When The Following Equation Is Balanced The Coefficients Are C8h18+o2

Imagine you’re in your kitchen, whipping up a delightful batch of cookies. You've got your flour, your sugar, maybe some chocolate chips. It all smells amazing, right? Well, just like a recipe needs the right amounts of ingredients to turn out perfectly, a little dance happening at the molecular level also needs its ingredients measured just so. This dance is called a chemical reaction, and it’s like a tiny, invisible party where molecules mingle and transform.
Today, we’re peeking in on one of these parties, a rather energetic one involving gasoline and air. You know, the stuff that makes your car zoom and your barbecue grill sizzle? It’s all thanks to this particular molecular get-together. The stars of our show are a molecule named octane, which is the main event in gasoline, and its enthusiastic dance partner, oxygen, the stuff we breathe.
Now, when these two get together, it's like a spark igniting a grand performance. They don't just casually say hello; they go all out, rearranging themselves to create something new. The amazing part is, for this dance to be perfectly balanced, for everything to be just right, we need specific numbers of each dancer on the floor. It’s not about throwing in a handful of ingredients and hoping for the best; it’s about precision, a secret code that unlocks the most efficient and satisfying outcome.
Think of it like this: if you’re baking a cake, and the recipe says you need two cups of flour but you only add one, your cake might be a little flat, a little sad. You won't get that perfect, fluffy texture you were dreaming of. Similarly, in this molecular dance, if our dancers, octane and oxygen, aren't in the right numbers, the whole performance can be a bit… off. It won’t be as energetic, and you might not get the best results.
The equation we’re looking at is the blueprint for this molecular fiesta. It tells us how many of our octane molecules need to show up, and how many of their spirited oxygen partners should join the fun. It's a bit like a choreographer for these tiny dancers, ensuring that everyone has a partner and that the energy is just right.
When this equation is balanced, it's a beautiful thing. It means that every atom, those tiny building blocks of everything, is accounted for. Nothing is lost, nothing is made out of thin air – it’s all just rearranged. It’s like tidying up your toy box; every piece is still there, just in a different formation.
So, what are these magic numbers? They’re not random. They’re carefully calculated, like the precise measurements in a gourmet meal. For our octane party, we need a certain amount of this fuel to get going. And to make sure it burns cleanly and powerfully, we need a specific, rather large, amount of oxygen to fuel the fire.

Let’s talk about octane for a second. It’s a hydrocarbon, a fancy name for a molecule made of carbon and hydrogen atoms all holding hands. Think of it as a little chain, and the longer the chain, the more energy it can potentially hold. Our octane has a nice, substantial chain, making it a great energy source.
And oxygen? It’s like the enthusiastic cheerleader for this reaction. It’s always ready to jump in and help things burn. Without enough oxygen, the party can fizzle out, or it can get a bit smoky and inefficient, leaving behind unwanted byproducts. We want a clean burn, a roaring fire, not a smoldering mess.
The Balancing Act: A Symphony of Numbers
Now, the balancing act itself is where the real fun begins. Imagine you have a scale, and on one side you have your octane and oxygen molecules before the reaction. On the other side, you have what they turn into after the dance – carbon dioxide and water. The balanced equation ensures that the weight on both sides is exactly the same. It’s a cosmic game of perfectly matched pairs.
So, for every group of octane molecules, we need a specific number of oxygen molecules to join them. It’s like a dance floor where you need a certain number of partners for every lead. If you have too many leads and not enough partners, some people are left standing around awkwardly. If you have too many partners, well, that’s just not efficient!

The coefficients in this equation are like the instructions for how many of each dancer to invite to the party. They tell us the exact ratio needed for the most successful and energetic transformation. It's a subtle art, but incredibly important for everything from how efficiently your car runs to how cleanly your stove cooks your dinner.
When you see the balanced equation, it’s like looking at a perfectly orchestrated symphony. The numbers are precise, and they ensure that all the atoms are accounted for, transformed, and ready to go. It's a testament to the underlying order in the universe, even in the most mundane of processes.
Think about the simple act of filling up your car. You’re not just pouring liquid into a tank; you’re loading up energy. And the efficiency with which that energy is released, the power that propels you down the road, is all thanks to this perfectly balanced dance between octane and oxygen.
It’s a little bit like a magic trick, isn’t it? You take two things, give them the right encouragement, and poof! You get something new, something useful, something powerful. And the secret to making that magic work every single time lies in those crucial, balanced numbers.

