What Role Do Electrons Play In Dehydration Synthesis And Hydrolysis
Ever felt that pang of hunger after a long day, or maybe that satisfied sigh after a delicious meal? Well, believe it or not, the tiny, zippy little things called electrons are playing a starring role in how your body handles all that food. It’s not just some complex chemistry mumbo-jumbo; it’s happening right inside you, all the time, and it's surprisingly down-to-earth when you break it down. Think of electrons as the ultimate matchmakers and divorce lawyers of the molecular world, and they’re super busy in two fundamental processes: dehydration synthesis and hydrolysis. These might sound like fancy terms from a Mad Libs gone wrong, but they're actually about building stuff up and breaking stuff down, just like you might build a LEGO castle and then, oops, accidentally kick it over.
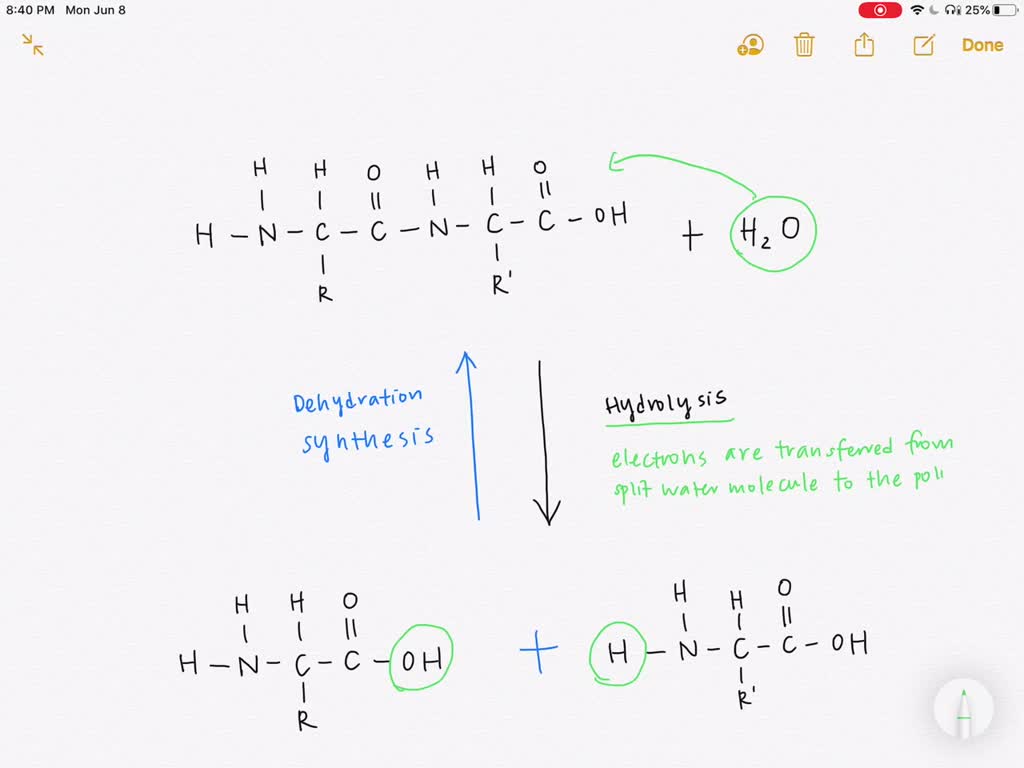
Let's start with dehydration synthesis. Imagine you have two tiny LEGO bricks, let's call them "Molecule A" and "Molecule B." These guys are happily minding their own business. Dehydration synthesis is like you, the master builder, deciding to connect these two LEGOs together. But here's the twist: to make them stick, you need to get rid of something. In the molecular world, that "something" is often a little water molecule. So, we're literally synthesizing (making something) by dehydrating (removing water).
Now, where do our electron buddies come in? Electrons are, at their core, about sharing and transferring energy. Think of them like tiny little energetic puppies. When Molecule A and Molecule B are getting ready to link up, their electrons are like, "Hey, let's get together and form a stronger bond! We’ll share our energy, and this will be awesome!"
Specifically, in dehydration synthesis, the electrons in the outer shells of atoms in Molecule A and Molecule B rearrange themselves. They get excited, so to speak, and start getting cozy. As they start to form new connections – a new bond that holds A and B together – a little piece of each molecule, a hydrogen atom from one and a hydroxyl group (an oxygen and a hydrogen) from the other, decide to go off and form a water molecule. It’s like they’re saying, "Okay, you two are going to be best friends now, and we'll just go make a little baby water molecule and scoot."
This is a super common process in your body. Think about building proteins from smaller units called amino acids. Each amino acid is like a LEGO brick. When your body needs to build a muscle, for example, it links up thousands of these amino acids through dehydration synthesis. The electrons are the unsung heroes, orchestrating the bond formation and the release of that tiny water molecule. It’s a chemical handshake, powered by electron rearrangement.

