Hydrolysis! It's like unzipping that gummy bear partnership.
When you eat something containing a disaccharide, your digestive system acts like a super-efficient unzipper. It uses water to break the bond between the two monosaccharides. So, sucrose gets split back into glucose and fructose, lactose into glucose and galactose, and so on. These individual monosaccharides are then absorbed into your bloodstream, ready to be used by your cells for energy.

Imagine your body as a car. Glucose is the high-octane fuel. But sometimes, that fuel comes in slightly larger, more stable packages (like sucrose). Your body needs to "unpackage" it into the usable form before it can burn it for power. Dehydration synthesis is how these packages are made in the first place, and hydrolysis is how they're opened up when needed.
Fueling Your Adventures
Every time you run, jump, think, or even just blink, you're using energy. And a huge portion of that energy comes from the breakdown of carbohydrates, which start as simple sugars like glucose. So, those two monosaccharides joining hands and losing a water molecule are indirectly powering your entire life! That afternoon slump? Maybe you need a little more of that dehydration-synthesized goodness broken down for energy. That burst of energy you feel after a sweet treat? Yep, that's the monosaccharides doing their job.

It's also important for plant life! Plants create their own food, and these simple sugars are their building blocks. They use dehydration synthesis to store energy in forms that are easier to manage and transport. So, the next time you admire a big, healthy tree or enjoy a crunchy apple, remember the tiny sugar chemistry that helped it grow.
A Little Bit of Science, A Whole Lot of Life
So, there you have it! Dehydration synthesis between two monosaccharides is a fundamental chemical reaction that’s happening all around us, from the food we eat to the energy we use. It's the elegant way nature builds more complex sugars from simpler ones, setting the stage for how our bodies fuel themselves.
Next time you enjoy something sweet, take a moment to appreciate the subtle dance of molecules. It’s not just about taste; it’s about an amazing process that keeps you going, keeps plants growing, and makes life a little bit sweeter and a lot more energetic. Pretty cool, right?
