What Is The Strongest Acid Among The Following
Imagine a superhero showdown, but instead of capes and laser eyes, we've got tiny chemical warriors battling it out. Today, we're peeking into the wild world of acids, those zesty substances that can add tang to your lemonade or make things… well, a bit too zingy.
We're going to look at a lineup of acids, not to give them all a full-blown origin story (that would take ages!), but to see who stands tall as the undisputed champion of "ouch, that's acidic!" Think of it as a friendly competition, where the prize is bragging rights for being the most… acidic. It's less about a dramatic fight and more about a friendly, slightly fizzy, comparison.
First up, let's give a little wave to Hydrochloric Acid. You might have heard of it before. It’s the stuff that helps your tummy digest that delicious burger you had for lunch. Pretty important, right? It’s like the trusty sidekick of your digestive system, always ready to get to work.
But don't let its helpfulness fool you. When concentrated, Hydrochloric Acid can be a real powerhouse. It's used in everything from cleaning rust off metal to making your favorite candies. So, it's a bit of a multitasker, this one.
Next on our imaginary stage is Sulfuric Acid. Now, this one is a bit of a reputation-builder. It’s sometimes called "oil of vitriol" because of its oily texture. Sounds a bit mysterious, doesn't it? It’s a workhorse in industries, making fertilizers, dyes, and even batteries for your car.
Sulfuric Acid is known for being a bit of a dehydrator, meaning it likes to steal water from things. It can turn sugar into a black, bubbly mess. Kind of like a mischievous wizard who can turn anything into coal, but with water as its target.
Then we have Nitric Acid. This one has a rather dramatic name, doesn't it? It's a strong oxidizer, which means it loves to give up its oxygen atoms in chemical reactions. Think of it as a generous chemist, always sharing its oxygen for the greater (or sometimes less great) good.

Nitric Acid is involved in making explosives, which gives it a bit of a "bad boy" image. But it’s also crucial for making fertilizers, so it’s not all boom and no bloom. It’s a complex character, this one, with a diverse resume.
Now, let’s introduce our dark horse, or should we say, our super-powered chemist. We’re going to talk about Superacids. These aren’t your everyday kitchen cupboard ingredients. They’re in a league of their own, far beyond the acids we’ve met so far.
Think of it this way: if Hydrochloric Acid is a strong gust of wind, and Sulfuric Acid is a powerful hurricane, then a Superacid is… well, it's more like a cosmic storm that rearranges planets. They are so incredibly strong that they can do things that seem almost magical.
One of the most famous examples of a superacid is Fluoroantimonic Acid. Say that ten times fast! This is the undisputed king of the acidic jungle, the grand poobah of protons. It’s a concoction that scientists brew up when they really need to make something happen.

Fluoroantimonic Acid is formed by mixing hydrogen fluoride and antimony pentafluoride. Don't worry too much about the names; just know that when these two get together, they create something truly extraordinary. It's like the ultimate power couple of the chemical world.
What makes Fluoroantimonic Acid so special? Well, it’s its incredible ability to attract and hold onto protons. Protons are like the tiny, positively charged building blocks of atoms. Acids are all about donating these protons.
The stronger the acid, the more eager it is to give away its protons. And Fluoroantimonic Acid? It's practically throwing them around like confetti at a party! It’s so good at it that it can even make some very stable molecules act like they’re made of something else entirely.
Imagine trying to dissolve a diamond. Most acids wouldn't even bat an eyelash. But Fluoroantimonic Acid? It can actually do it! It can break down some of the toughest substances known to science. It's like the ultimate dissolver, capable of tackling things that would make other acids weep.
Scientists use these superacids for very specific, high-tech applications. They are essential for creating new materials, understanding complex chemical reactions, and pushing the boundaries of what we thought was possible. They are the tools of discovery for the most adventurous chemists.
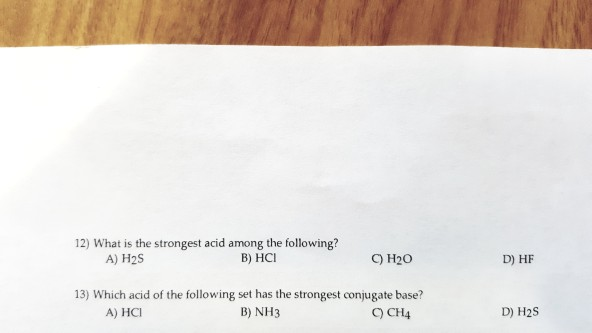
So, when we ask what’s the strongest acid among the ones we’ve discussed, the answer is clear: Fluoroantimonic Acid reigns supreme. It’s not just strong; it’s in a category all its own. It’s the superhero that makes the other heroes look like they’re just starting their training.
It’s easy to think of acids as just dangerous chemicals, but they are also the unsung heroes of so many things we take for granted. From helping us digest our food to enabling the creation of advanced materials, acids play a vital role. They are the tiny engines that drive many important processes.
And understanding the extremes, like the incredible power of Fluoroantimonic Acid, gives us a new appreciation for the subtle but profound forces at play in the world around us. It’s a reminder that even the smallest things can have the biggest impact.
So next time you enjoy a tart lemon or marvel at a new piece of technology, remember the world of acids, and give a little nod to the extraordinary strength and versatility they possess. They are truly remarkable, in their own special, sometimes sparkly, way.

It’s like a secret handshake for molecules, and Fluoroantimonic Acid knows all the best moves!
The sheer power of these substances can be both awe-inspiring and a little bit humbling. They remind us that nature has its own set of incredible forces, waiting to be understood and harnessed.
Think about how much chemistry is happening all around us, all the time. Acids are a big part of that unseen world, quietly doing their work. They are the silent architects of many marvels.
The journey from a simple stomach acid to a superacid like Fluoroantimonic Acid is a testament to human curiosity and ingenuity. We’ve taken something fundamental and pushed its capabilities to the absolute limit.
It’s a story of chemistry, yes, but it’s also a story of exploration. It's about asking "what if?" and then finding out just how powerful an answer can be. The world of acids is full of surprises!
