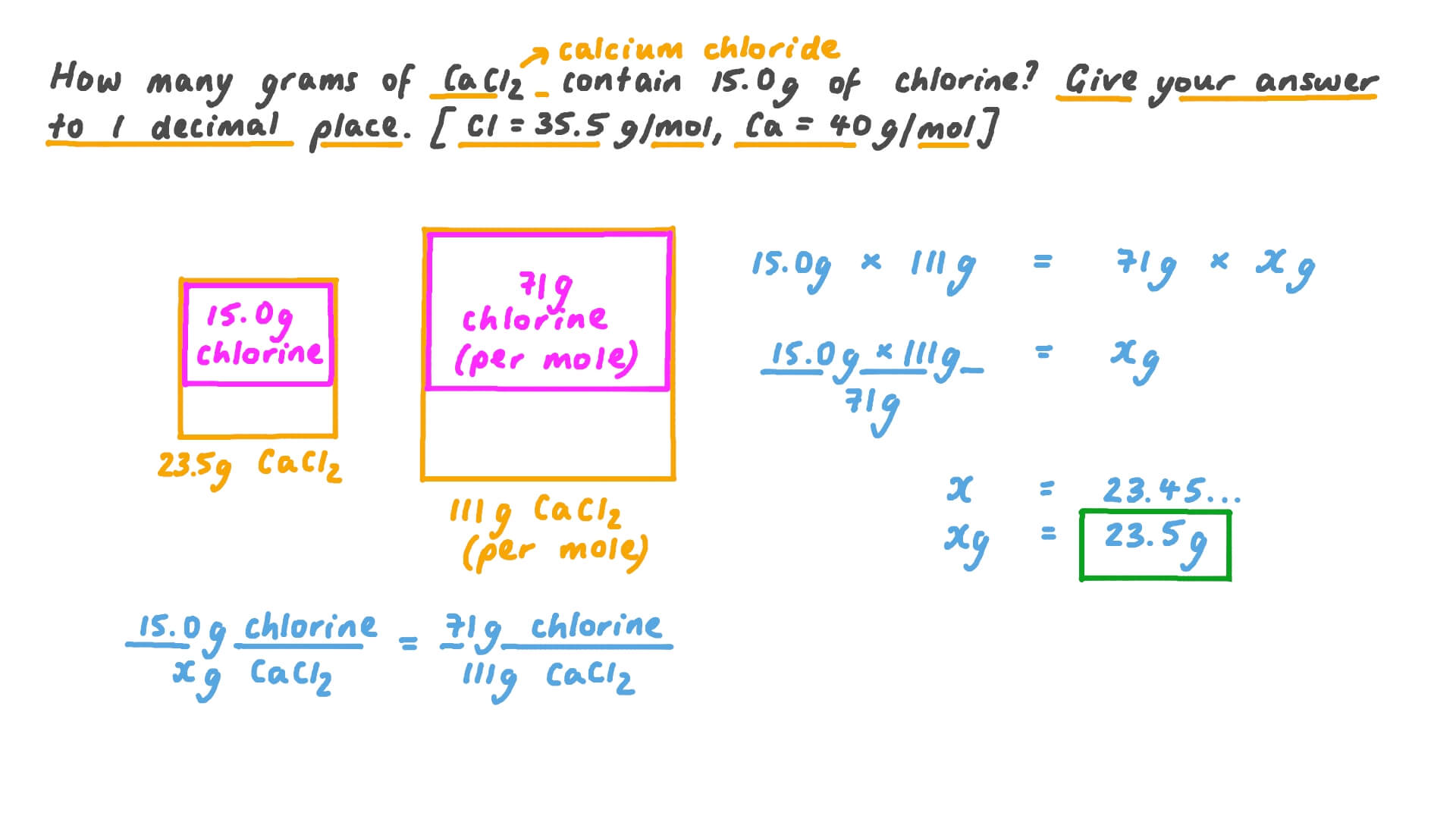
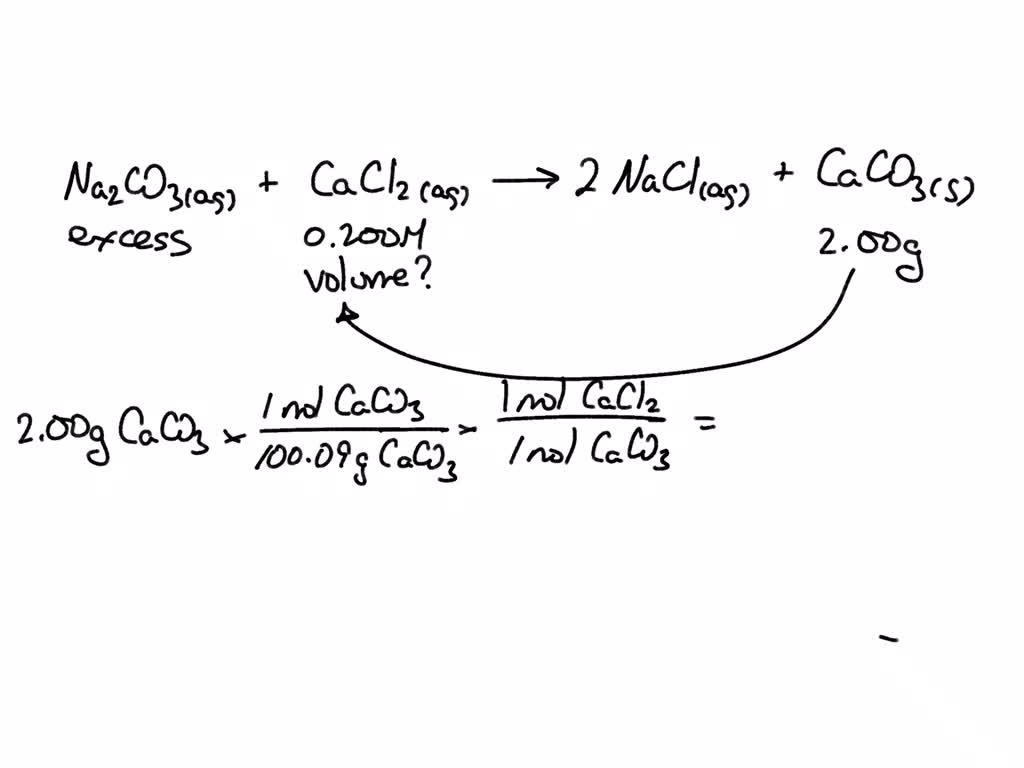
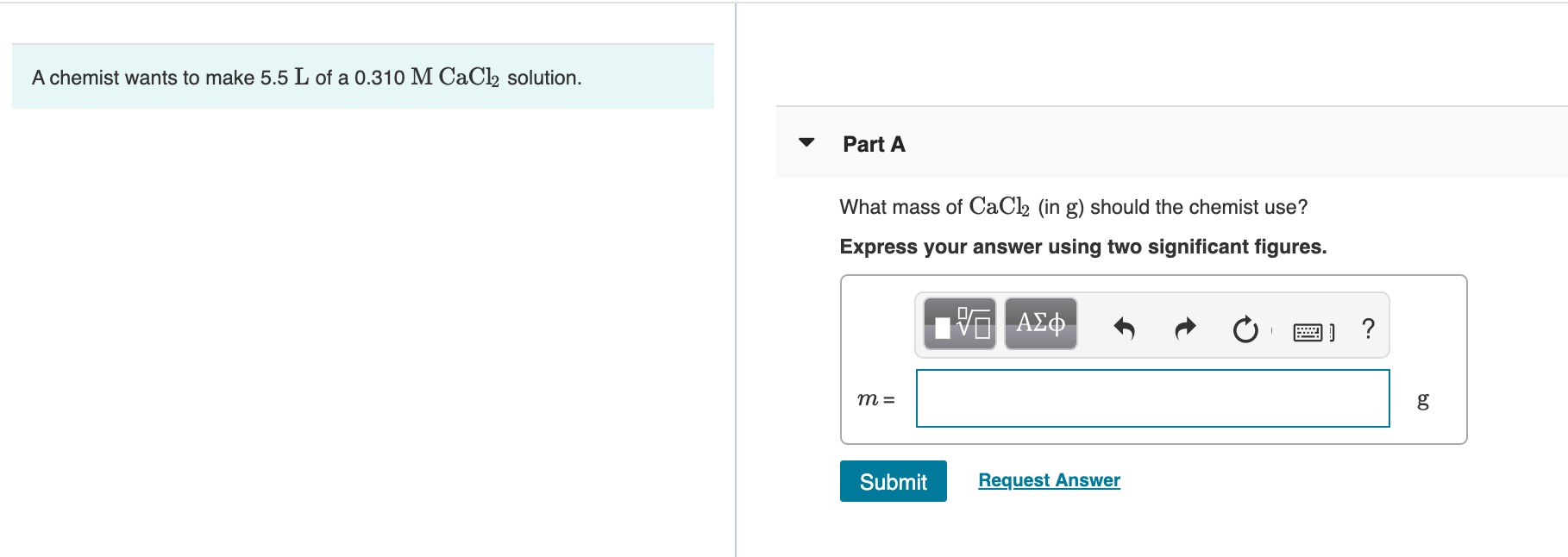
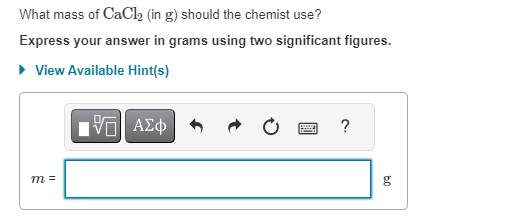
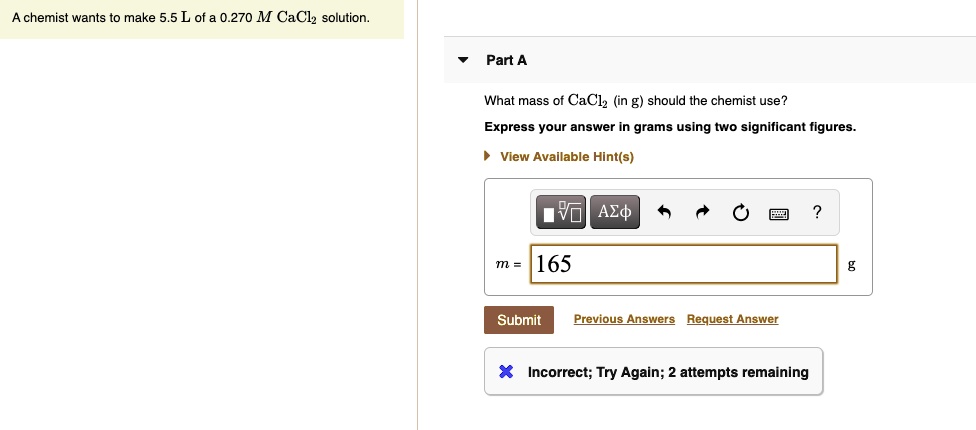
What Mass Of Cacl2 In G Should The Chemist Use
Ah, chemists. Those mystical beings who speak in formulas and can whip up a bubbling concoction with a flick of their wrist. We've all seen them, right? Staring intently at tiny vials, muttering about molarity and stoichiometry like it's the latest celebrity gossip. And then there's the weighing. Oh, the weighing!
Picture this: A chemist, let's call her Dr. Anya, is in her lab. She needs a specific amount of a chemical. Today, it's calcium chloride. Or, as it's more commonly known by its fancy chemical name, CaCl₂. Now, Anya is a whiz. She knows her stuff. But even the best can get a little… creative when it comes to measurements. Especially when it comes to the humble, yet crucial, CaCl₂.
