What Organic Functional Group Does Terephthalic Acid Contain

Hey there, curious minds! Ever wondered about the building blocks of some of the stuff we use every day? Like, you know, that super useful plastic that makes bottles and fabrics? Well, today we're diving into the fascinating world of terephthalic acid. Sounds a bit science-y, right? But trust me, it's more like a cool puzzle than a dry textbook chapter. And the best part? We're going to figure out its secret identity – its main organic functional group! Think of it like a superhero having a secret superpower. What's terephthalic acid's?
So, what even is terephthalic acid? Imagine a molecule, a tiny little structure made of atoms all linked up. Terephthalic acid is one of these molecular architects. It's not something you'd find just lying around in your fruit bowl, but it's a super important ingredient in making things like PET plastic, which you'll find in everything from your water bottles to your cozy fleece jackets. Pretty neat, huh?
Now, when we talk about "organic functional groups," what are we really talking about? Think of it like this: a molecule is like a Lego creation. The main body of the Lego creation might be all the same type of brick, but the "functional group" is like a special, uniquely shaped Lego piece that gives it a specific job or property. It’s the part that really makes the molecule do things. It's where the magic happens, chemically speaking.
So, what makes terephthalic acid tick? What's its special Lego piece? Let's break it down. If you were to draw out the structure of terephthalic acid, you'd see a few key things. There's a ring structure, kind of like a hexagon, made of carbon atoms. That's pretty common in organic chemistry. But then, attached to that ring, are some other bits. And those are the bits that tell us about its functional groups.
The Big Reveal: What's the Star Player?
The main, and arguably most important, functional group in terephthalic acid is the carboxylic acid group. You might have heard of acids before, right? Like lemon juice is acidic. Well, in chemistry, a carboxylic acid group has a very specific structure: it's a carbon atom double-bonded to one oxygen atom and single-bonded to another oxygen atom, which in turn is bonded to a hydrogen atom. It looks like this: -COOH. It’s a super versatile group!
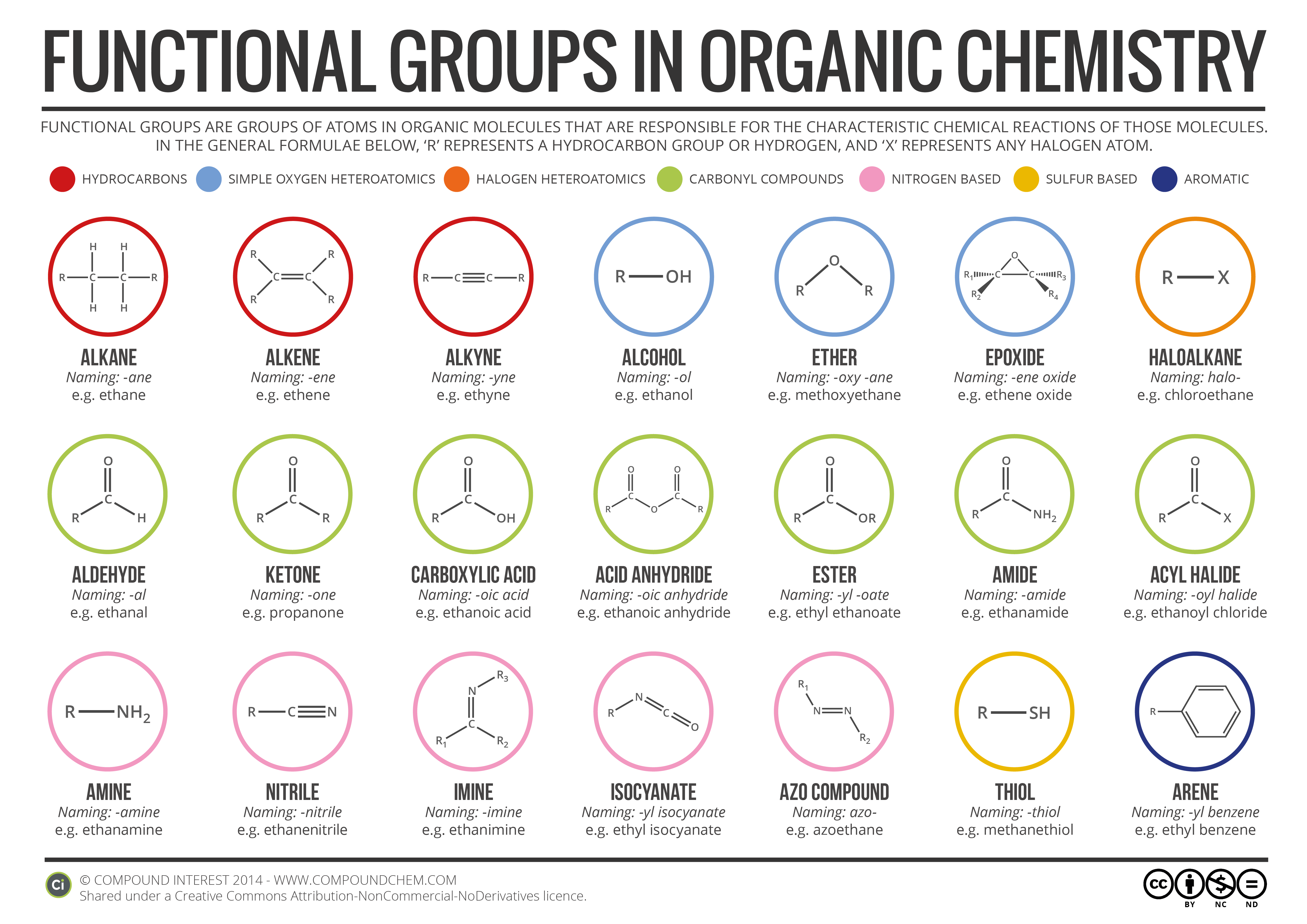
Imagine it as a little chemical appendage. This -COOH group is like the business end of the molecule. It's where a lot of the reactivity happens. It can donate a hydrogen ion, which is what makes it "acidic." It's also what allows terephthalic acid to link up with other molecules and form those long chains that make up polymers like PET. Think of it like a tiny connector that can join up with other connectors to build something much bigger.
But wait, there's a twist! Terephthalic acid doesn't just have one of these amazing carboxylic acid groups. It actually has two of them! And they're positioned on opposite sides of that central ring structure. This "di-" prefix is important – it means "two." So, we have a dicarboxylic acid. This is a big deal because having two of these reactive groups allows terephthalic acid to be a fantastic building block for making polymers. It can connect on both ends, forming long, sturdy chains.

