What Mass Of Water Is Produced From The Complete Combustion
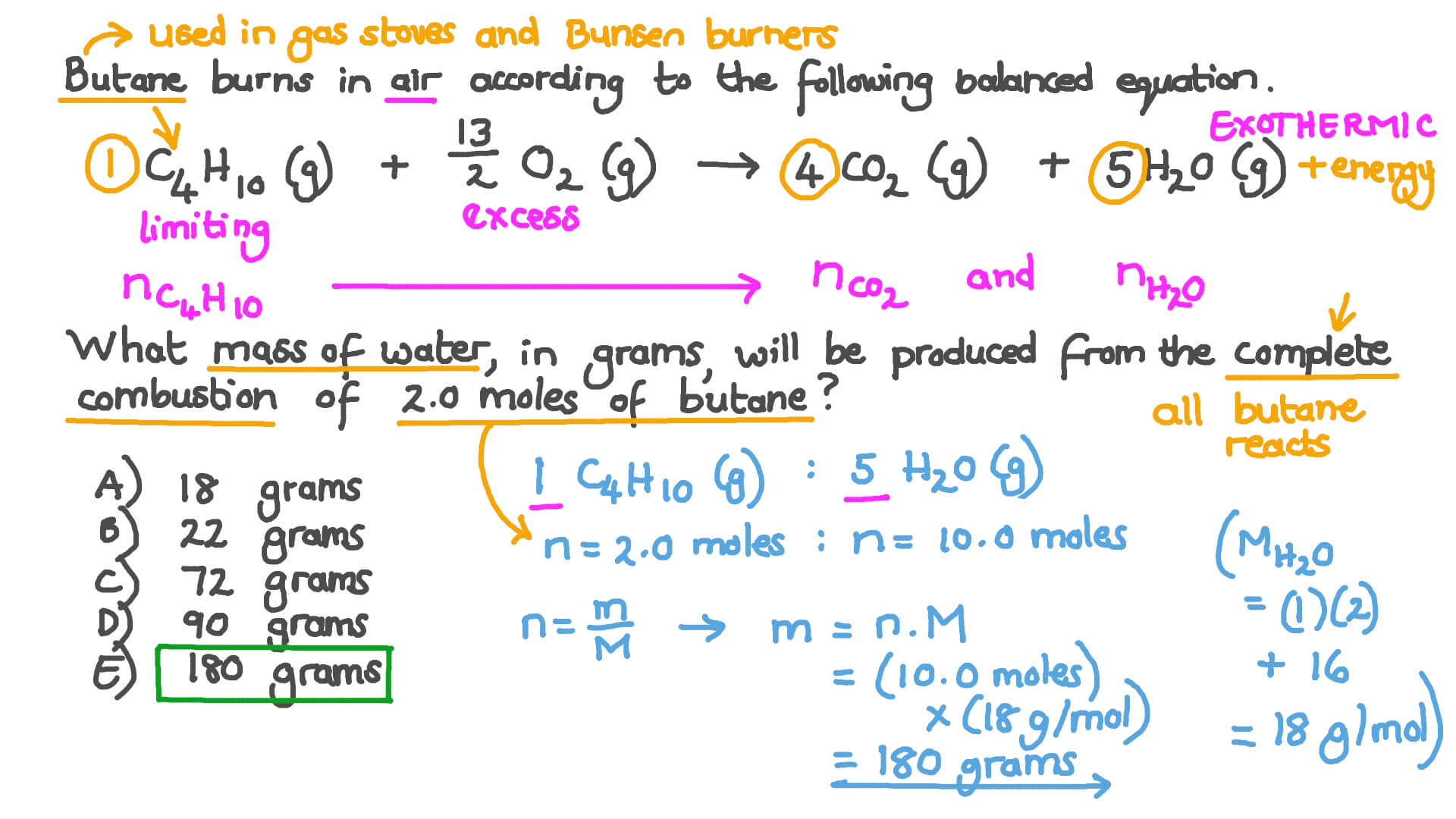
Get ready for a splash of science that's so cool, it'll make you want to dive right in! We're talking about something you see every single day, something so ordinary you might overlook its amazing magic. But hold onto your hats, because what we're about to uncover is going to be a real eye-opener. It’s like discovering a secret superpower hidden in plain sight!
