What Types Of Intermolecular Forces Are Found In Xef4
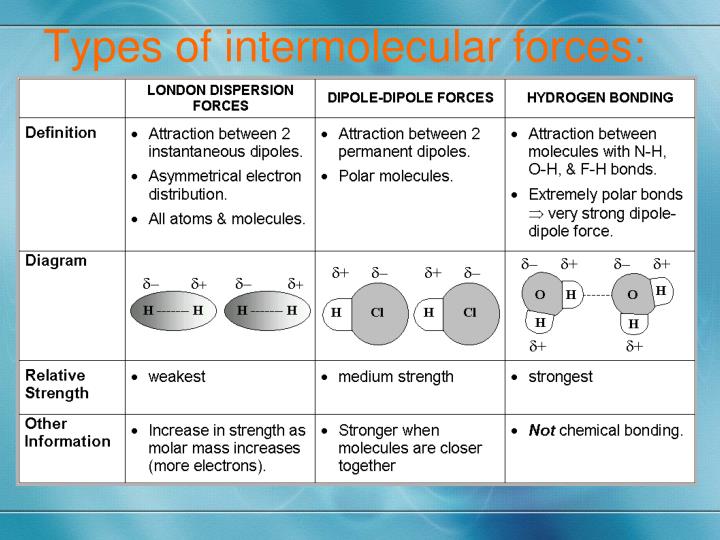
Hey there! So, you wanna chat about Xenon Tetrafluoride, huh? XeF4? Cool choice! It’s like the shy but surprisingly complex kid in chemistry class. We’re gonna dive into what makes it, well, stick together. Think of it like trying to figure out why some people just naturally click, right? Except, you know, with atoms and stuff. And forces. Lots of forces.
So, the big question is: what kinds of intermolecular forces are hanging out with XeF4? This is where things get a little… interesting. Because, honestly, it's not as straightforward as you might think. It's not like there are little XeF4 molecules holding hands with hydrogen bonds or anything as cute as that.
First off, let's get one thing straight. XeF4 itself is a molecule. We're talking about the forces between these molecules, not the forces holding the atoms inside one XeF4 molecule together. That’s called intramolecular force, and for XeF4, that’s the strong, unbreakable covalent bond. But we're gossiping about the inter-molecular stuff here, the ones that make them want to be near each other. Or, you know, not.
Now, the first thing we gotta do is look at the shape of this XeF4 molecule. This is crucial. Like, super-duper important. Because the shape tells us if the molecule is polar or nonpolar. And that, my friend, is the gateway to understanding its intermolecular forces. It’s like knowing if someone’s an introvert or an extrovert – it tells you a lot about how they’ll interact with others!
So, let’s get our VSEPR (Valence Shell Electron Pair Repulsion) hats on. It’s a fancy name, I know, but it basically says that electron pairs around an atom hate being close to each other. They want to spread out as much as possible. For Xenon, it’s got 8 valence electrons. It bonds with four Fluorines, using 4 electrons. That leaves 4 electrons, which form two lone pairs. So, we have 4 bonding pairs and 2 lone pairs. That’s a total of 6 electron domains. And when you have 6 electron domains, the ideal arrangement to minimize repulsion is octahedral. Think of a soccer ball, with points all around it. Very symmetrical.
But here’s the twist! The molecular geometry is different from the electron geometry. Because the lone pairs, even though they’re invisible, they still take up space and push the bonding pairs around. So, instead of an octahedron, we get a square planar shape. Imagine a flat square, with the Xenon in the middle and the four Fluorines at the corners. And those two lone pairs? They’re chilling out above and below the plane, pointing away from everything. Like they’re saying, “We’re here, but we’re not really here.”
And guess what? This square planar shape is super symmetrical. You've got the Fluorines all arranged perfectly around the Xenon. Even though Fluorine is way more electronegative than Xenon (meaning it pulls electrons towards itself like a magnet), the symmetry of the square planar shape means that the little dipoles created by those Xe-F bonds actually cancel each other out. It’s like having a tug-of-war where both teams are perfectly balanced, and the rope doesn’t move. Poof! The molecule ends up being nonpolar. Voila!
