What Types Of Intermolecular Forces Exist Between Hi And H2s

Hey there, fellow curious minds! Ever find yourself staring at two seemingly simple molecules, like hydrogen iodide (HI) and hydrogen sulfide (H₂S), and wondering what invisible forces are holding them together (or pushing them apart)? It’s not just chemistry class jargon; understanding these intermolecular forces is like peeking behind the curtain of how the world around us works, from the way your morning coffee brews to the very air you breathe. And honestly, it’s kinda cool!
We’re talking about the whispers between molecules, the subtle attractions and repulsions that dictate everything from boiling points to how well certain substances dissolve. Think of it like a cosmic dance, where molecules twirl and sway based on their internal makeup. Today, we’re going to dive into the delightful world of intermolecular forces, specifically focusing on the intriguing interactions between HI and H₂S. No need for a lab coat or a complex formula sheet; we're keeping it breezy, stylish, and totally accessible.
The Molecule Mashup: HI vs. H₂S
So, let’s get acquainted with our players. First up, we have hydrogen iodide (HI). It’s a simple diatomic molecule, meaning it’s made of just two atoms: one hydrogen and one iodine. Now, iodine is a pretty hefty atom compared to hydrogen. This size difference and the electronegativity (think of it as an atom’s hunger for electrons) play a crucial role. Iodine is more electronegative than hydrogen, meaning it pulls the shared electrons closer to itself. This creates a slight partial negative charge on the iodine and a slight partial positive charge on the hydrogen. We call this a polar molecule. Think of it like a tiny, invisible magnet with a positive end and a negative end.
Our other contestant is hydrogen sulfide (H₂S). This one’s a bit more of a party animal, with one sulfur atom and two hydrogen atoms. Sulfur, like iodine, is more electronegative than hydrogen. It grabs the electrons from both hydrogens, making the sulfur atom partially negative and the hydrogen atoms partially positive. But here’s the twist: H₂S has a bent shape, kind of like a little V. This geometry means the individual partial charges don't cancel each other out. The molecule as a whole has a distinct positive side and a distinct negative side, making it another polar molecule. So, both HI and H₂S are polar buddies!
The Intermolecular Force Fiesta: What's Cooking?
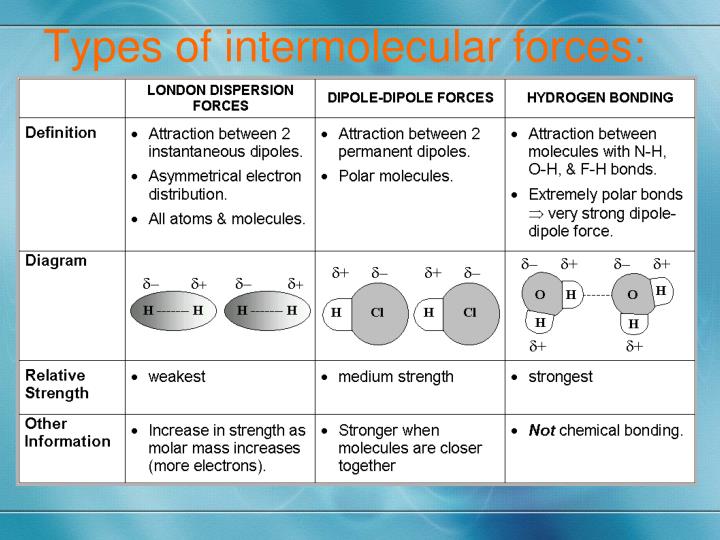
Now that we know our molecules are polar, what kind of shindig are they going to throw when they meet? Since both HI and H₂S are polar, they’re going to attract each other through a primary force called dipole-dipole interactions. Remember those partial positive and partial negative ends we talked about? Well, the positive end of one molecule will be attracted to the negative end of another. It’s like the universe’s way of saying, "Opposites attract!"
Imagine a crowded room at a party. People naturally gravitate towards others who have complementary vibes. That’s a bit like dipole-dipole. The partially positive hydrogen of an HI molecule might get cozy with the partially negative iodine of an H₂S molecule, and vice versa. This attraction is what keeps liquids from instantly becoming gases and solids from crumbling into dust. It’s the glue that holds matter together in a liquid or solid state.
Beyond the Basics: London Dispersion Forces
But wait, there’s more to this molecular soirée! Even non-polar molecules have some kind of attraction. And polar molecules? They’ve got a little extra something-something going on too. Enter London dispersion forces (sometimes called London forces or dispersion forces). These are the universal connectors, present in all molecules, polar or not. How do they work?

