When 2-butene Reacts Completely With Bromine The Product Is
Hey there, science curious folks! Ever wonder what happens when some everyday-ish ingredients get together for a little chemical shindig? Today, we're diving into a particularly fun little reaction that's got a bit of a theatrical flair. Imagine this: we've got 2-butene, a simple molecule that’s kind of like a flexible chain with a double bend in the middle. And then we've got our guest star, the ever-exciting bromine. Think of bromine as a two-for-one deal, always ready to split and join in on the fun.
Now, when these two get together, and we’re talking about a complete reaction, meaning everyone plays nice and uses up their partners, something pretty neat happens. It’s not just a casual hello; it's a full-on chemical party! The 2-butene, with its special double bond, is like a welcoming handshake waiting to happen. And bromine, well, it's just itching to break apart and get involved.
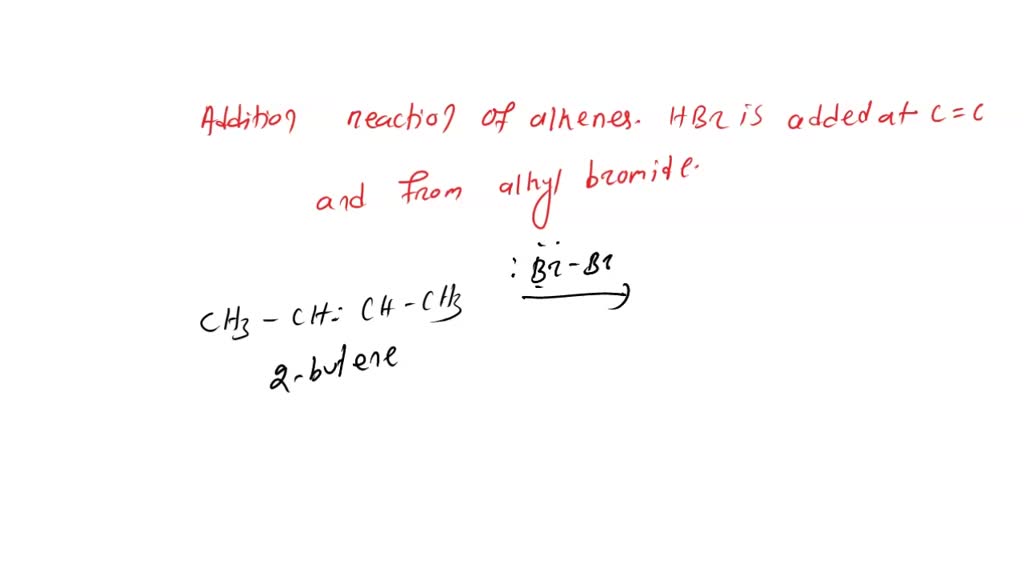
Here’s where the magic really begins. That double bond in 2-butene is the key. It’s a spot where the atoms are held together a little more loosely, making it super eager to react with other things. Bromine, as we mentioned, comes as a pair of bromine atoms stuck together. When it meets 2-butene, this pair can’t resist. They decide to go their separate ways.
Each individual bromine atom is now ready to join the 2-butene party. And guess what? They find perfect spots to attach themselves! It’s like a perfect fit, a jigsaw puzzle snapping into place. The double bond in 2-butene breaks open, and each of those two bromine atoms latches onto one of the carbon atoms that were part of the original double bond. Pretty slick, right?
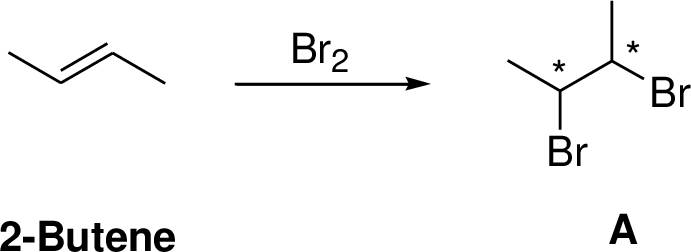
The result? We get a brand new molecule. This isn’t just any old molecule; it’s got a name that sounds a bit like a secret agent’s designation: 2,3-dibromobutane. Say that five times fast! What's so cool about this 2,3-dibromobutane? Well, it's no longer got that flexible double bend. It’s now a more solid, single-bonded chain. The bromine atoms have essentially "saturated" the molecule, filling in those reactive spots.
