What Period 3 Element Having The Following Ionization
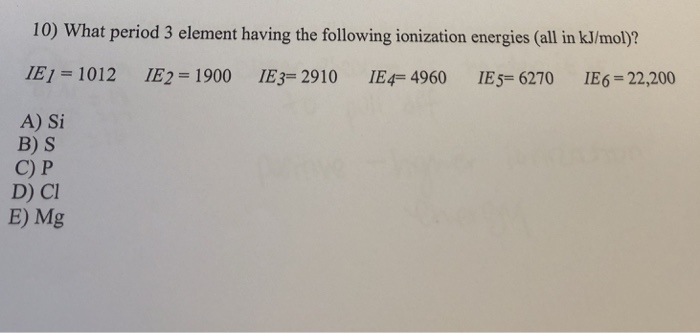
Welcome, fellow curious minds and budding chemists! Ever found yourself staring at a periodic table, wondering about the hidden personalities of those elemental squares? Today, we’re diving into a fun little game of elemental detective work, specifically focusing on a particular Period 3 element. If you enjoy a good puzzle, a bit of scientific deduction, or just want to impress your friends with some fascinating trivia, then this is for you! It’s like a crossword puzzle, but with atoms, and trust me, the rewards are just as satisfying.
So, what’s the big deal about ionization, you ask? Well, ionization energy is essentially the amount of energy required to remove an electron from an atom. Think of it as the atom’s “grip strength” on its electrons. This property is incredibly important because it dictates how an element will behave in chemical reactions. Understanding ionization energy helps scientists predict how elements will bond, what kind of compounds they'll form, and even how they'll conduct electricity or heat. This knowledge is the bedrock of countless everyday marvels, from the batteries in your phone to the aluminum foil in your kitchen, and even the silicon chips that power your computer.
Now, for our mystery element from Period 3 (that’s the third row of the periodic table, for those not yet fluent in chart-speak). We're looking for an element with a relatively high first ionization energy, but not the absolute highest. This element is a real workhorse in industry and has a truly remarkable range of applications. Think about the sleek, lightweight frames of your bicycle, the sturdy pots and pans you cook with, or even the windows you look out of – all thanks to this element! Its compounds are also vital in everything from antiperspirants to flame retardants. This element is none other than Aluminum (Al)!
Aluminum sits comfortably in Period 3. While not as difficult to ionize as elements further to the right (like chlorine or argon), it requires a significant amount of energy to pull away one of its electrons. This characteristic is what makes it so useful. It’s reactive enough to form strong, stable compounds, but not so reactive that it spontaneously combusts. Its metallic nature, combined with its ability to form a protective oxide layer, makes it exceptionally resistant to corrosion, a dream for engineers and manufacturers alike.

To enjoy this elemental mystery even more, try visualizing the process. Imagine electrons as tiny planets orbiting a central sun (the nucleus). Ionization energy is the energy needed to launch a planet into deep space, away from its star. When you look at the periodic table, you can see trends in this “launching energy.” For Period 3, as you move from left to right, the ionization energy generally increases because the nucleus’s pull gets stronger. Aluminum, being in the middle-ish of Period 3, has that sweet spot of reactivity that’s so beneficial.
Here are some practical tips for your elemental adventures: Firstly, get your hands on a periodic table! Keep it handy while you’re reading about chemistry or even watching documentaries. Secondly, don't be afraid to look up the ionization energies for different elements. Comparing them can be incredibly illuminating. Finally, connect the dots between the properties you're learning about and the objects you encounter daily. The next time you see an aluminum can or a piece of foil, you’ll know that its very nature, its chemical personality, is a direct result of its ionization energy and its place on that fascinating grid of elements.
