What Type Of Interparticle Forces Holds Liquid N2 Together
Ever wondered what keeps things from just floating apart? We’re not just talking about glue or magnets; the universe has a much subtler, yet incredibly powerful, way of holding matter together. Today, let's dive into the fascinating world of intermolecular forces, specifically focusing on what makes liquid nitrogen, well, liquid! It might sound a bit technical, but understanding these tiny, invisible attractions is like unlocking a secret code to how so much of our world behaves. It’s a journey into the heart of matter, and it’s surprisingly fun and relevant!
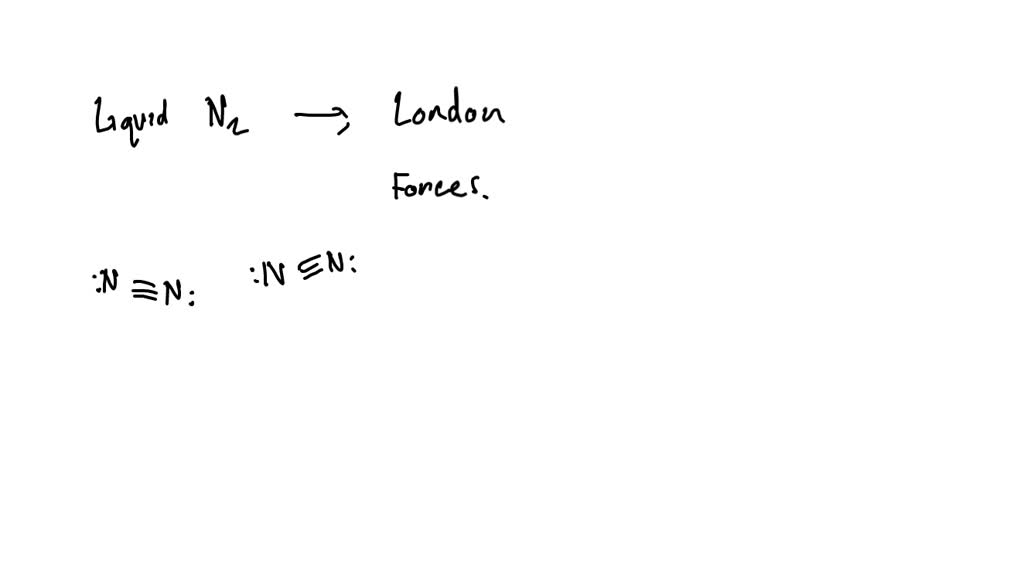
The big question is: what type of interparticle forces holds liquid nitrogen together? Nitrogen, as you probably know, is a gas at room temperature. It makes up about 78% of the air we breathe! But when you cool it down to extremely low temperatures (around -196 degrees Celsius or -321 degrees Fahrenheit), it transforms into a mesmerizing, misty liquid. This transformation isn't magic; it's a direct result of these subtle attractions between individual nitrogen molecules (N2). Unlike water, which has strong hydrogen bonds holding it together, nitrogen molecules are relatively simple and nonpolar. This means they don't have a significant positive or negative end. So, what’s doing the holding?
The answer lies in a type of force called London dispersion forces (sometimes called van der Waals forces). Now, don't let the fancy name intimidate you! Even though nitrogen molecules are neutral, at any given moment, the electrons within them are constantly in motion. This means that for a fleeting instant, one side of a nitrogen molecule might have a slightly more concentrated negative charge, while the other side has a slightly more positive charge. This creates a temporary, induced dipole. This temporary dipole in one molecule can then influence the electron distribution in a neighboring molecule, creating a weak, temporary attraction. It's like a chain reaction of tiny, fleeting nudges and pulls!
These London dispersion forces, while individually very weak, become significant when you have a huge number of molecules packed closely together, as they are in a liquid. The more electrons a molecule has, the stronger these dispersion forces tend to be. Nitrogen molecules, with their 14 electrons each, have enough of these weak attractions to overcome the tendency to spread out as a gas and stay huddled together as a liquid at very cold temperatures. Think of it like a group of shy people at a party; individually, they might not interact much, but if everyone is packed in, they'll inevitably bump into each other and form tentative connections.

Understanding these forces has practical implications everywhere. In education, it helps students grasp the difference between gases, liquids, and solids, and why different substances have vastly different boiling and melting points. In daily life, the principles behind these forces are crucial for industries dealing with gas liquefaction, like the production of liquid oxygen for medical use or liquid hydrogen as a rocket fuel. Even the way perfumes evaporate or how certain materials stick together can be explained by these fundamental interactions.
Want to explore this yourself? While creating liquid nitrogen is definitely not a DIY project, you can observe its effects! Many science museums have demonstrations where you can see liquid nitrogen in action – watch it boil vigorously when exposed to air, or see how it can freeze everyday objects like flowers instantly. You can also research videos online showcasing these fascinating transformations. Next time you see something cool happening with gases and liquids, remember the invisible dance of electrons and the subtle, yet powerful, interparticle forces at play!
