What Transition Metal Has The Smallest Atomic Radius

Get ready, folks, because we're about to dive into the super-duper, teeny-tiny world of atoms! Today, we're on a quest to find the absolute champion of smallness, the undisputed heavyweight in the bantamweight division of the periodic table. We're talking about transition metals, those dazzling darlings of chemistry that make our everyday lives so much more interesting.
Now, the periodic table is like a giant, super-organized closet of all the elements. And within this closet, there's a special section called the "transition metals." Think of them as the sparkly, colorful clothes – the ones that add a little pizzazz and a lot of function. They're famous for their incredible abilities to do all sorts of cool things, from conducting electricity like a champ to forming stunningly vibrant colors.
But we're not here to talk about their superpowers today, oh no! We're here to talk about their size. Imagine a bunch of balloons, all different sizes. Some are big and boisterous, others are more modest. We want to find the very, very smallest balloon in the transition metal bunch. This is serious business, people!
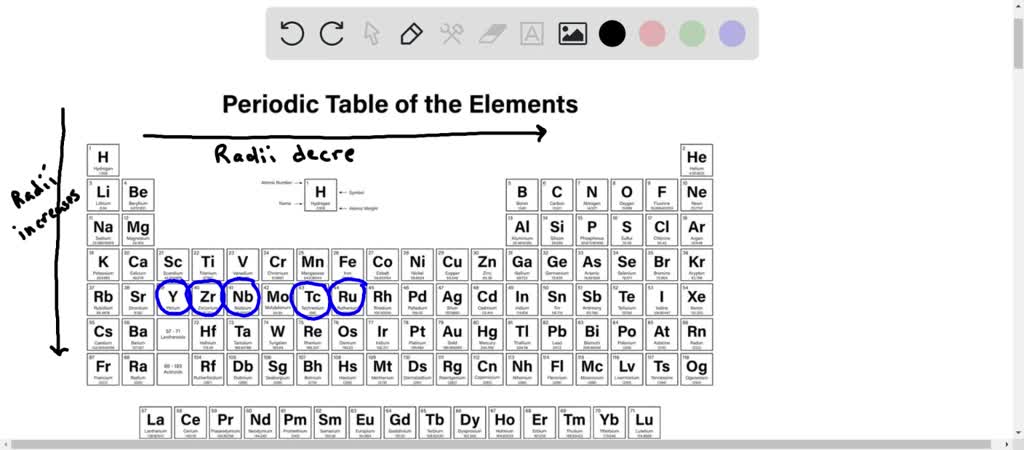
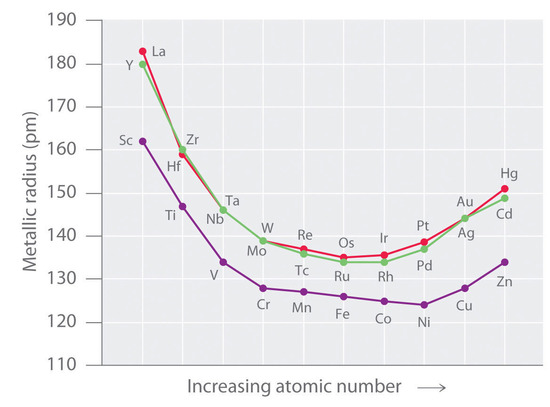
So, who is this microscopic marvel, this pint-sized powerhouse? Drumroll, please! It's none other than... Yttrium! Yep, that's right, Yttrium is the transition metal that takes the crown for having the smallest atomic radius. It's like the little engine that could, proving that good things really do come in small packages.
Now, you might be thinking, "Atomic radius? What's that?" Think of it like the distance from the very center of an atom (the nucleus, which is like the atom's cozy little home) to the edge of its electron cloud. It's basically how much space the atom takes up. And Yttrium, our little champ, just doesn't take up much space at all!
