What Structures Are Formed When Water Molecules Surrounds Individual Ions

Ever stare at a glass of water, maybe with a pinch of salt or sugar in it, and wonder what’s really going on in there? It’s not just a boring old liquid, oh no. It’s a miniature party, a microscopic mosh pit, and water molecules are the enthusiastic partygoers making it all happen. Today, we’re diving deep (pun intended!) into the super-cool structures that form when these energetic water guys decide to surround individual ions. Think of it as water molecules throwing a personalized welcome party for each charged particle that swims into their neighborhood.
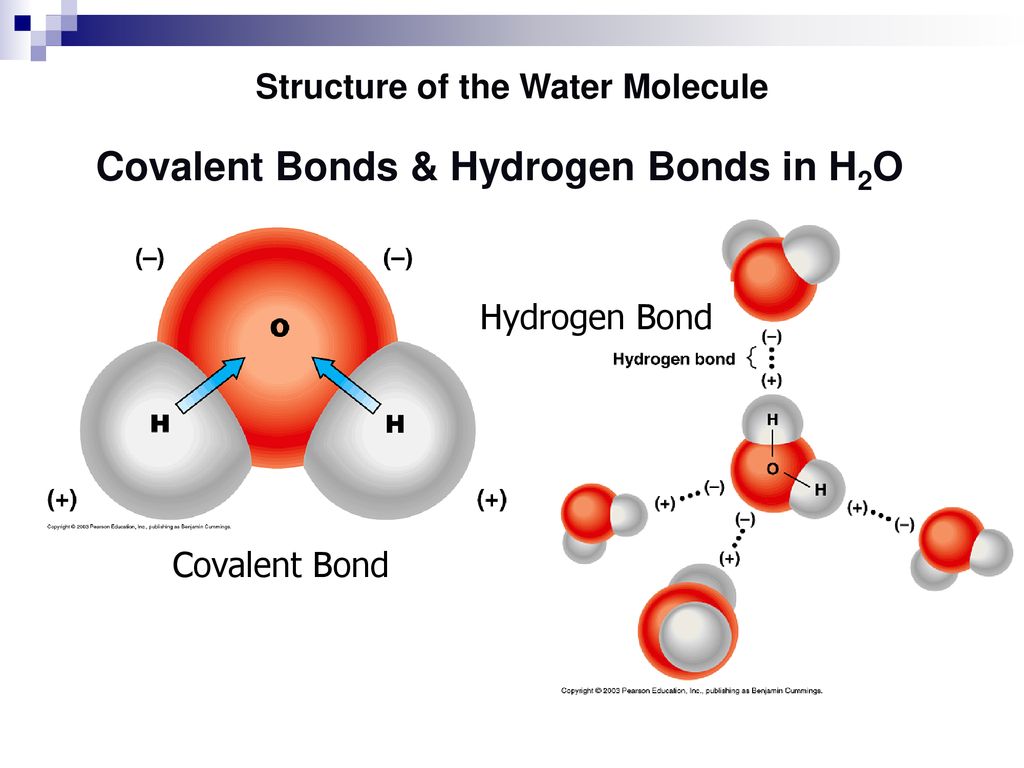
Let’s start with the star of the show: the water molecule. Picture it like a tiny Mickey Mouse head. You’ve got the oxygen atom as the head, and then two hydrogen atoms sticking out like ears. Now, here’s the juicy bit: this Mickey Mouse isn't perfectly balanced. The oxygen end is a bit like a grumpy teenager who's a little stingy with their electronics – it hogs the electrons, making it slightly negative. The hydrogen ends, on the other hand, are like those friends who are always a bit too generous, always sharing their snacks – they have a slight positive charge.
This charge difference, even though it’s super small, is what makes water so special. It’s like a tiny, built-in magnet. And when you toss some salt (sodium chloride, NaCl) into water, things get really interesting. Salt, as you know, is made of sodium ions (Na+) and chloride ions (Cl-). These guys are like the celebrities walking into the party, and the water molecules are all clamoring for their attention.
Imagine you’re at a buffet, and there’s a super popular celebrity chef. Everyone wants to get a selfie, right? The water molecules are doing the same thing, but instead of selfies, they’re forming little hydration shells around the ions. It’s like each ion gets its own entourage of water molecules.
Let’s focus on the positive celeb: the sodium ion (Na+). Remember how the hydrogen ends of water are a little positive? Well, opposites attract! So, the negative oxygen ends of the water molecules are going to get cozy with the positive sodium ion. They’ll practically be hugging it, forming a sphere of water molecules all pointing their oxygen “ears” towards the Na+.
It’s like a bunch of enthusiastic fans, all holding out their phones (the oxygen atoms) to get a picture with their favorite star (the sodium ion). They’re not just randomly there, either. They arrange themselves in a pretty organized way, creating a hydration shell around the sodium. This shell is usually just one layer thick, but it’s a tight group, like a celebrity security detail.
Now, let’s switch gears to the negative celeb: the chloride ion (Cl-). This one’s got a negative vibe, like a misunderstood artist. And who’s going to be drawn to that? Yep, the positive hydrogen ends of the water molecules! They’re going to flock to the chloride ion, like moths to a flame, or like puppies to a dropped piece of bacon.
So, the water molecules surround the chloride ion, but this time, their hydrogen “ears” are pointing inwards, towards the Cl-. It’s another hydration shell, but with the water molecules oriented in the opposite direction compared to when they surround a positive ion. Think of it as a different kind of fan club, with a different pose for the camera.
These hydration shells are not just for show. They’re crucial for keeping the ions dissolved in the water. Without them, the positive and negative ions would just stick back together, and your salt would un-dissolve, leaving a gritty surprise at the bottom of your glass. It’s like the water molecules are saying, “Nope, not on my watch! You’re staying separate and mingling!”
The number of water molecules that surround an ion depends on a few things. The main factor is the charge density of the ion. This is basically how much charge is packed into a given size. Smaller ions with a higher charge are like super-compact celebrities. They have a more intense gravitational pull (electrostatic attraction) and can attract more water molecules to form a larger, more robust hydration shell. Think of a petite pop star with a massive, screaming fan base versus a more laid-back indie musician with a smaller, dedicated following.
For example, a small, highly charged ion like magnesium (Mg2+) will have a very tightly packed and extensive hydration shell. The water molecules are practically plastered to it, struggling to get any closer. It’s like trying to get backstage at a Beyoncé concert – a serious mission!

