When A Catalyst Is Added To A Reaction Mixture It

Ever feel like you're stuck in a rut? Like your morning commute is a slow-motion zombie apocalypse, or your brain just refuses to get going until that second cup of coffee? Yeah, we’ve all been there. It’s that feeling of “ugh, this is taking FOREVER.” Well, guess what? In the wild and wacky world of chemistry, there’s a magical little helper that swoops in and says, “Hold my beaker, folks, we’re gonna speed this party up!” This little marvel is called a catalyst, and when you add one to a reaction mixture, things… well, they get interesting.
Think of a catalyst like that one friend who’s always buzzing with energy. You know the one. The one who can turn a sleepy Sunday afternoon into an impromptu road trip or convince everyone to go to that ridiculously early spin class. They don’t really do the driving or the pedaling themselves, but man, oh man, do they make it happen way faster and a whole lot more fun. That’s your catalyst in a nutshell. It’s the ultimate wingman for chemical reactions.
Let’s ditch the beakers for a sec and get real. Imagine you’re trying to get your kids to clean their room. You’ve asked, you’ve pleaded, you’ve offered bribes that would make a pirate jealous. Nothing. The toys are multiplying, the socks are staging a hostile takeover of the floor, and the general vibe is one of chaotic stillness. You’re like a lone knight facing a dragon made of Lego and dust bunnies. It’s a slow, arduous battle, and frankly, you’re starting to question all your life choices that led you to this exact moment.
Now, what if you brought in… a challenge? Maybe you say, “Okay, whoever cleans their room the fastest gets to pick the movie tonight and gets an extra cookie!” Suddenly, it’s like someone flicked a switch. The energy levels surge. They’re scrambling, they’re strategizing, they’re even (gasp!) cooperating. The room, miraculously, starts to resemble a place where human beings might actually reside.
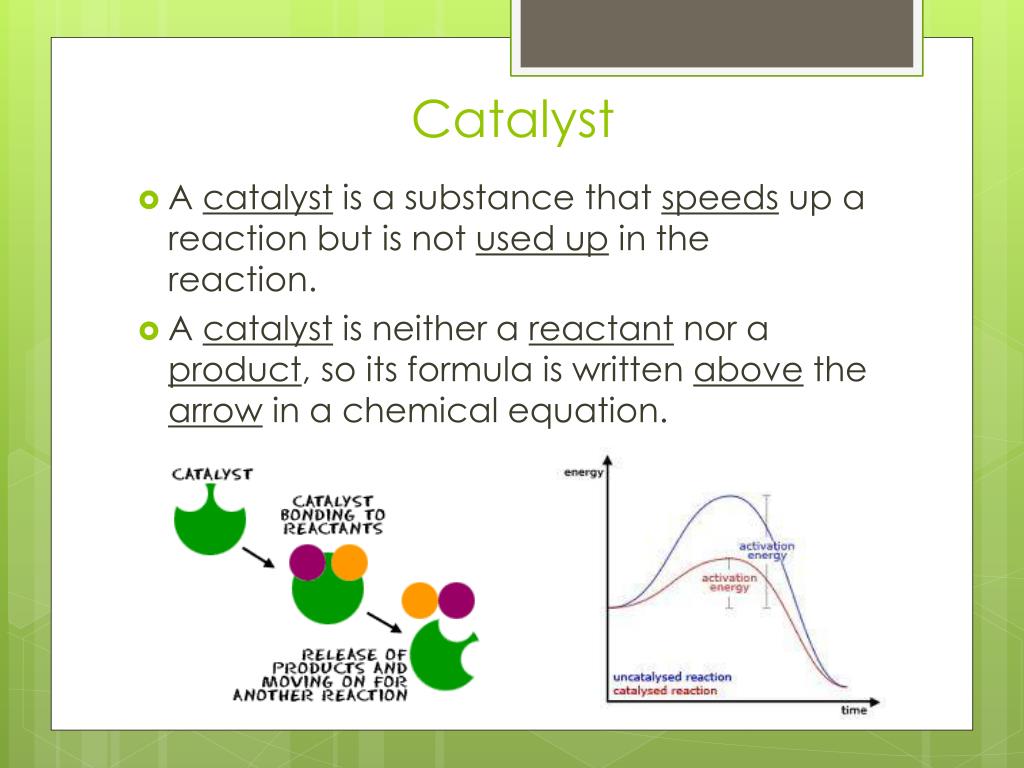
That’s your catalyst, my friends! The challenge. It didn’t clean the room itself, did it? Nope. It just lowered the barrier to entry. It made the whole process seem less like a monumental chore and more like… well, a slightly less monumental chore, but with a shiny reward. In chemistry, a catalyst does something similar. It lowers the activation energy, which is basically the energy needed to kick-start a reaction. Think of it as the tiny hill you have to push your car over before it can roll downhill. A catalyst makes that hill a gentle slope, or even just a slight dip in the pavement.
So, what does this mean for our reaction mixture? It means things are about to get a wiggle on. Instead of a snail’s pace, we’re talking about a cheetah on a caffeine binge. Reactions that might have taken days, weeks, or even eons (yes, eons are a thing in chemistry!) can now happen in the blink of an eye. It’s like going from dial-up internet to fiber optics. The whole experience is just… zippier.

