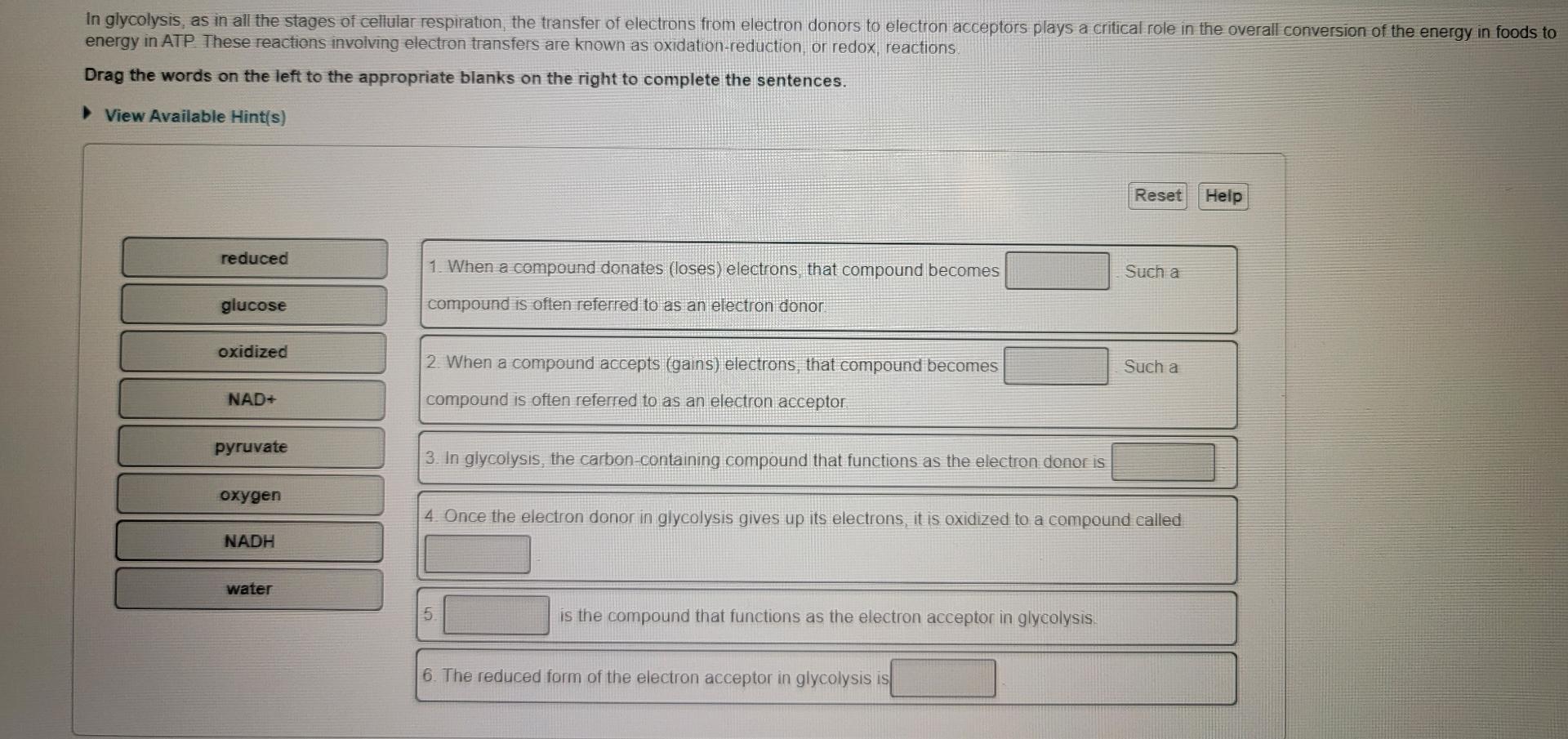
When A Compound Donates Loses Electrons That Compound Becomes
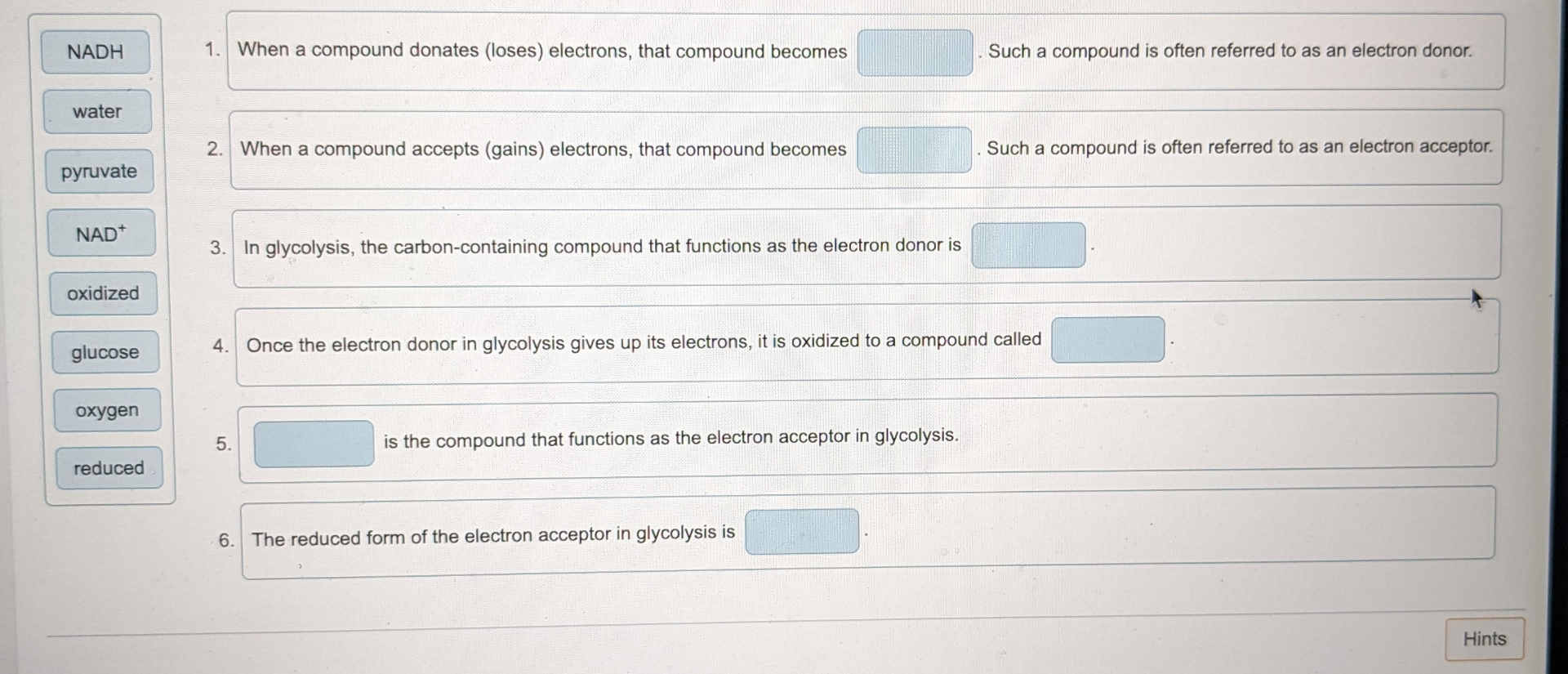
Hey there, science curious pals! Ever feel like you've lost a few electrons yourself? Maybe after a long day of adulting or trying to assemble IKEA furniture? Well, guess what? In the quirky world of chemistry, when a compound donates some of its electrons, something super cool happens. It's not quite the same as forgetting where you put your keys, but it's a fundamental shift, and it’s all about becoming… positively charged!
Seriously, think about it like this: electrons are these tiny, negative little party animals zipping around atoms. They're the ones who get all the attention, forming bonds and making things happen. When a compound decides to be generous and hand over some of these electrons to another compound, it's like it's giving away a piece of its negative mojo.
And what happens when you lose something negative? Bingo! You become positive. It’s basic math, really. Imagine you have a bunch of $-1$ bills. If you give away one of those $-1$ bills, your total wealth just went up by $1$. Ta-da! You're feeling a bit richer, a bit more… positive!
The Electron Exchange Program: It's More Exciting Than It Sounds!
This whole electron-donating gig is a HUGE deal in chemistry. It’s the bedrock of so many reactions and processes that keep our world humming. We're talking about everything from how batteries work (yep, those trusty powerhouses are all about electron transfer!) to how our bodies digest food. So, when a compound decides to go full electron-donor, it's essentially becoming an oxidized agent. Ooh, fancy word, right? Don't let it intimidate you. It just means it’s lost electrons.
Think of it like a super popular person at a party who has tons of cool friends (electrons). They’re so popular, they start handing out invitations (electrons) to others who might be feeling a bit left out or just need some extra sparkle. Once they've given away a few of those invitations, they're still cool, but they’re now carrying a slightly different vibe. They've become the host with the most, but in a way that makes them less… negative.
This loss of electrons isn't a sad story, though. It’s often the start of something new and exciting. It’s like the compound is saying, "You know what? I've got these electrons, and I'm happy to share them with someone who needs them more right now. This will allow them to do something awesome." It's a team sport, you see. One compound's loss is another compound's gain – and sometimes, that gain is a whole lot of chemical fun!
What Do We Call These Generous Givers?