The question on everyone's lips, the one that keeps aspiring chemists up at night and seasoned ones chuckling into their beakers, is this: What mass of CaCl₂, in grams, should the chemist use?
My unpopular opinion? It’s a bit of a guessing game, isn't it? Of course, there are precise calculations. There are perfect, textbook answers. But let's be honest. Sometimes, the lab has its own rhythm. Its own little quirks.
Think about it. Dr. Anya consults her notes. The recipe calls for, let's say, 15.73 grams of CaCl₂. Fifteen point seven… three. That's a lot of pressure. That's a number that demands respect. But what if the tiny scoop is a tad too generous? What if Anya accidentally nudges the scale just as she's aiming for that perfect decimal point?
Suddenly, we're at 15.78 grams. Is that a catastrophe? Will the entire experiment go up in a puff of smoke, or perhaps, more dramatically, a fizzle of disappointment? Probably not. The universe, and by extension, the chemical reaction, is often more forgiving than we give it credit for.
This is where the art of chemistry truly shines, isn't it? It's not just about following the rules; it's about understanding the spirit of the experiment. It's about intuition. It's about that little voice in the back of your head that whispers, "Eh, it's close enough."
Imagine a baker. They don't meticulously weigh every single grain of flour, do they? They have a feel for it. They have a sense of when the dough is just right. Chemists, in their own way, develop a similar instinct. Especially with common reagents like CaCl₂. It's not like handling a volatile explosive; it's a pretty sturdy, if slightly hygroscopic, salt.
So, Anya measures. She hovers. She adds a tiny pinch. She scrapes a minuscule amount off. The digital display flickers. 15.74… 15.72… 15.75. Her brow furrows for a fleeting moment, then a confident smile spreads across her face. She's got it. Or, at least, she thinks she's got it.
"A good chemist knows the theory, but a great chemist knows the practicalities, and sometimes, the practicalities involve a bit of optimistic approximation."
And that's the beauty of it. We're not talking about rocket science here, although sometimes it feels like it. We're talking about making something happen. A reaction. A solution. A discovery. And for that, a few milligrams here or there of CaCl₂ might just be the difference between a successful experiment and a slightly less successful, but still educational, one.
What if the bottle of CaCl₂ itself isn't perfectly pure? What if there's a speck of dust that found its way in? Does Anya get out her miniature vacuum cleaner and meticulously remove it? Probably not. She adds it in, trusts that the impact will be negligible, and moves on. Because that's what chemists do. They adapt. They improvise. They make it work.
So, to answer the burning question: What mass of CaCl₂ should the chemist use? My honest, slightly heretical answer is: Enough to get the job done, with a healthy dose of confidence.
It’s the spirit of adventure, isn't it? The thrill of the unknown. Will it be exactly 15.73 grams? Maybe. Will it be 15.70? Possibly. Will it be 15.81? Hey, it could happen! And in the grand tapestry of scientific endeavor, that little bit of human variability makes things… interesting.
We should celebrate those moments. The tiny sighs of relief when the scale lands close enough. The quiet nods of understanding between chemists when one subtly hands the other a slightly over-measured reagent. It's a fraternity, a sisterhood, a shared understanding that perfection is an ideal, but functionality is a practical necessity.
So next time you see a chemist carefully (or perhaps not-so-carefully) measuring out their CaCl₂, give them a knowing smile. They're not just following a recipe; they're engaging in a delicate dance between precision and practicality. And in that dance, the exact gram of CaCl₂ is sometimes less important than the confident hand that measures it.
Ultimately, the chemist should use the mass of CaCl₂ that leads to the desired outcome. And if that outcome is achieved with a few milligrams more or less than the textbook suggests? Well, that’s just good chemistry in action, wouldn’t you agree?
