What Makes Balancing Redox Reactions Different From Balancing Other Reactions

Hey there, chemistry curious cats and aspiring alchemists! Have you ever looked at a chemical reaction and thought, "Whoa, that looks like a tangled mess of atoms!"? Well, you're not alone. Balancing chemical reactions is like making sure you have exactly the right number of ingredients for a super-secret recipe. You know, so you don't end up with too many flour molecules and not enough sugar for your epic cookie batch. We've all been there, right?
Now, most of the time, balancing these atomic recipes is pretty straightforward. It's like making sure you have two hydrogen atoms and one oxygen atom to whip up some good old H2O – water! You just count up what you have on one side of the reaction arrow (the "reactants," where the magic starts) and then make sure you have the exact same count on the other side (the "products," where the delicious (or sometimes explosive!) results appear). It's all about the conservation of stuff, keeping everything nice and even-steven.
But then, oh boy, then we stumble upon a certain special kind of reaction. These are the ones that make you scratch your head and maybe even do a little jig of confusion. We're talking about the wild and wonderful world of redox reactions! Think of them as the rockstars of the chemical world, a little more dramatic, a little more unpredictable, and definitely a lot more exciting than your average Tuesday night stoichiometry. They've got a secret handshake, a hidden agenda, and a whole different set of rules for getting everything perfectly balanced. It’s like trying to balance a unicycle while juggling flaming torches – possible, but requires a bit more… flair!
So, what's the big deal? What makes these redox reactions such divas of the balancing act? Well, it all boils down to a couple of sneaky little characters that are doing more than just showing up for the party. We're talking about electrons! In regular balancing, we’re just counting atoms. Easy peasy. But in redox reactions, the atoms are not just hanging out; some are losing electrons (they get oxidized – like going on a wild spending spree and losing all their money!) and some are gaining electrons (they get reduced – like finding a treasure chest of electrons and becoming super-rich!).
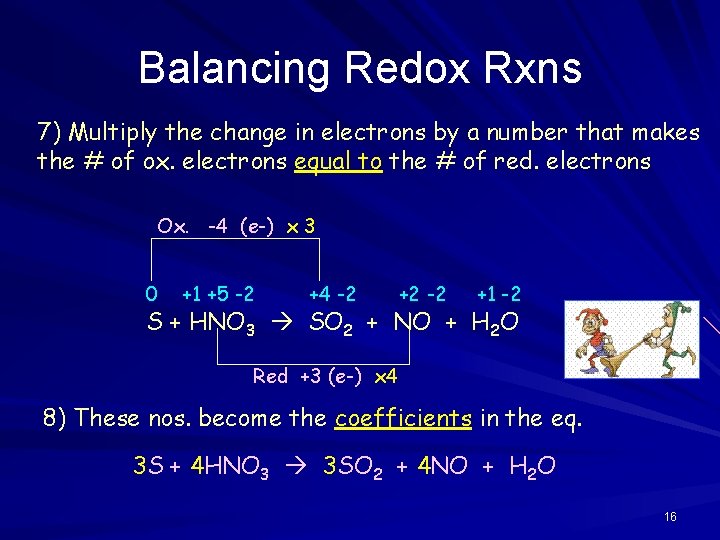
Imagine you're at a party, and you have a bunch of balloons. In a normal reaction, you just need to make sure you have the same number of red balloons and blue balloons on both sides of the room. Simple. But in a redox reaction, it's like some people are handing out balloons (oxidation) and others are eagerly snatching them up (reduction). You not only have to make sure the total number of balloons is the same, but you also have to keep track of who gave away how many balloons and who received them! It's a whole lot more detective work.
This electron-swapping business adds a whole new layer of complexity. It’s not just about the number of atoms anymore; it’s about the electrical charge that these atoms are carrying. Sometimes, the reactions happen in weird places too, like in an acidic solution where there are tons of hydrogen ions running around, or a basic solution where you have hydroxide ions causing mischief. These environments can totally throw a wrench in your balancing plans, making you feel like you’re trying to navigate a maze blindfolded.
