What Orbitals Overlap To Create The H-c Bond In Ch3+
Ever found yourself staring at a chemical formula, like CH3+, and wondering, "What's really going on in there?" It's easy to get lost in the shorthand, but behind those letters and charges lies a fascinating world of atomic interactions. Today, we're going to peek into the heart of the methyl cation, CH3+, and uncover the secrets of how its carbon-hydrogen bonds are formed. It’s a journey into the realm of quantum mechanics, but don't worry, we'll keep it light, curious, and surprisingly relevant!
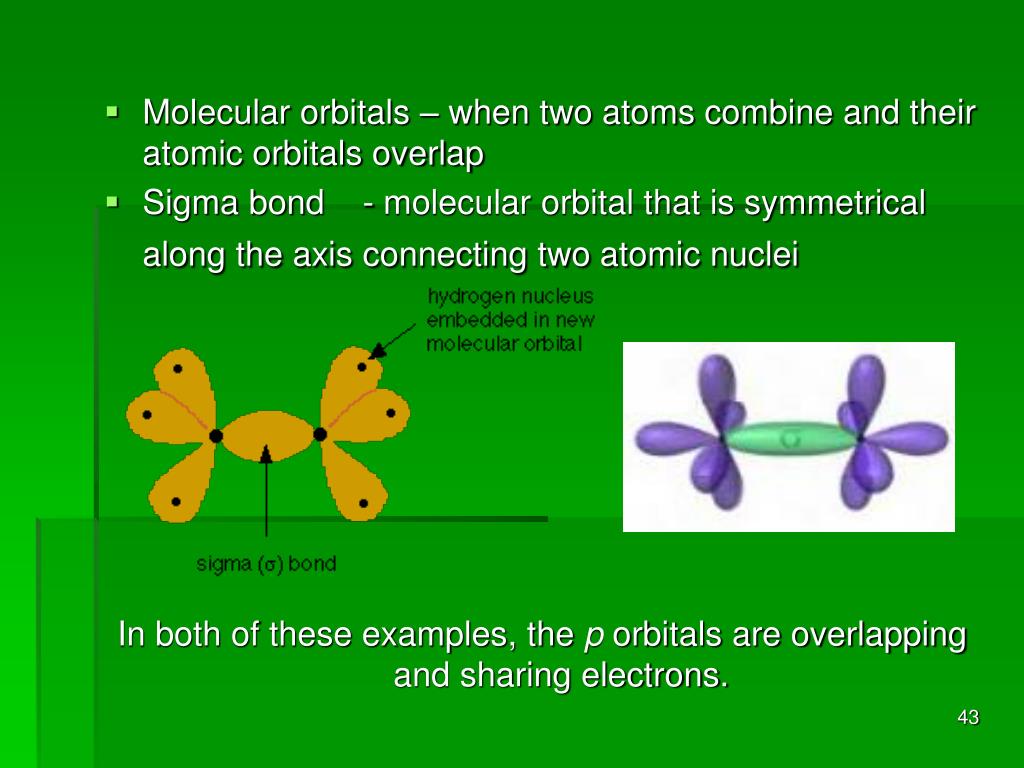
Understanding how atoms bond is fundamental to chemistry. It's like learning the alphabet before you can read a book. When we talk about CH3+, we're looking at a molecule with a positive charge, which means it's a bit electron-deficient and quite reactive. The question is, how does that central carbon atom, with its limited electron-sharing capacity, manage to hold onto those three hydrogen atoms? The answer lies in the magical concept of orbital overlap. Think of atomic orbitals as fuzzy clouds of probability where electrons hang out. When atoms get close enough to bond, these clouds can merge, creating a stronger, more stable connection.
So, what specific clouds are doing the mingling to create those H-C bonds in CH3+? The central carbon atom, in its typical bonding behavior, uses its s and p atomic orbitals. To form these three identical bonds with hydrogen, the carbon atom undergoes a process called hybridization. It mixes one 2s orbital and three 2p orbitals to create four new, identical orbitals called sp3 hybrid orbitals. These sp3 orbitals are arranged in a tetrahedral fashion around the carbon atom, pointing outwards like the spokes of a wheel. Each of these sp3 hybrid orbitals then overlaps with the 1s orbital of a hydrogen atom, which is a simple, spherical cloud. This overlap is what forms the strong sigma (σ) bond between the carbon and each hydrogen atom. It’s a beautiful dance of electron clouds coming together!
Why is this useful? Well, understanding orbital overlap helps chemists predict the shape and reactivity of molecules. For CH3+, its positive charge and sp3 hybridization make it a key intermediate in many organic reactions. It’s like a fleeting, charged handshake between atoms that sets the stage for further chemical transformations. This knowledge is crucial in fields like drug design, material science, and understanding complex biological processes. Even in everyday life, the principles of bonding underpin everything from the plastic in your water bottle to the fuels that power your car.

For students, visualizing this orbital overlap can be a game-changer. Instead of just memorizing shapes, you start to understand why they have those shapes. In education, molecular modeling kits and even simple diagrams showing overlapping spheres and dumbbells can make these abstract concepts tangible. In a daily context, think about how different materials have different properties. This is directly related to how their atoms are bonded and the types of orbitals involved!
Want to explore this curiosity further? You can find fantastic 3D molecular visualization software online that allows you to rotate and examine molecules like CH3+ up close. Even sketching out the orbitals with colored pens on paper can help solidify the concept. Don't be afraid to ask "why" and "how" – that's where the real fun of chemistry begins!