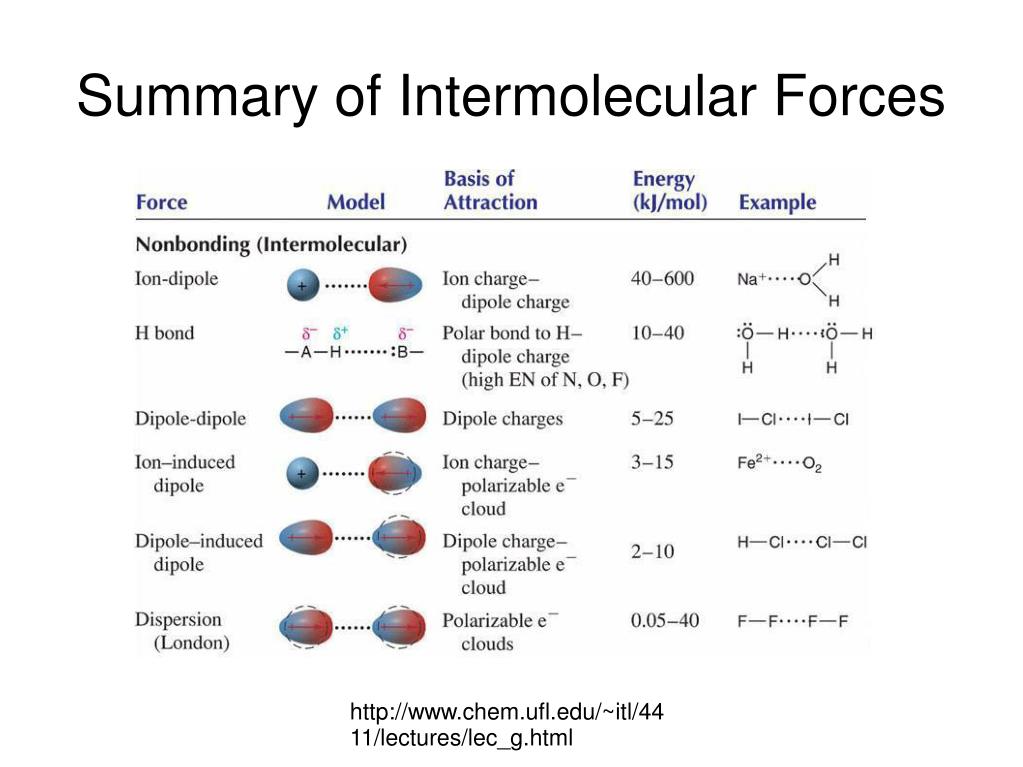
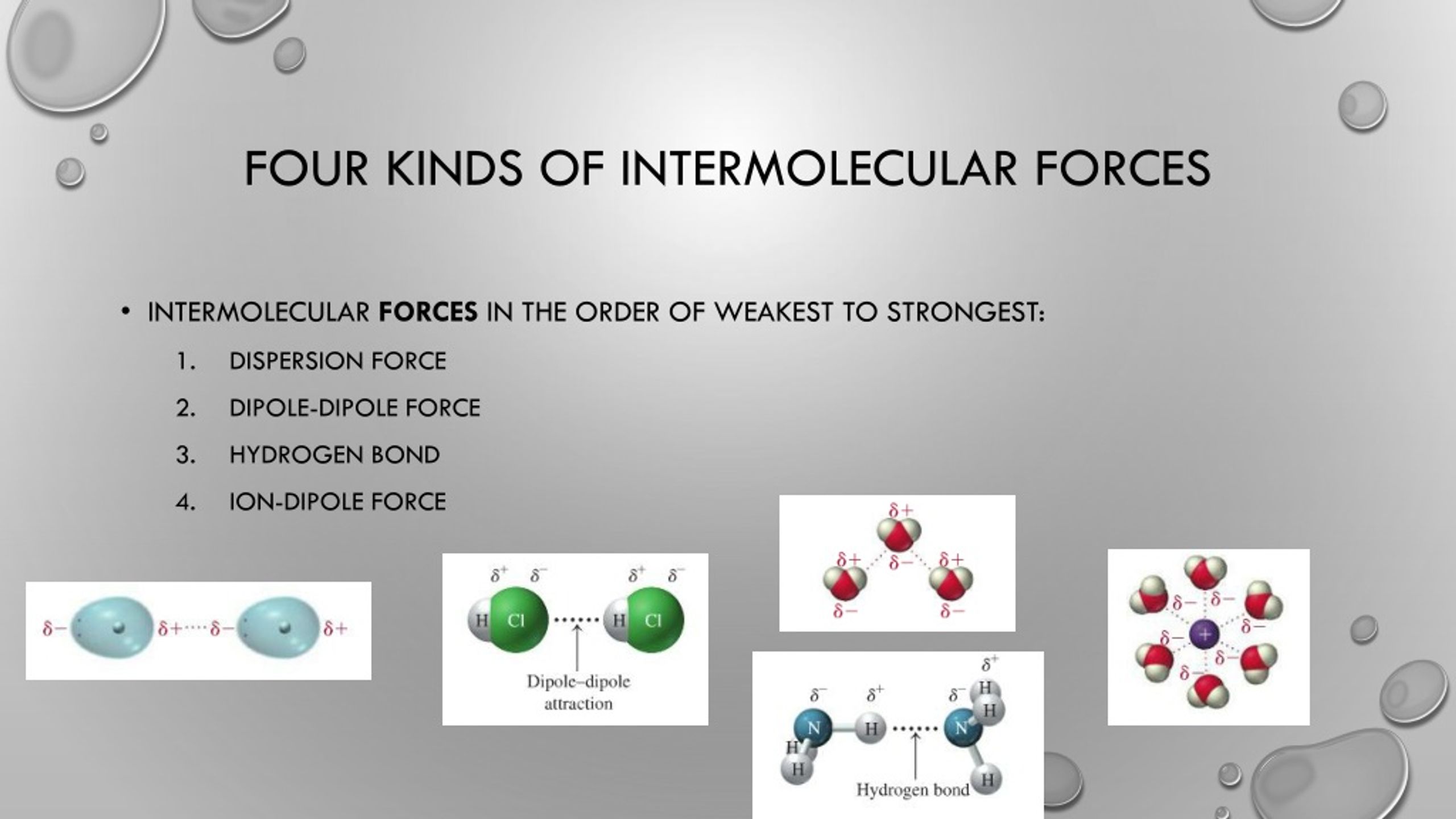
So, if XeF4 is nonpolar, what kind of intermolecular forces are we talking about? This is where the plot thickens, or rather, thins out. For nonpolar molecules, the main players in the intermolecular force game are the London dispersion forces (also known as Van der Waals forces, which is a bit of an umbrella term, but let’s stick with LDF for now). These are the weakest of the intermolecular forces, but they’re everywhere. Like the dust bunnies under your bed – you might not notice them, but they’re there.
How do these LDFs work, you ask? It’s all about temporary electron cloud fluctuations. Imagine the electrons in a XeF4 molecule are like a bunch of super-energetic teenagers at a party. They’re zipping around, and at any given instant, they might all be hanging out on one side of the molecule. This creates a temporary, instantaneous dipole. It’s like a little puff of negative charge on one side and a little puff of positive charge on the other. It's super fleeting, like a blink of an eye.
And what does this temporary dipole do? It can induce a dipole in a neighboring XeF4 molecule. So, if one molecule suddenly gets a little negative charge here, it can nudge the electrons in the next molecule to shift away from that spot, making that molecule temporarily positive on the side closest to the first molecule. And that slight attraction between these temporary, induced dipoles is your London dispersion force. It’s like a chain reaction of mild nudges and polite acknowledgements. “Oh, you’ve got a bit of a charge there? So do I, for a millisecond!”
The strength of these LDFs depends on a couple of things. First, the size of the molecule. Bigger molecules have more electrons, and more electrons mean more chances for these temporary fluctuations. XeF4 is a pretty decent-sized molecule, so it’s got a reasonable number of electrons to play with. More electrons, more potential for LDFs. It’s like having more people at the party – more possibilities for awkward bumping into each other.