You might also see this happening when your body builds carbohydrates, like the complex sugars that give you energy. Simple sugar molecules link up, again, with the help of electron sharing and a little water being kicked out. So, every time you eat something that needs to be built up – from the complex molecules that make your hair shiny to the energy stores in your liver – those electrons are on the job, making sure the connections are solid.
It’s a bit like when you’re at a party, and two people are about to hit it off. Their initial conversations (electron sharing) lead to a deeper connection (a covalent bond), and maybe, as a side effect, someone else leaves to grab another drink (the water molecule). The party (your body) keeps going, with new connections being formed!
On the flip side, we have hydrolysis. This is the exact opposite of dehydration synthesis. If dehydration synthesis is about building, hydrolysis is about breaking down. Think of it as the molecular divorce lawyer, or perhaps the meticulous chef chopping up ingredients. The name itself gives it away: hydro- meaning water, and -lysis meaning to break apart. So, we’re literally breaking something apart using water.

This is crucial for digestion. When you eat a big sandwich, your body can't just absorb the whole thing. It needs to break down the complex carbohydrates into simpler sugars, the proteins into amino acids, and the fats into their smaller components. Hydrolysis is the star of this show.
How do electrons get involved here? Well, remember how electrons were busy making strong bonds in dehydration synthesis? Now, in hydrolysis, those strong bonds need to be broken. And what’s the secret weapon for breaking these bonds? Yep, you guessed it – a water molecule. But it's not just about the water itself. It's about how the electrons in the water molecule interact with the molecule being broken down.
When a water molecule approaches a bond between, say, two linked amino acids in a protein, its electrons get a little agitated. The oxygen atom in water, which is a bit greedy for electrons, can attract the electrons in the bond of the protein. Simultaneously, a hydrogen atom from the water can get involved on the other side.

Think of the bond between the two amino acids as a tug-of-war rope. The water molecule comes in, and its electrons help to weaken that rope. One part of the water molecule (the oxygen side) might pull a bit from one amino acid, while the hydrogen part of the water molecule attaches to the other amino acid. Essentially, the water molecule inserts itself into the bond, breaking it. The electrons in the water molecule are key players in facilitating this disruption.
It's like having a stubborn knot in your shoelaces. You can try to pull it with all your might, but it’s tough. Then, someone suggests using a little bit of water to loosen it up. The water seeps in, and the fibers of the shoelace (the electrons) shift, allowing you to finally untie that pesky knot. The water molecule, with its electron configuration, is the catalyst for this loosening.
Every time you digest food, hydrolysis is happening on a massive scale. Those complex carbohydrates from your pasta? Broken down into glucose. Those proteins from your chicken? Broken down into amino acids. Those fats from your olive oil? Broken down into fatty acids and glycerol. And in every single one of these breaking-down steps, electrons are playing a crucial role in how the water molecule interacts to sever the chemical bonds.

It’s not just about eating, either. Your body constantly needs to recycle and break down old or damaged molecules to make new ones. For instance, when your body breaks down old red blood cells, hydrolysis is involved. When your muscles need energy from stored glycogen, hydrolysis is used to break down that glycogen into glucose. It’s the constant cycle of building and dismantling that keeps you going, and electrons are the essential currency of these reactions.
So, the next time you’re enjoying a meal, or even just feeling that energy surge after a good night’s sleep, give a little nod to the invisible world of electrons. They’re the tireless workers, the meticulous organizers, and the efficient dismantlers, all working in concert through dehydration synthesis and hydrolysis to keep your incredible body running like a well-oiled, molecule-building, molecule-breaking machine. They’re not just orbiting nuclei; they’re the architects and demolition crews of your very existence!
It’s pretty wild to think about, right? That these tiny, almost imperceptible particles are responsible for everything from the energy you use to walk to the very structure of your cells. They’re the ultimate go-getters of the chemical universe. Without their ability to shift, share, and facilitate the involvement of other molecules like water, we’d just be a pile of unconnected atoms, not a vibrant, living, breathing organism. So, next time you feel a bit hungry or satisfied, remember the electrons hard at work, making all the magic happen, one bond at a time.