Think of electrons as being in constant, random motion around the nucleus of an atom. At any given instant, the electrons might be distributed unevenly, creating a temporary, fleeting dipole. It’s like a tiny, spontaneous blink of a magnetic field. This temporary dipole can then induce a similar, temporary dipole in a neighboring molecule, leading to a weak, short-lived attraction. It’s incredibly subtle, like a shared whisper in a loud room.
For HI and H₂S, these London dispersion forces are definitely at play. Since iodine and sulfur are relatively large atoms with lots of electrons, they can develop more significant temporary dipoles. So, even though dipole-dipole forces are the main event here, the London dispersion forces contribute to the overall attraction. It's like having a charismatic main speaker at an event, but also a really engaging background hum that keeps everyone comfortable.
The Hydrogen Bond Question: A Special Guest?
Now, a common intermolecular force that people often think about is the hydrogen bond. This is a particularly strong type of dipole-dipole interaction. It happens when a hydrogen atom is bonded to a highly electronegative atom like oxygen (O), nitrogen (N), or fluorine (F). Because these atoms are so electronegative, they pull the hydrogen’s electron so strongly that the hydrogen atom becomes almost like a bare proton, with a significant partial positive charge. This highly positive hydrogen is then strongly attracted to a lone pair of electrons on a highly electronegative atom in a neighboring molecule.
So, do we have hydrogen bonds between HI and H₂S? Let’s look closely. In HI, the hydrogen is bonded to iodine. Is iodine highly electronegative in the same league as oxygen, nitrogen, or fluorine? Not quite. While it’s more electronegative than hydrogen, the difference isn’t as dramatic. In H₂S, the hydrogen is bonded to sulfur. Again, sulfur is electronegative, but not to the same extreme degree as O, N, or F.

Therefore, while there might be a weak form of attraction that some chemists might categorize as a very, very weak hydrogen bond, it's generally not considered a true hydrogen bond in the way we see in water (H₂O) or ammonia (NH₃). The primary forces at play are dipole-dipole interactions and London dispersion forces. Think of it as the difference between a firm handshake (dipole-dipole) and a fleeting wave (London dispersion), versus a warm, prolonged hug (hydrogen bonding). HI and H₂S are more in the handshake and wave territory.
Why Does This Even Matter? Practical Pop-Culture Applications!
Okay, so we’ve got these invisible forces. Why should you care beyond acing a pop quiz? Because these forces dictate the physical properties of substances, and those properties affect our lives in countless ways!
Boiling Points and States of Matter: Molecules with stronger intermolecular forces tend to have higher boiling points because more energy is required to overcome those attractions and turn a liquid into a gas. So, HI and H₂S, with their dipole-dipole and dispersion forces, will have higher boiling points than, say, non-polar molecules like methane (CH₄). This is why water (with strong hydrogen bonding) is liquid at room temperature, while methane (with only weak dispersion forces) is a gas.
Solubility: Ever heard the saying "like dissolves like"? It’s a direct consequence of intermolecular forces! Polar solvents (like water) tend to dissolve polar solutes (like salt), and non-polar solvents (like oil) tend to dissolve non-polar solutes (like grease). So, if you were trying to dissolve HI in H₂S, they’d likely mix pretty well because they’re both polar and can engage in similar dipole-dipole interactions. This principle is why your salad dressing separates (oil and vinegar don't mix because oil is non-polar and vinegar is mostly water, which is polar).
Biological Processes: At a much grander scale, intermolecular forces are fundamental to life! The way proteins fold into their complex shapes, the way DNA strands pair up, and how enzymes bind to their substrates are all governed by these subtle attractions. Even the way your brain cells communicate involves the interaction of molecules mediated by intermolecular forces.
Industrial Applications: In the world of industry, understanding these forces is crucial. From designing refrigerants to creating effective pharmaceuticals, chemists and engineers rely on this knowledge. For instance, the strength of intermolecular forces influences how gases behave under pressure, which is vital for everything from making fizzy drinks to storing natural gas.
Fun Facts and Cultural Tidbits
Did you know that hydrogen sulfide (H₂S) is the compound responsible for the “rotten egg” smell? It’s produced naturally by decaying organic matter and is also found in volcanic gases. While it’s toxic in high concentrations, it’s present in very low concentrations in our atmosphere. So, the next time you catch a whiff of that distinct aroma, you’re experiencing the presence of H₂S molecules interacting with your olfactory receptors!
Hydrogen iodide (HI) is a component of hydroiodic acid, a strong acid. It’s used in various chemical syntheses and has historically been used as a reducing agent. Imagine the vigorous bubbling and the distinct properties of such a solution – all thanks to the molecular interactions within!

Thinking about those dipole-dipole interactions reminds me of old-school romance novels where characters are drawn to each other by an undeniable, almost magnetic, pull. It’s a bit cheesy, but the analogy holds! Similarly, London dispersion forces are like those quiet, supportive friendships that just work without needing a lot of fanfare. They’re the reliable background players.
A Daily Dose of Molecular Magic
So, there you have it! The seemingly simple interactions between molecules like HI and H₂S are a vibrant tapestry of forces. We’ve got the steady pull of dipole-dipole interactions, the universal shimmer of London dispersion forces, and a nuanced look at why true hydrogen bonds aren't quite the main act here.
The next time you brew your coffee, notice how the water (polar, with hydrogen bonds!) extracts the soluble compounds from the coffee grounds. Or consider why your favorite lotion (often containing oils and water-based ingredients) feels smooth on your skin – it’s a carefully orchestrated balance of intermolecular forces. Even the way soap cleans grease is a testament to these molecular attractions and repulsions.
It’s a beautiful reminder that the macroscopic world we experience is built upon these incredibly small, yet powerful, molecular conversations. From the subtle scent of a rose to the stability of a skyscraper, it all comes down to how molecules choose to interact. And isn't that just wonderfully fascinating?
