What Type Of Alcohol Undergoes Oxidation To Yield A Ketone

There's a delightful transformation happening in kitchens and studios around the world, a chemical dance that turns a common, beloved beverage into a powerful creative catalyst. We're not talking about a new cocktail recipe, but rather a fascinating scientific process that involves a specific type of alcohol. Prepare to be amazed, because when it comes to unlocking new artistic possibilities, secondary alcohols are the unsung heroes!
You might be familiar with secondary alcohols in their delicious liquid forms. Think of the rich, complex flavors of many wines and beers. But the magic happens when these alcohols undergo a process called oxidation. With the right reagents and conditions, the hydroxyl group (-OH) on the middle carbon of a secondary alcohol is converted into a carbonyl group (C=O), resulting in a ketone. This might sound like advanced chemistry, but the results are surprisingly accessible and incredibly beneficial for anyone with a creative itch.
For artists and hobbyists, understanding this transformation can open up a world of new techniques. Imagine using ketones derived from fermented grapes to create vibrant, unique pigments for your paintings. Or perhaps you're a baker who discovers how ketones extracted from carefully aged spirits can add an unexpected depth and complexity to your pastries. Even for the casual learner, experimenting with these reactions offers a hands-on, engaging way to connect with the principles of chemistry. It's about seeing the world around you, from the ingredients in your pantry to the hues on your canvas, in a whole new light.
Consider the variations: a secondary alcohol like 2-propanol (isopropyl alcohol), when oxidized, yields acetone. While industrial acetone is readily available, imagine the satisfaction of understanding its origin from a simpler alcohol. In the realm of beverages, the oxidation of certain alcohols present in fermented products can lead to the formation of ketones that contribute to their characteristic aromas and flavors. Think of the fruity esters and nutty notes you detect in a well-aged cider or a complex Belgian ale – these are often the result of intricate chemical pathways involving various alcohols and their oxidized derivatives.
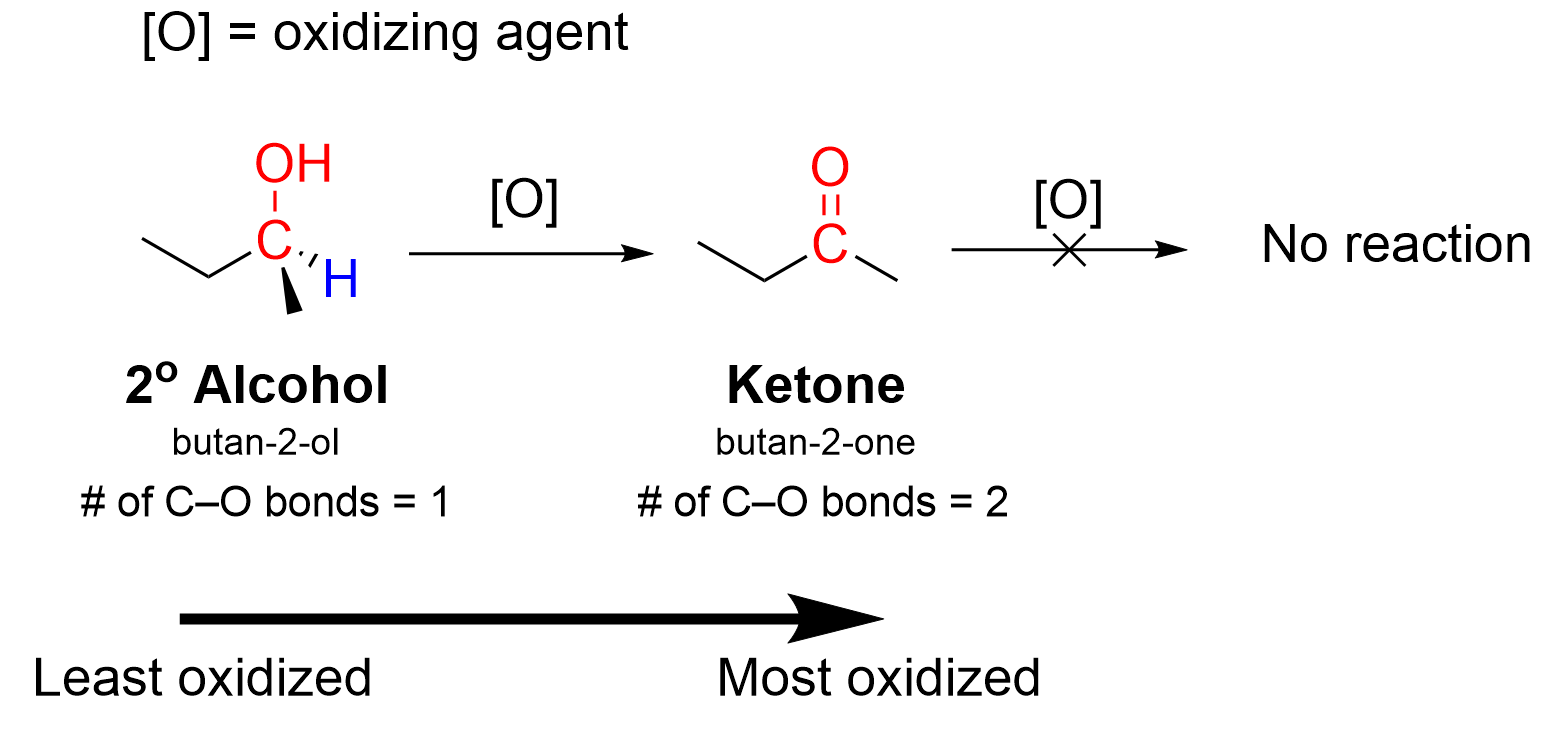
Curious to dip your toes into this world at home? While complex chemical synthesis is best left to professionals, you can explore related concepts safely. Many common household cleaners contain solvents that are, in fact, ketones. By researching the chemical makeup of these products and understanding their properties, you can gain an appreciation for how these compounds are used. For those with a keen interest in the scientific aspect, there are educational kits available that allow for safe and supervised demonstrations of oxidation reactions using more basic, readily available secondary alcohols. Always prioritize safety and follow instructions meticulously if attempting any home experiments.
Ultimately, the enjoyment of this particular chemical transformation lies in its ability to bridge the gap between the ordinary and the extraordinary. It's about appreciating the hidden complexity within everyday substances and harnessing that knowledge for creative expression. Whether you're a seasoned artist, a budding scientist, or simply someone who appreciates a good story, the journey of a secondary alcohol becoming a ketone is a testament to the fascinating and often beautiful world of chemistry that surrounds us.