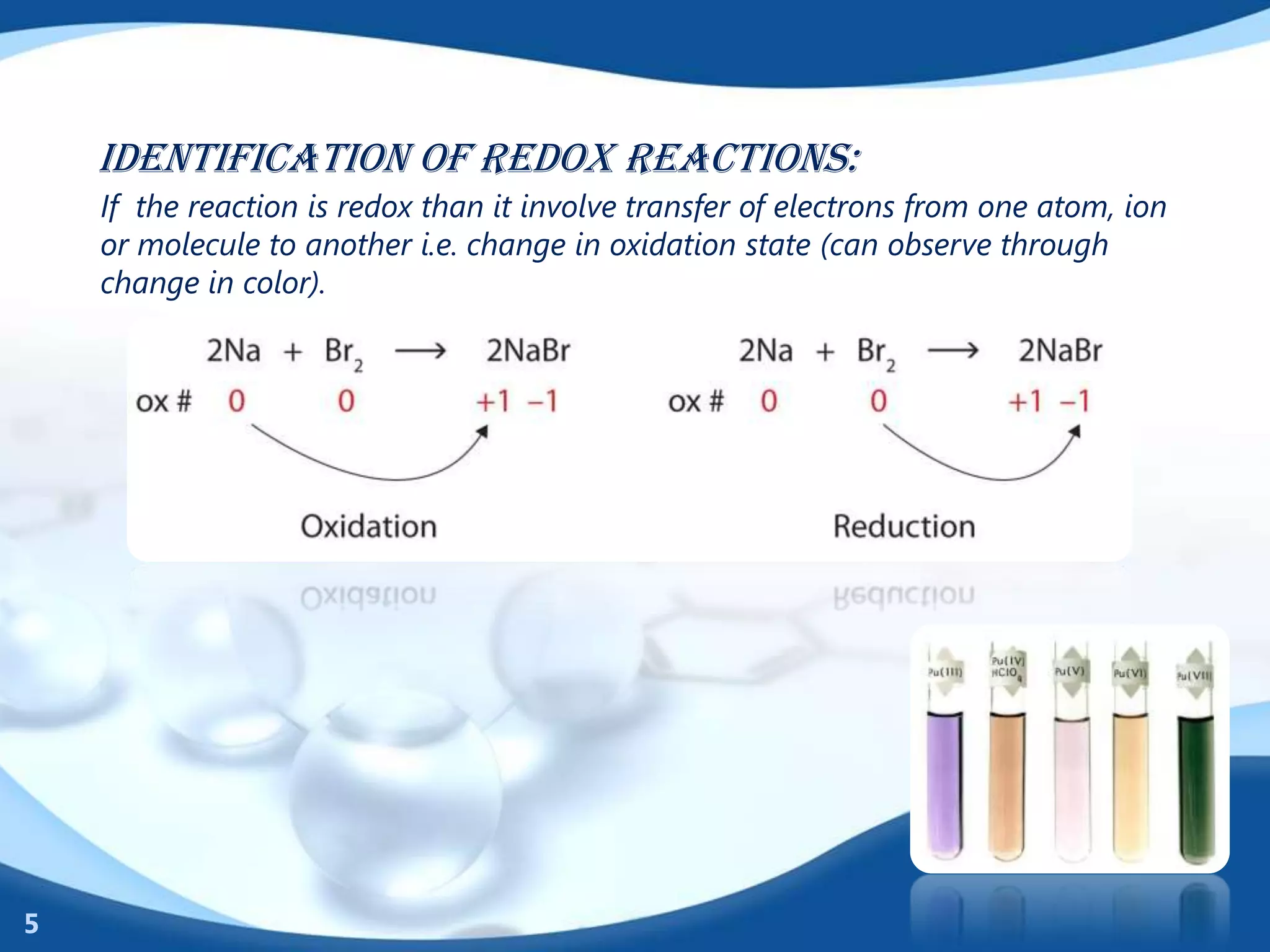
Think of it this way: balancing a regular reaction is like following a recipe for a simple sandwich. You need two slices of bread and one filling. Boom. Done. But balancing a redox reaction is like trying to make a soufflé in zero gravity during a hurricane. You have to consider not only the ingredients but also the atmospheric pressure, the wind speed, and the fact that your eggs might float away! You need special techniques, like the half-reaction method (which sounds a bit like a secret spy mission, doesn’t it?) to break down the big, scary reaction into smaller, more manageable pieces. You balance the electron transfer in one piece, the atom changes in the other, and then you carefully stitch them back together, ensuring that every single electron is accounted for. It's a beautiful, intricate dance of atoms and charge, and when you finally get it right, it feels like you've just solved the world's most satisfying puzzle.
So, while other reactions are content with a simple headcount, redox reactions demand a deeper understanding, a keen eye for electron movement, and a willingness to embrace a little bit of chemical chaos. They're the ones that fuel your car, power your batteries, and even allow you to breathe! They're the unsung heroes, and mastering their balancing act is like unlocking a secret level in the game of chemistry. It’s a challenge, sure, but oh-so-rewarding. Keep experimenting, keep wondering, and don’t be afraid of those electrifying redox reactions – they’re where the real fun happens!
