What Minimum Frequency Of Light Is Required To Ionize Boron
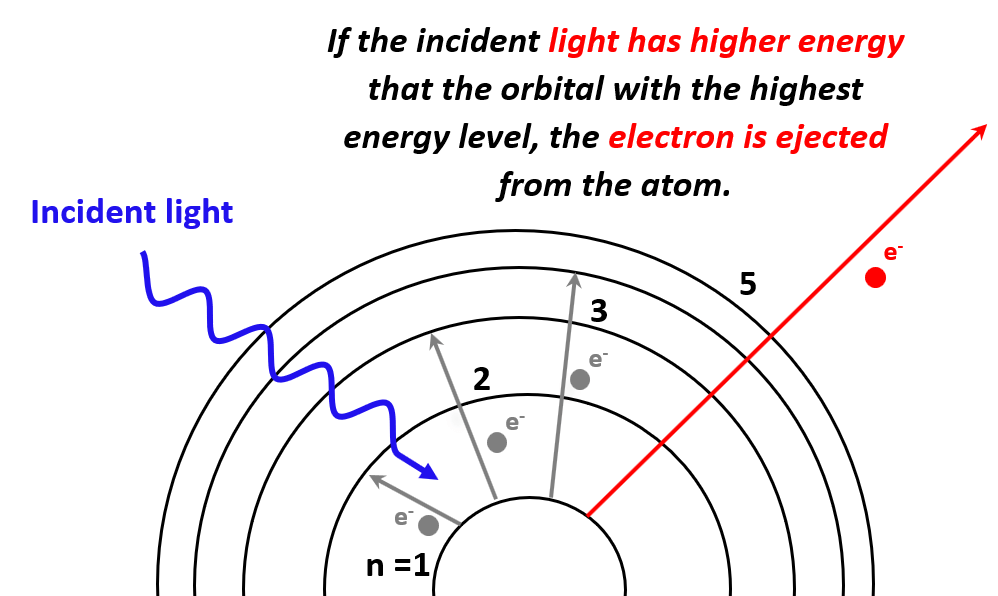
Alright, so imagine this: you're at a cosmic café, right? And you've got this little dude, Boron. He's not exactly the life of the party, more of a quiet type, hanging out with his electrons like they're his prized collection of vintage stamps. Now, Boron, bless his little metallic heart, has his electrons held on pretty darn tight. We're talking a grip tighter than a toddler with a cookie they just snagged. So, if you want to have a little fun and, you know, ionize him – which is just a fancy word for kicking one of his electrons to the curb – you can't just whisper sweet nothings at him.
Nope. You gotta bring the thunder. And by thunder, I mean light. But not just any light. You can't just wave your phone flashlight at Boron and expect him to shed an electron. That's like trying to open a bank vault with a toothpick. It just ain't gonna happen.
We're talking about the minimum frequency of light required. Think of frequency as how energetic the light is. It's like the espresso shot of the light world. Low frequency is like a weak decaf; high frequency is a triple ristretto that’ll make your eyeballs vibrate. For Boron, we need something with a bit of a kick, but not so much that it blows his whole atom to smithereens. We're aiming for a precise eviction notice, not a demolition crew.
Now, how do we figure this out? It’s not like there’s a handy dandy Boron Ionization Frequency chart tacked to the wall of the universe. (Though, wouldn't that be neat? Imagine a cosmic cafeteria with helpful signage!) We gotta do a little bit of science. And when I say "we," I mean some very clever folks who spent a lot of time staring at stuff and scribbling equations.
The key concept here is the ionization energy. This is the amount of energy you need to completely remove an electron from an atom. For Boron, this magic number is around 8.30 electronvolts (eV). Now, electronvolts are like the tiny little units of energy used for wimpy things like atoms. Think of it as trying to measure the weight of a feather in tons. It's a unit, but you need a lot of them for anything that actually matters in your everyday life.

So, we have the energy. But the question asks about frequency. Luckily, the universe has a pretty straightforward way of linking energy and frequency for light. It’s like a cosmic universal translator: E = hf. Don't let the letters scare you. 'E' is the energy (that's our 8.30 eV, converted into something more useful), 'f' is the frequency we're looking for, and 'h' is Planck's constant. Poor Max Planck, he was the guy who figured this out, probably while staring at his tea and having a "eureka!" moment. He’s basically the cosmic matchmaker for energy and frequency.
So, we plug in the numbers. We take that 8.30 eV, convert it into Joules (the more common unit of energy that doesn't make physicists cry), which is roughly 1.33 x 10-18 Joules. Then we divide by Planck's constant (which is a teensy tiny number, like 6.626 x 10-34 Joule-seconds – imagine trying to count all the grains of sand on Earth, then divide that by a gazillion). And voilà! We get a frequency. Drumroll, please…
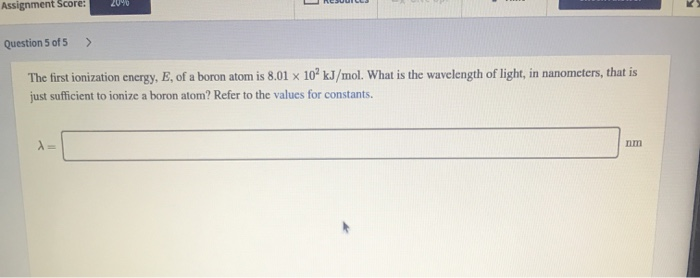
The minimum frequency required to tickle Boron’s electron right out of its cozy little shell is approximately 2.01 x 1015 Hertz. Hertz, by the way, is just cycles per second. So, that's over 2 quadrillion wiggles per second. That’s a lot of wiggling!
Now, what kind of light is that? Is it the gentle glow of a sunset? The blinding flash of a camera? Nope. This frequency is way, way up there on the electromagnetic spectrum. We're talking about ultraviolet (UV) light. Not the kind that gives you a nice tan, mind you. This is the gnarly, energetic UV that can actually damage your DNA if you’re not careful. It’s like the party animal of the light world, the one that’s a little too enthusiastic.

Think about it: Boron is like a very shy librarian, and his electron is his most treasured first edition. You need a certain level of… oomph… to make him let go. Visible light, the kind we see, is like a gentle murmur. It’s not enough to rouse him from his studious habits. Infrared? That's just warm fuzzies; Boron is immune. Radio waves? That’s like humming a lullaby; he’ll just fall asleep.
But UV light? That’s a high-energy concert. It’s got the punch to make that electron say, "Whoa, okay, that's enough excitement for me!" and decide to go on an adventure. This is why sunscreen is important, folks. It's not just to prevent sunburn; it's to act as a shield against this electron-evicting cosmic energy. Your skin cells are basically made of atoms that, if bombarded with enough high-frequency UV, might start losing their own precious electrons, and trust me, that's a party nobody wants to be at.
So, there you have it. The next time you're lounging in the sun (with adequate protection, of course!), remember that the light hitting you, while mostly harmless visible stuff, also contains a spectrum of energies. And somewhere in that energetic punch of UV light is the specific frequency that can convince a Boron atom, in its own quiet way, to finally let one of its electrons break free. It’s a testament to the universe’s incredibly specific, and sometimes rather forceful, ways of doing things. Who knew a little Boron and a lot of UV could be such a dramatic duo?