Think of it like having two hands ready to hold onto things. If it only had one hand, it could only build a line. But with two hands, it can build intricate networks and long chains, which is exactly what you need to create plastics and other advanced materials. This symmetry and dual functionality are what make terephthalic acid so incredibly useful.
Why is This Functional Group So Cool?
So, why should we be excited about a carboxylic acid group? Well, it's one of the workhorses of organic chemistry! It's found in so many important molecules. Think about amino acids, the building blocks of proteins. They have carboxylic acid groups! Even things like acetic acid (that's vinegar!) have this group. It's a sign of a molecule that can participate in a lot of interesting reactions.
For terephthalic acid, having those two carboxylic acid groups is like having a superpower multiplied by two! This allows it to react with other molecules, like diols (which are molecules with two alcohol groups), in a process called esterification. This is how you get PET plastic. The carboxylic acid groups from terephthalic acid link up with the alcohol groups from the diol, forming ester linkages. It's like a chemical handshake that creates a strong bond.

Imagine you're building a bridge. Terephthalic acid is like a super-strong beam that has attachment points on both ends. When you combine it with other beams (like diols), you can build a really long, robust bridge. This is essentially what happens when you make PET. You're linking up lots and lots of these terephthalic acid "beams" with other molecules to create that familiar plastic material.
The fact that it has two carboxylic acid groups, and they're positioned so nicely on the ring, makes it particularly good for creating linear polymers. These are the long, chain-like molecules that give plastics their strength and flexibility. Without this specific arrangement and the presence of those two crucial carboxylic acid groups, we wouldn't have many of the plastics we rely on today.
More Than Just Plastic?
While its role in PET production is huge, terephthalic acid and its dicarboxylic acid nature have other applications too. It's used in the production of polyester fibers, which are super common in clothing, home furnishings, and industrial textiles. Think of those durable, wrinkle-resistant fabrics you love? Yep, terephthalic acid is often a key ingredient.
It's also used in making other types of polymers, like certain alkyd resins which are found in paints and coatings. So, even the paint on your walls might have a little bit of terephthalic acid's magic in it! It's quite amazing how one molecule, with its distinct functional groups, can be so versatile and find its way into so many different aspects of our lives.
So, the next time you grab a bottle of water or pull on a comfy fleece, you can give a little nod to terephthalic acid and its awesome carboxylic acid groups. It's a reminder that even the most ordinary objects around us are built from fascinating molecular structures, and understanding their functional groups is like unlocking their secret powers. It’s a little bit of chemistry magic that makes our modern world possible!
