When Two Amino Acids Combine Via A Dehydration Reaction
Ever wondered what makes your muscles strong, your hair shiny, or even how your body carries out its most basic functions? It all boils down to tiny, incredible building blocks called amino acids! These are the fundamental ingredients for life as we know it, and their ability to link together is a truly fascinating process. Think of them like LEGO bricks for your body – individually, they’re interesting, but when you start clicking them together, you can build amazing things! Today, we’re going to dive into one of the most important ways these amazing molecules interact: when two amino acids combine via a special kind of reaction. It’s not just a chemical process; it’s the very foundation of proteins, the workhorses of our cells.
The "Click" That Builds Life
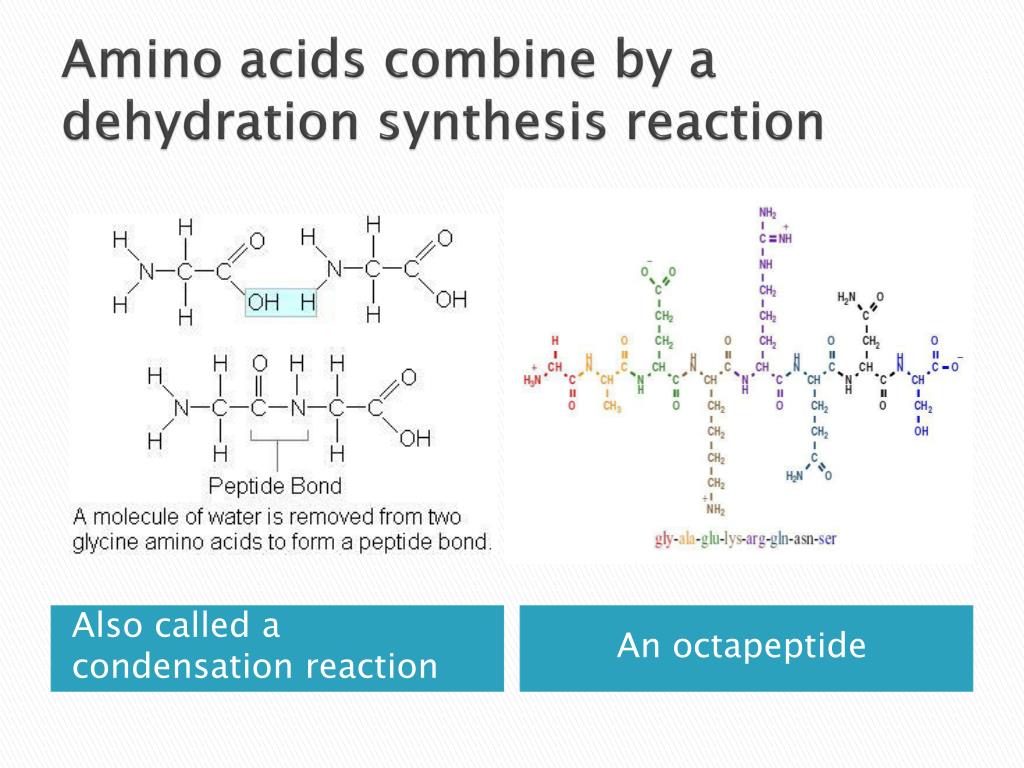
So, what happens when two of these versatile amino acids decide to get acquainted and form a bond? It’s a bit like a friendly handshake, but with a chemical twist! This process is called a dehydration reaction. The name itself gives us a clue: "de-" means to remove, and "hydration" relates to water. So, in essence, a water molecule is removed when these two amino acids link up.
Imagine each amino acid has a specific "hand" it likes to shake. One hand is called the amino group (NH₂), and the other is the carboxyl group (COOH). When they come together, the amino group of one amino acid partners up with the carboxyl group of another. During this union, a hydrogen atom (H) from the amino group and a hydroxyl group (OH) from the carboxyl group are released. These two pieces, H and OH, then join forces to form a molecule of water (H₂O) – and voilà! A new bond is formed between the two amino acids. This new connection is a special type of bond called a peptide bond.
Think of the peptide bond as the strong, reliable zipper that holds these building blocks together, creating longer chains.
This simple act of linking two amino acids is incredibly significant because it’s the very beginning of creating a polypeptide. A polypeptide is essentially a chain of many amino acids linked together by peptide bonds. And guess what? These polypeptides are the precursors to proteins. So, that single peptide bond formed during a dehydration reaction is the first step in building everything from the enzymes that help you digest your food to the antibodies that fight off infections.

Why This "Click" is So Beneficial
The beauty of the dehydration reaction lies in its efficiency and its ability to build complexity from simplicity. Here are some of the amazing purposes and benefits of this fundamental process:
- Building Proteins: This is the most obvious and crucial benefit. Proteins are the workhorses of our cells, performing a vast array of functions. Without the formation of peptide bonds, we wouldn't have the structural components of our bodies (like collagen in our skin and bones), the enzymes that catalyze vital chemical reactions, the hormones that regulate bodily processes, or the transporters that move molecules around.
- Creating Diversity: There are 20 different types of amino acids. The order in which they are linked together in a polypeptide chain determines the final protein's structure and function. This means that a simple dehydration reaction, repeated over and over with different combinations of amino acids, can create an almost infinite variety of proteins, each with its unique job. It’s like having a massive vocabulary where the arrangement of letters creates countless words.
- Energy Efficiency: The removal of a water molecule during the reaction is a clever way for the cell to conserve energy. Instead of requiring a lot of energy input to form the bond, the cell essentially gets a small energy boost or neutralizes an otherwise potentially disruptive molecule (water) in a constructive way.
- Structural Integrity: The strong peptide bond is stable, ensuring that the protein chain remains intact. This stability is essential for proteins to fold into their specific three-dimensional shapes, which is critical for their function. Think of it like building a sturdy structure that can withstand the rigors of cellular life.
- Signal Transduction: Many signaling molecules in the body are proteins or peptides. The ability to create these specific chains of amino acids allows for precise communication within the body, coordinating complex processes.
In essence, the humble dehydration reaction, by forming a single peptide bond, unlocks the door to the incredible world of proteins. It’s a testament to the elegant simplicity and profound power of molecular interactions that underpin all life. So, the next time you marvel at the complexity of your own body, remember these tiny amino acid handshakes – they’re doing some seriously important work!