This isn’t just about chemistry in a textbook; it’s about the world around you. It’s about the heat from your stove, the light from a candle, the hum of your car engine. All of these things are powered by these tiny, invisible molecular dances, and their success hinges on precise, balanced choreography.
The next time you see a flame, or feel the warmth of a fire, remember the unsung heroes: the precisely measured molecules of octane and oxygen, dancing together in perfect harmony. It’s a reminder that even in the smallest interactions, there’s a beautiful, ordered universe at play, making our world work, one balanced equation at a time.
It’s amazing to think that such fundamental processes, the ones that power our lives, are governed by such elegant mathematical relationships. The coefficients are the guardians of this order, ensuring that energy is released efficiently and that the results are as intended. They are the unsung heroes of everyday marvels.
So, while it might seem like just a jumble of letters and numbers, the balanced equation for C8H18 + O2 is actually a tiny window into the incredible, often invisible, workings of the universe. It’s a story of energy, transformation, and the quiet beauty of perfect balance, happening all around us, all the time.
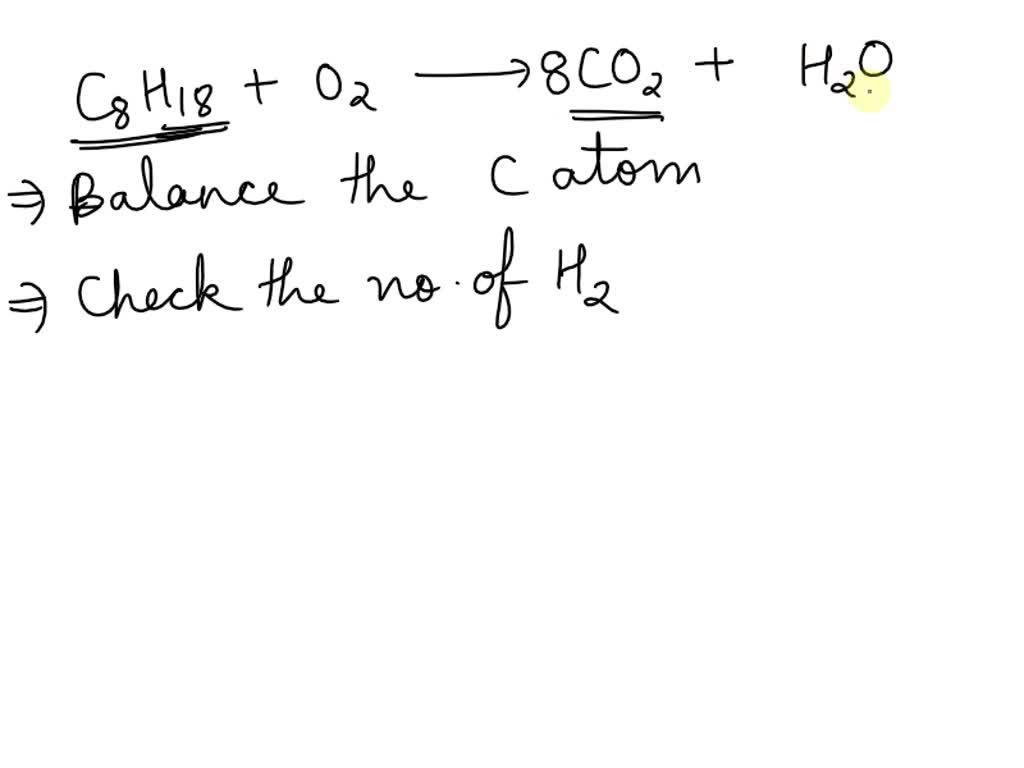
It’s a bit like a perfectly timed performance, where every dancer knows their cue and every step is just right. The magic happens when the numbers are aligned, allowing for the most energetic and clean transformation. It’s a testament to the underlying order that governs even the most explosive of reactions!
From powering your car to lighting your home, the precise ratio of reactants is crucial. This isn't just about creating a reaction; it's about creating the best reaction, the most efficient, the cleanest, the most powerful. The balanced equation ensures that all the atoms have their perfect dance partners, leading to the desired products.
Think of the satisfaction of a perfectly baked cake, or a perfectly tuned engine. There’s a joy in seeing things work exactly as they should, and that joy extends to the molecular world. The balanced equation is the recipe for that perfection, ensuring that every atom plays its part beautifully.
So, the next time you hear about a chemical equation, don’t just see numbers. See the potential for energy, the promise of transformation, and the elegant dance of molecules. It's a reminder that even in the most scientific of subjects, there's a touch of wonder and a whole lot of everyday magic waiting to be discovered.