So, what’s the official title for our electron-donating hero? In the esteemed halls of chemistry, these compounds are known as reducing agents. Wait, what? Reducing? That sounds like it's making something less, not more positive. I know, I know, the names can be a little cheeky. It's one of those chemistry quirks that makes you scratch your head and go, "Really?"
The idea is that by donating electrons, the compound is helping another compound to be reduced. Reduction, in chemistry terms, is the gain of electrons. So, our generous donor is helping someone else gain electrons, thereby reducing that other compound. It’s like a chain reaction of helpfulness. The donor is the agent of reduction, hence the name: reducing agent.
It’s a bit like a superhero who doesn’t fight directly but instead empowers others to be heroic. Our reducing agent is the one who gives the power (electrons) to another substance, allowing that substance to undergo reduction. So, the reducing agent itself gets oxidized (loses electrons), while the substance it helps gets reduced (gains electrons). It’s a beautifully balanced, albeit slightly confusing, partnership.
Let's break it down with an analogy. Imagine you have a really powerful flashlight (your compound). You see a friend fumbling around in the dark (another compound needing electrons). You shine your flashlight on them, illuminating their path. Your flashlight (the reducing agent) has given off its light (electrons), and in doing so, it has helped your friend (the substance being reduced) to see. Your flashlight is now a bit dimmer (oxidized), but your friend is no longer lost in the dark (reduced).
Electrons: The Tiny Powerhouses of the Chemical World
So, why are electrons so important in this whole shebang? Well, they're not just tiny, negatively charged particles. They’re the glue that holds atoms together, the sparks that ignite reactions, and the currency that’s exchanged in countless chemical processes. They are, in essence, the movers and shakers of the molecular world.

When a compound has an abundance of electrons, or a certain eagerness to share them, it can become a potent reducing agent. Think of elements like sodium (Na) or potassium (K). These guys are notorious for their desire to shed that single, outermost electron. They're like, "Here, take it! I have another one just like it!" This makes them excellent reducing agents.
Conversely, some elements are electron-hoarders. They love to snatch up electrons. These are the oxidizing agents – the electron-takers. They're the ones who benefit from our generous reducing agents. It's a constant give-and-take, a perpetual dance of electrons that keeps the universe in motion. And honestly, it’s pretty darn fascinating when you think about it.
This electron exchange is what fuels chemical reactions. Without it, you wouldn’t have fire, or rust, or even the complex processes that allow us to breathe and live. It’s a fundamental principle, a grand design that’s playing out all around us, all the time.
The "Donates Loses Electrons" Rule: A Simple Truth
Let's circle back to our core concept: "When a compound donates, it loses electrons." This is a fundamental rule of chemistry, often referred to as the definition of oxidation. When a substance donates or loses electrons, it is said to have been oxidized.

It’s important to remember that oxidation can’t happen in isolation. You can’t just have a compound decide to lose electrons into thin air. Electrons need to go somewhere! This is where the concept of a redox reaction comes in. Redox is a portmanteau of reduction and oxidation. In any redox reaction, one substance is oxidized (loses electrons), and another substance is reduced (gains electrons).
So, our electron-donating compound, the one that loses electrons, is undergoing oxidation. And in doing so, it’s enabling another compound to undergo reduction. It’s a symbiotic relationship, a chemical handshake. One can’t happen without the other.
Think of it like a trade. You have apples (electrons) and your friend has oranges (a need for electrons). You donate your apples, and in the process, you lose them. Your friend gains those apples, and you both end up with something new. Your friend’s collection of fruit has increased, while yours has changed.
This is the beauty of chemistry – it’s all about how things interact and transform. And understanding this simple rule, that donating electrons means losing them and becoming oxidized, is a key to unlocking a whole universe of chemical understanding. It's not just a dry fact; it's a window into the dynamic nature of matter.

From Donor to Delight: The Uplifting Outcome
So, the next time you hear about a compound donating electrons, don’t think of it as a loss. Think of it as a transformation. Think of it as a selfless act that enables something new and wonderful to occur. Our electron-donating compound, by becoming oxidized, is playing a vital role in the grand chemical ballet.
It’s a reminder that sometimes, giving something away is the most powerful way to make a difference. It’s how batteries get their juice, how fuels burn, and how life itself is sustained. The seemingly simple act of losing a few electrons can have monumental consequences.
And you know what? That's a pretty inspiring thought, isn't it? In our own lives, when we share our talents, our time, or even just a kind word, we might feel like we're "losing" a bit of ourselves. But in reality, we're often empowering others, creating positive change, and contributing to something bigger than ourselves.
So, let's embrace this idea! When a compound donates, it loses electrons, yes, but it also becomes a catalyst for something greater. It becomes a player in the ongoing, vibrant story of chemistry. And in a way, we can all be like that generous compound, making the world a little brighter, one electron, one act of kindness, at a time. Go forth and be positively charged, my friends!