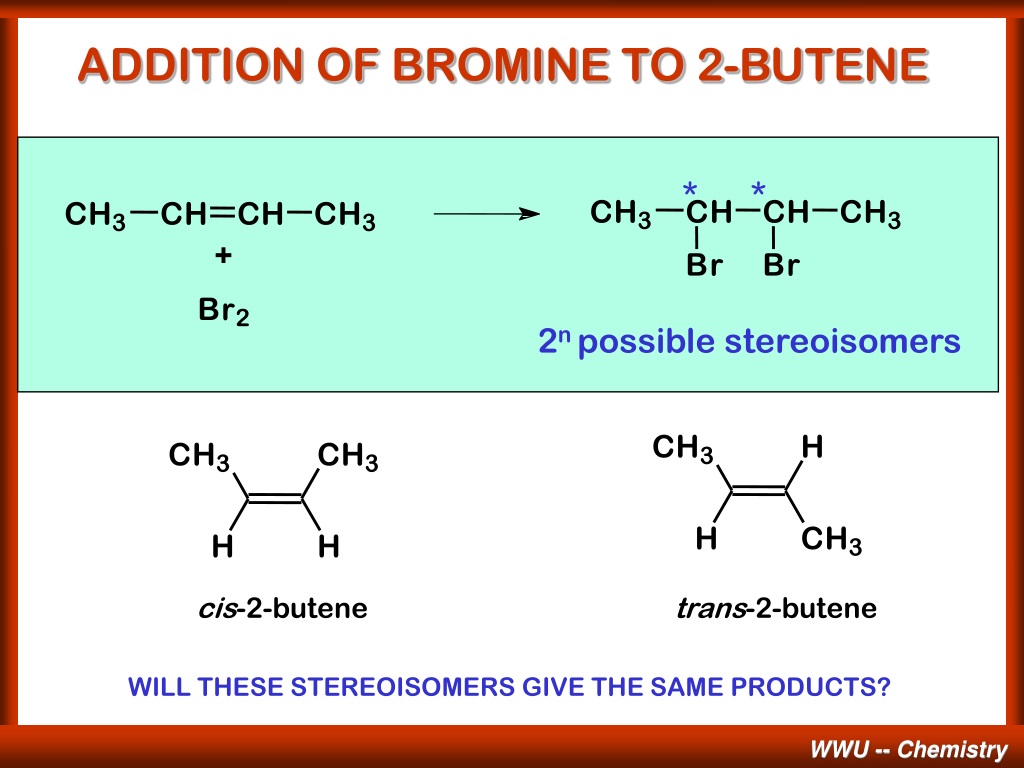
Think of it like this: 2-butene is a road with a temporary bridge. Bromine comes along, and instead of just passing by, it decides to reinforce that bridge. It breaks its own connection and uses both its parts to build stronger, permanent supports. The result is a sturdier, more complete structure. The 2,3-dibromobutane is the finished product of this diligent construction work.
What makes this so entertaining to learn about? It’s the sheer predictability and the satisfaction of seeing a clear-cut transformation. There are no messy leftovers, no "maybe this, maybe that." When 2-butene reacts completely with bromine, the product is always 2,3-dibromobutane. It’s like a perfectly executed magic trick every single time. No smoke and mirrors, just pure, reliable chemistry.
It’s also special because it shows us how atoms and molecules can rearrange themselves. They’re not static; they’re constantly interacting and changing. This reaction is a fantastic illustration of this dynamic dance. The bromine addition is a classic example of an electrophilic addition reaction. Don't let the fancy name scare you! It just means that the bromine molecule, with its tendency to split into positively and negatively charged parts, is attracted to the electron-rich double bond of the 2-butene.
Imagine the 2-butene as a friendly crowd, and the bromine as a pair of very social individuals. When they meet, the crowd's excitement (the double bond's electrons) attracts the social butterflies. The bromine pair breaks up, and each individual bromine finds a welcoming spot to mingle and make friends within the 2-butene structure. The result is a happy, more tightly knit group – the 2,3-dibromobutane.

Why should you be curious? Because this isn't just about memorizing a formula. It's about understanding the fundamental ways matter behaves. It’s about recognizing patterns and predicting outcomes. This simple reaction is a building block for understanding more complex chemical processes. Plus, it’s just plain cool to know that with a bit of 2-butene and some bromine, you can create something entirely new, with a neat, orderly structure.
So, the next time you hear about 2-butene and bromine having a little chemical chat, remember the exciting transformation that occurs. It's a neat, clean, and predictable reaction that results in the formation of 2,3-dibromobutane. It’s a testament to the elegant rules that govern the universe, one molecule at a time. Maybe it’ll even inspire you to look at everyday substances with a little more wonder and curiosity!