Let’s dive into a slightly more… sticky situation. You’re trying to make caramel. You know, that glorious, golden goo that makes everything better? You’ve got your sugar, you’ve got your heat, and you’re waiting. And waiting. And waiting. The sugar just sits there, being stubbornly sugary. It’s like it’s having an existential crisis, contemplating its own granular existence. You might even start to doubt the very concept of caramel. Is it even real? Or is it just a myth whispered by greedy dentists?
But then! You add a tiny little squeeze of lemon juice, or maybe a drop of water. And BAM! Suddenly, that sugar starts to melt. It starts to bubble and transform. It’s like the lemon juice whispered sweet, acidic nothings into the sugar’s ear, telling it, “Come on, buddy, you can do it! Embrace the melt!” The lemon juice, in this case, is acting like a catalyst. It’s not becoming caramel itself, but it’s making the whole sugar-to-caramel transformation happen way more readily.
This is precisely what happens in countless industrial processes. Think about the stuff that makes your everyday life possible. The gasoline in your car? Made possible by catalysts. The fertilizer that grows your food? Guess what? Catalysts are involved. The plastic in your phone case? Yep, catalysts again. They’re the silent, unsung heroes of modern living, working tirelessly behind the scenes to make sure things happen efficiently and effectively.
It’s kind of like a really good party planner. A great party planner doesn’t do all the dancing or eat all the canapés. But they orchestrate everything. They make sure the music is right, the food arrives on time, and everyone’s having a blast. They lower the stress levels for the host, and they elevate the fun for the guests. They’re the catalyst for a fantastic soirée. And a catalyst in a chemical reaction is pretty much the same deal – it makes the whole event run smoother and faster.
Now, here’s where it gets really cool. Catalysts are recyclable! Unlike that friend who borrows your favorite sweater and it magically disappears into their laundry pile forever, a catalyst pretty much shows up for the reaction, does its magic, and then… pops back out, ready for its next gig. It doesn’t get used up in the process. It’s like a boomerang of chemical helpfulness. This is a huge deal, both for the environment and for our wallets. Imagine if your coffee maker just kept brewing you coffee all day without you having to add more grounds. That would be a dream, right? Catalysts are kind of like that, but for chemical reactions.
Consider the Haber-Bosch process, which is responsible for producing ammonia, a key ingredient in fertilizers. Without this process, feeding the world would be a whole lot harder. And at the heart of it? A catalyst. It takes nitrogen from the air (which is super unreactive and basically says, “Nah, I’m good”) and hydrogen, and with the help of a catalyst, they get persuaded to become ammonia. It’s like convincing two introverts to go on a blind date; it needs a little push, and that push is the catalyst.
Sometimes, adding a catalyst is like adding a dose of enthusiasm to a dull conversation. You’re talking to someone, and it’s all just… “Uh-huh.” “Yeah.” “Mmm-hmm.” It’s like trying to extract sunshine from a cloud. You feel like you’re doing all the heavy lifting, and the other person is just… there. Then, someone else jumps in, someone with a really interesting story or a hilarious observation. Suddenly, the conversation sparks! It becomes lively, engaging, and actually enjoyable. That new person is the catalyst. They didn’t change the topic entirely, but they injected energy and made everyone else more likely to participate and contribute.
In a chemical reaction, the reactants are those people in the dull conversation. They’re there, they have the potential to interact, but they’re just not quite ready to go there. The catalyst comes in, and it’s like it’s saying, “Hey guys, I’ve got this neat little pathway that makes it super easy for you to bump into each other and do your thing!” It provides a sort of chemical social lubricant. It creates an alternative route for the reaction to take, one that requires less energy to get going.
You might even have experienced this in your own kitchen. Making bread, for example. You’ve got your flour, your water, your yeast. The yeast is the star player here, acting as a biological catalyst. It munches on the sugars in the flour and, in doing so, produces carbon dioxide gas. This gas gets trapped in the dough, making it rise. If you just mixed flour and water and waited, nothing would happen. But add that little packet of yeast, and the dough starts to puff up like a happy little balloon. The yeast didn’t turn into bread itself, but it made the bread-making process possible and, more importantly, it made it rise!
The beauty of catalysts is their specificity. It’s not a one-size-fits-all kind of deal. Just like you wouldn’t use a hammer to screw in a lightbulb, chemists have very specific catalysts for very specific reactions. Some catalysts are metals, like platinum or iron. Others are more complex organic molecules. It’s a whole intricate dance of molecular matchmaking. A good catalyst is like a perfectly tailored suit – it fits the reaction just right, making everything smooth and efficient.
So, the next time you hear about a catalyst in a science class, or see one mentioned in an article, don’t just think of some abstract chemical entity. Think of the energetic friend, the helpful nudge, the perfectly timed witty remark. Think of that spark that ignites a process, making it faster, easier, and more likely to happen. Because that, my friends, is the magic of a catalyst. It’s the ultimate enabler, turning sluggish reactions into dynamic transformations, all without breaking a sweat… or, in its case, without getting used up.
It’s the unsung hero of countless processes, from the mundane to the monumental. It’s the reason why we have so many of the materials and products that make our lives comfortable and convenient. Without catalysts, our world would be a much slower, and frankly, a much less interesting place. They’re the secret sauce, the magic ingredient, the invisible hand that keeps the wheels of industry turning, and they do it all with a graceful, non-consuming efficiency that’s frankly, a little bit inspiring. So, here's to the catalysts, the ultimate reaction accelerators!