Second, the shape can play a role. More elongated, "stringy" molecules can have stronger LDFs because they have more surface area to interact with their neighbors. Square planar XeF4 is pretty compact, so it might not have as much surface area for these interactions as, say, a long, skinny molecule. But it's still got those electrons, you know?
Now, you might be thinking, “But wait a minute! Fluorine is really electronegative! Surely there’s some dipole-dipole interaction happening, right?” And that's a great question! For a truly polar molecule, where the dipoles don't cancel out, you'd definitely have dipole-dipole forces. These are the attractions between the permanent positive end of one polar molecule and the permanent negative end of another. Think of two little magnets that are always attracted to each other.
But, as we established (with all our fancy VSEPR knowledge!), XeF4 is nonpolar. The individual Xe-F bond dipoles are there, absolutely. The Fluorines are tugging hard on those electrons. But because of the perfect square planar symmetry, those pulls are balanced. They cancel each other out. So, there are no permanent dipoles to create those strong, consistent dipole-dipole attractions. It’s like having a perfectly balanced scale; no matter how much you push on one side, the other side pushes back with equal force, and the whole thing stays level. No net tilt, no permanent attraction.
What about hydrogen bonding? Oh, hydrogen bonding! That’s like the VIP club of intermolecular forces. It’s a special, super-strong type of dipole-dipole interaction that happens when hydrogen is bonded to a highly electronegative atom like oxygen, nitrogen, or fluorine. Think water molecules holding hands! It's super strong and gives things like water its amazing properties. But, alas, XeF4 doesn't have any hydrogen atoms. So, no hydrogen bonding for our friend here. It’s not invited to that particular party, sadly.
So, to recap our little chemical chat, XeF4 is a nonpolar molecule. And the primary intermolecular force that holds these nonpolar molecules to each other is London dispersion forces. These are the temporary, fleeting attractions that arise from electron cloud fluctuations. They’re not the strongest, but they’re definitely present.
It’s a bit like the difference between a passionate, all-consuming love (hydrogen bonding) and a comfortable, friendly acquaintance (LDFs). XeF4 is firmly in the acquaintance zone. It’s polite, it’s there, but it’s not exactly swooning over its neighbors.
Why is this important, you ask? Well, understanding these forces tells us a lot about the physical properties of XeF4. For instance, because it only has weak London dispersion forces, it doesn’t have a very high boiling point or melting point. It’s not going to be a solid rock at room temperature, holding on tight. It’s more likely to be a gas or a liquid because it doesn’t take much energy to pull those molecules apart.
Think about it. If you have strong intermolecular forces, like in water with its hydrogen bonds, it takes a lot of heat to break those connections and turn it into steam. But with XeF4, because the LDFs are relatively weak, it's much easier to overcome them. Less energy needed, lower boiling point. Makes sense, right?
Also, nonpolar molecules tend to be insoluble in polar solvents like water. They don't really "like" to mix. They're like oil and water – they just don't play well together. This is because the polar water molecules are busy attracting each other with their strong hydrogen bonds, and they don't have much interest in interacting with the nonpolar XeF4 molecules. The XeF4 molecules, in turn, are more attracted to each other (via LDFs) than they are to the water molecules.
So, in summary, for XeF4, you're mainly looking at London dispersion forces. It’s the humble, but essential, force that keeps these nonpolar molecules in the same general vicinity. It’s the molecular equivalent of a gentle nod hello, rather than a warm hug. And that’s all thanks to its neat, tidy, and perfectly balanced square planar shape!
Isn’t chemistry fun? We take this seemingly simple compound, XeF4, and unlock all these little secrets about how its molecules behave. It’s like peeling back layers of an onion, but way less tear-inducing! And all because of electron pairs and shapes and electronegativity. Who knew something so small could have such an impact on how things interact?
So, next time you hear about Xenon Tetrafluoride, you can impress your friends (or at least confuse them a little) by talking about its LDFs and its nonpolar nature. You’re basically a molecular matchmaker now! You know what kind of forces are (or aren't!) drawing these little guys together. Pretty neat, huh? Keep those chemistry questions coming – I’m always up for a good molecular chat!