Imagine trying to pack a whole bunch of marbles into a small box. You want the smallest marbles so you can fit more in, right? Well, in the world of transition metals, Yttrium is like the perfectly petite marble that lets you cram in the most amazing chemistry possible into a tiny spot.
Let's just take a moment to appreciate the sheer adorableness of this. While some of its transition metal buddies are practically doing the atomic equivalent of a belly flop, stretching out and taking up all the room, Yttrium is just quietly, efficiently, and oh-so-neatly existing. It's the Marie Kondo of the transition metals, tidying up its electron cloud and keeping things super compact.
So, why is Yttrium so darn small? It's all about the tug-of-war happening inside the atom. You've got the positively charged protons in the nucleus pulling on the negatively charged electrons. The more protons you have, the stronger that pull, and the tighter the electrons are held. Yttrium, with its specific number of protons, just happens to have the perfect balance to keep its electrons snug as a bug in a rug.
It's like when you're at a party, and everyone's trying to get closer to the snack table. Some people are pushing and shoving, trying to get the biggest scoop. But Yttrium is the person who politely, but firmly, secures their spot right at the front, without needing to be overly dramatic or physically imposing. It's all about efficiency and a well-placed gravitational pull!
Think about it. We're talking about atoms here, which are already mind-bogglingly small. We're talking about numbers so tiny they make your brain do a little happy dance of disbelief. And within that microscopic universe, Yttrium is the undisputed king of compact. It's the ninja of the periodic table, silent, efficient, and incredibly small.
While other transition metals might be busy showing off their expansive electron clouds, like a peacock with its tail feathers spread wide, Yttrium is the quiet, unassuming individual who has mastered the art of fitting in. It's the little black dress of transition metals – classic, elegant, and always perfectly proportioned.
And the best part? This tininess doesn't stop Yttrium from being incredibly useful. Oh no! This little guy plays a starring role in some seriously cool technologies. Think about the vibrant red color in your TV screen – yep, Yttrium is often involved in making those colors pop! It's also found in those super-bright lights we use for things like stadium floodlights and even in some high-tech lasers.

It’s like having a tiny superhero who can lift incredibly heavy loads, or a miniature artist who can paint the most breathtaking masterpieces. Yttrium proves that you don't need to be big to be mighty. In fact, sometimes, being small is exactly what makes you so powerful and so special.
So next time you're marveling at the colors on your screen or enjoying the brilliance of a bright light, give a little nod to Yttrium. This humble, pint-sized transition metal is out there, working its magic, all while being the teeniest, tiniest titan of its group. It's a true testament to the wonders of the atomic world, reminding us that even the smallest things can have the biggest impact.
Isn't that just the coolest? To know that this unassuming element, Yttrium, is the smallest among a whole family of fascinating metals, yet it contributes so much. It’s like finding a tiny, perfect pebble on a beach filled with grander rocks, and realizing that pebble has a unique beauty and importance all its own. Yttrium, you little champion, we salute your compact magnificence!
So, the next time someone asks you about transition metals and their sizes, you can confidently declare: "It's Yttrium! The undisputed, teeny-tiny king of the atomic radius!" You'll sound like a chemistry whiz, and you'll be absolutely right. It’s a fun fact that’s as small and as brilliant as Yttrium itself.
And just for fun, imagine if other transition metals were trying to be like Yttrium. They'd be doing all sorts of contortions, trying to squeeze themselves smaller. But Yttrium just naturally does it, effortlessly. It's like a ballet dancer compared to someone trying to do a handstand for the first time – pure, natural grace in its minuscule form.
So there you have it, the secret's out! The transition metal with the smallest atomic radius is Yttrium. It’s a tiny giant, a microscopic marvel, and a testament to the fact that size isn't everything. In fact, in the case of Yttrium, being small is its superpower. It’s a feel-good story for the periodic table, a reminder that even in the grand scheme of elements, the little guys can shine the brightest. So let's all give a cheer for Yttrium, the smallest and perhaps the most subtly brilliant of the transition metals!
