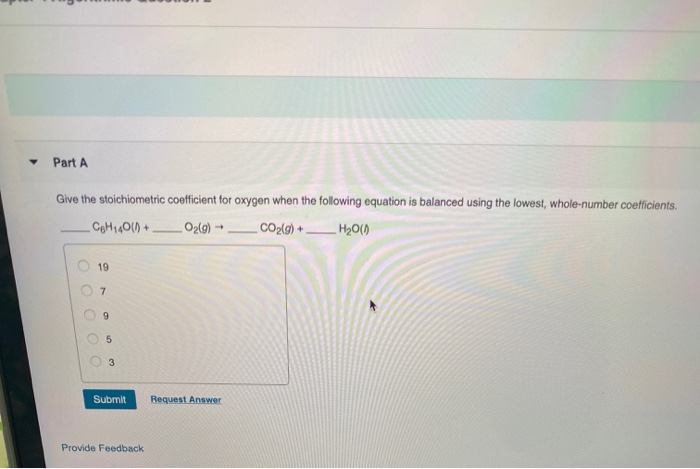
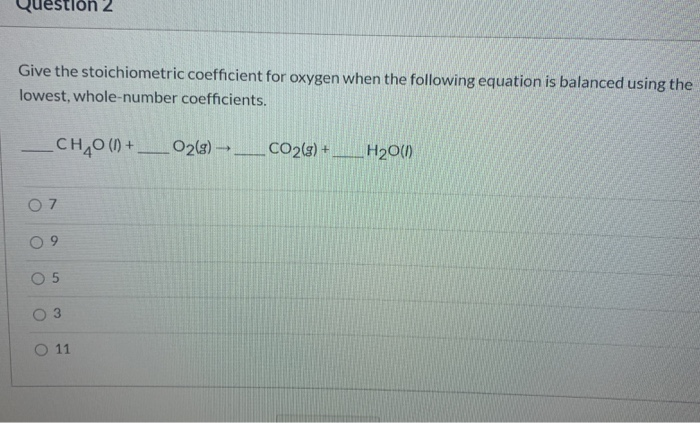
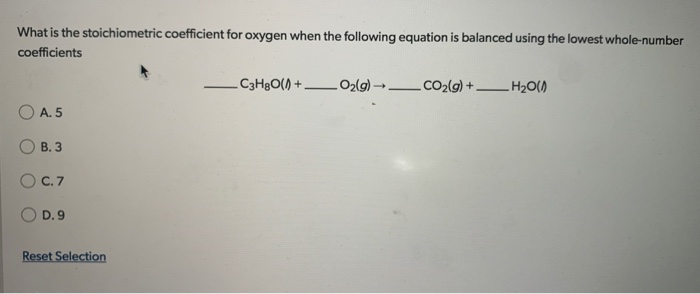
What Is The Stoichiometric Coefficient For Oxygen

You know that feeling when you're baking, and the recipe calls for exactly two cups of flour? It's a delicate balance, right? Too much, and your cookies turn into little hockey pucks. Too little, and they might just crumble into dust. Well, the world of chemistry has its own version of this precise baking, and it involves something we all need to live: oxygen!
When we talk about the "stoichiometric coefficient for oxygen," we're essentially talking about its perfect amount in a chemical recipe. Think of it like the magic number that makes a chemical reaction just right. It’s not just some abstract number; it’s the key to unlocking energy, creating new materials, and even keeping our planet breathing.
Let’s zoom in on something super familiar: burning. When you light a candle, or when a car engine roars to life, you're witnessing a chemical reaction where oxygen plays a starring role. It's like the enthusiastic partner that helps everything else get going. Without enough oxygen, the flame sputters, and the engine coughs. Too much, and well, that’s a whole different, usually much bigger, story!
The stoichiometric coefficient tells us the exact amount of oxygen needed for that particular chemical dance. It’s the silent orchestrator of combustion, making sure that fuel and oxygen meet in the perfect ratio to produce the desired outcome. Imagine a grand ballroom dance, and the coefficient is the choreographer, ensuring everyone steps in time.