On the other hand, a larger ion with a lower charge, like potassium (K+), will have a less dense hydration shell. The water molecules are still there, but they’re a bit more spread out, more like casual acquaintances than intense superfans.
This whole process is called hydration. It’s the reason why many ionic compounds dissolve in water. The water molecules essentially "insulate" the ions from each other, preventing them from rejoining and crystallizing. It’s like water is the ultimate peacemaker, or perhaps the ultimate bouncer, keeping the ions in their designated spots.
Think about making a cup of tea. You add sugar, which is made of sucrose molecules, not ions. But let’s imagine for a second. If it were ions, the water molecules would be surrounding each positively charged bit and each negatively charged bit. For sugar, it’s a bit different; the water molecules surround the whole sugar molecule, pulling it apart and dispersing it. Still a party, just a different kind of dance.
The hydration shells also influence the properties of the solution. For example, they can affect how easily the ions move around in the water. Ions with larger, more tightly bound hydration shells tend to move slower because the water molecules are essentially "dragging" them along. It’s like trying to run through a crowd of people trying to get autographs versus running through an empty field.

And it's not just about salt. This hydration phenomenon happens with all sorts of ionic compounds, from the calcium in your milk to the potassium in your bananas. It's a fundamental process that allows life as we know it to exist. Our bodies are mostly water, and all those essential electrolytes, like sodium and potassium, are dissolved and doing their jobs thanks to these tiny water molecule entourages.
So, next time you’re sipping on something that’s been dissolved in water, take a moment to appreciate the microscopic dance party happening within. The water molecules, with their quirky positive and negative ends, are diligently working, forming hydration shells, keeping everything separate and functional. They are the unsung heroes of dissolution, the invisible DJs of the ionic world.
It’s a bit like a busy restaurant kitchen. You’ve got all these ingredients (the ions) that need to be prepared and served. The water molecules are the chefs and waiters, carefully plating each ingredient, making sure it’s presented correctly and doesn’t interact with the wrong neighbors before it’s time. They are the culinary artists of the molecular world!
The interactions between water molecules and ions are also pretty dynamic. These hydration shells aren’t static; they are constantly forming and breaking. Water molecules are zipping around, swapping places, like a really energetic dance floor where partners are changing every second. But the overall structure, the organized shell, tends to persist as long as the ion is present and the water is around.
This dance is governed by the principles of electrostatics – the attraction between opposite charges. It’s the same force that makes a balloon stick to your hair after you rub it, or keeps the planets in orbit around the sun (though on a much smaller scale, of course!). Water’s polar nature, that uneven distribution of charge, is its superpower, allowing it to interact with and dissolve so many different substances.
So, when you’re watering your plants, or even just sweating on a hot day, remember that water isn't just a passive substance. It’s an active participant, constantly forming these amazing structures to keep things in balance. It’s like the ultimate organizer, ensuring that charged particles get their personal space and can go about their business without causing molecular mayhem.
It’s a beautiful, intricate dance of attraction and organization. The humble water molecule, with its seemingly simple structure, is responsible for so much of the chemistry that makes our world work. And the way it surrounds and stabilizes ions is just one of its many, many talents. It’s like the Swiss Army knife of molecules!
Next time you see water, don't just see H2O. See the energetic partygoers, the tireless organizers, the microscopic celebrity entourage managers. They’re the reason your sports drink tastes like, well, a sports drink, and why the ocean isn’t just a giant, undissolved salt crystal. It's all thanks to the amazing hydration shells formed around individual ions.
It's a fascinating glimpse into the unseen world, a world where even a simple glass of water is a bustling metropolis of molecular activity. And it all boils down to the clever way water molecules embrace charged particles, creating order out of potential chaos. Pretty neat, huh?