Think about your cozy campfire, the cheerful hiss of your gas stove, or even the mighty roar of a car engine. What do all these everyday marvels have in common? They’re all busy little chefs, cooking up something truly spectacular. And what’s the secret ingredient they’re all using? It’s something so common, so readily available, you probably have gallons of it in your house right now!
That’s right, we’re talking about water! But not just any water, oh no. We’re talking about the water that pops into existence when something burns completely. It’s like a tiny, invisible water factory working overtime, and the results are surprisingly… well, wet! Imagine if you could bottle up that magic!
Now, you might be thinking, "Wait a minute, when things burn, don't they just turn into smoke and ash?" And usually, you'd be absolutely right! But here's where the "complete combustion" part swoops in like a superhero with a cape. This is when things get really, really efficient in the burning department. It’s like a perfectly orchestrated performance, with nothing left to chance.
When something like, say, a chunk of wood or a gulp of gasoline burns completely, it’s not just about heat and light. It’s also about a chemical dance that involves oxygen from the air and the stuff the fuel is made of. And in this dazzling dance, a rather surprising byproduct emerges: water! Yes, the very same stuff you drink, swim in, and use to brush your teeth.
So, the big question, the one that might have you scratching your head in delightful wonder, is: how much of this magical, invisible water gets produced? It’s not like you see puddles forming after a perfectly burning candle, right? Well, that’s because this water is born as a gas, like steam, and it happily mixes with the air. It’s a master of disguise, really!

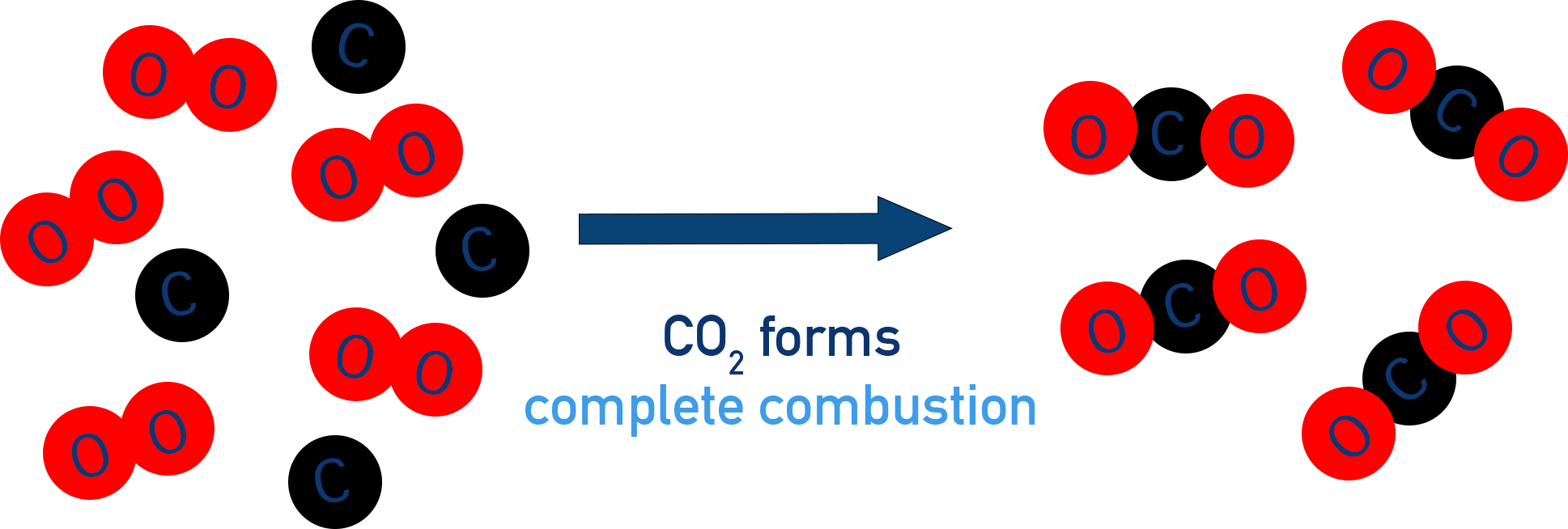
Let’s get a little bit into the nitty-gritty, but don’t worry, it’s going to be fun! Think about a simple molecule of methane, which is the main ingredient in natural gas. It’s made of one carbon atom and four hydrogen atoms. When this little fella decides to go through complete combustion, it teams up with oxygen.
The carbon atom is so happy to find oxygen, it snuggles up and forms carbon dioxide – that’s the invisible gas we breathe out. But the real star of our show is what happens to those four hydrogen atoms. They each grab an oxygen atom, and poof! Four little water molecules are born. It’s like a tiny chemical nursery creating pure H2O!
Now, here’s where the mass comes in, and it’s a concept that’s surprisingly easy to grasp. Think of it like counting LEGO bricks. Different atoms have different "weights" or masses. For instance, a carbon atom is heavier than a hydrogen atom. And oxygen is even heavier still.
When we look at how much stuff goes in versus how much stuff comes out, the total mass always stays the same. This is a fundamental rule of the universe, a bit like gravity, but for molecules. Nothing is lost, and nothing is magically gained. It’s all about rearranging the building blocks!

So, back to our methane. For every 12 units of mass from the carbon atom, we have 4 units of mass from the hydrogen atoms. When they go through combustion, they team up with oxygen. The carbon and some oxygen become carbon dioxide (12 + 32 = 44 units of mass), and the hydrogen and the rest of the oxygen become water.
Here’s the super cool part: those four hydrogen atoms (weighing 4 units in total) end up as water. And when they combine with oxygen to form water, the mass of the water produced is actually more than the mass of the hydrogen that went into it! Isn't that wild? It’s like a chemical magic trick where the output weighs more than a part of the input!
Specifically, for every 4 units of hydrogen mass that become water, you get a whopping 36 units of water mass! That means the water produced from the combustion of hydrogen is nine times heavier than the hydrogen itself. It’s like a tiny hydrogen atom eating a whole lot of oxygen and then becoming a much, much bigger water molecule. Incredible, right?

So, if you were to burn, say, one kilogram of pure hydrogen gas completely, you wouldn’t just get a kilogram of something else. You’d get a kilogram of hydrogen PLUS the oxygen it grabbed, resulting in 9 kilograms of water! That’s a massive jump, and it highlights just how much water can be a byproduct of seemingly simple processes.
Let's think about something more common, like wood. Wood is mostly made up of complex molecules containing carbon, hydrogen, and oxygen. When wood burns, the hydrogen atoms within its structure combine with oxygen from the air to form water. And there’s a surprisingly significant amount of it!
For every kilogram of wood that burns completely, you can get around 0.5 to 0.6 kilograms of water! That’s like getting half a liter of water from burning just a small log. Imagine if you could collect all that steam from a bonfire and make a giant water balloon!
And what about gasoline? That’s a whole different beast, a cocktail of hydrocarbons. When a car engine runs, it’s a symphony of controlled explosions, and each one is diligently producing water vapor. Over time, all those tiny puffs of steam add up to a significant amount!
For every kilogram of gasoline that’s completely combusted, you produce roughly 1.2 to 1.3 kilograms of water. That’s more water than the gasoline you started with! It’s like the engine is a secret hydration machine for the planet, albeit an invisible one.
Think about all the cars on the road, all the stoves in our kitchens, all the power plants burning fuel. They are all, in their own way, contributing to the grand cycle of water. It’s a constant, silent production line, turning everyday fuels into the lifeblood of our planet. So next time you see a flame, remember the hidden water being born!
It’s a fascinating thought, isn’t it? The very things that give us warmth, energy, and convenience are also tiny, tireless water factories. They are quietly and efficiently contributing to the world’s water supply, one molecule at a time. So, while you might not see puddles, rest assured, there’s a whole lot of invisible H2O being created! It's a beautiful reminder of the interconnectedness of everything in our amazing universe.
So, to wrap it up, the mass of water produced from the complete combustion of a substance depends on the amount of hydrogen it contains. The more hydrogen, the more water! It’s a simple yet profound principle that showcases the incredible transformations happening all around us, every single day. Prepare to be amazed by the everyday magic of chemistry!