Take the simple act of burning wood. For every bit of wood that wants to turn into ash and warmth, there's a specific, beautiful relationship with oxygen molecules. It’s a precise partnership, like a perfectly matched pair of dancers on the floor.
This concept isn't just for science geeks in labs. It's at play every time you take a breath! Your body is a marvelous chemical factory, and oxygen is the fuel that powers it all. The stoichiometric coefficient for oxygen in our own bodies ensures we get just enough to keep our cells humming, without overdoing it.

Think about when you exercise. Your body needs more energy, right? So, you breathe deeper and faster, bringing in more oxygen. This isn't random; it's your body's clever way of adjusting the "stoichiometric coefficient" of oxygen it needs for that heightened activity. It’s like turning up the dial just enough to meet the demand.
It’s also pretty heartwarming when you think about it. This precise relationship allows for the release of energy that sustains life. Every beat of your heart, every thought you have, is powered by this careful chemical ballet where oxygen's coefficient is a crucial step.
And it's not always about fire. Consider how plants make food. They take in carbon dioxide and sunlight, and then, through photosynthesis, they release oxygen! This is another amazing chemical reaction where oxygen’s coefficient is critical. It’s the byproduct of their healthy living, which, in turn, is essential for ours.
So, when plants are busy making the air we breathe, the stoichiometric coefficient for oxygen in that process is like a guarantee. It ensures that the right amount of this life-giving gas is produced, creating the atmospheric balance that makes Earth so special. It’s a cosmic exchange, a give-and-take that keeps everything in harmony.
Sometimes, these coefficients can be a bit surprising. They might seem like arbitrary numbers, but they are derived from the fundamental way atoms bond together. It's like discovering the secret handshake of molecules.
Let’s take another look at burning fuel, like in a car. The engine is designed to mix fuel with a specific amount of air (which is mostly nitrogen, but also contains our star, oxygen). The stoichiometric coefficient for oxygen here is key to making sure the combustion is efficient, leading to less waste and more power.
If the car engine had too little oxygen, it would run poorly, like trying to run a race with a heavy backpack. If it had too much, it might overheat or waste fuel. The coefficient is the sweet spot, the perfect balance for optimal performance. It's like a finely tuned instrument.
And think about the incredible feat of space travel! Rockets burn massive amounts of fuel, and oxygen is a crucial component. The precise control of oxygen's coefficient is vital for generating the immense thrust needed to escape Earth's gravity. It's a fiery dance on a colossal scale!
Scientists spend a lot of time figuring out these coefficients. It's like solving a giant, intricate puzzle to understand how the universe works at its smallest level. And for us, it helps us build better engines, understand our own bodies, and even create new medicines.
It’s also a reminder of how interconnected everything is. The oxygen we breathe might have been released by a tree miles away, or it might be the result of a carefully controlled industrial process. The stoichiometric coefficient is the invisible thread connecting these events.
Consider something as simple as a gas stove. When you turn it on, natural gas mixes with oxygen from the air. The stoichiometric coefficient ensures a clean, efficient flame that cooks your dinner. It’s a little bit of chemical magic happening right in your kitchen.
Even when we talk about pollution, the stoichiometric coefficient plays a role. Sometimes, incomplete combustion happens because the oxygen isn't in the perfect ratio. This can lead to harmful byproducts. Understanding these coefficients helps us design cleaner technologies.
It’s funny to think that something as simple as a number can have such a profound impact on our lives. The stoichiometric coefficient for oxygen isn't just a number; it's a promise of energy, a facilitator of life, and a cornerstone of the natural world.
So, the next time you see a flame, or take a deep breath, remember the quiet, precise work of the stoichiometric coefficient for oxygen. It's a little scientific secret that makes our world, and our lives, possible. It’s the unsung hero of countless reactions, ensuring everything happens just right.
It’s like the best supporting actor in a play, always there, always doing its job perfectly, without needing a spotlight. And that, in its own way, is pretty amazing. It’s a testament to the beautiful, intricate order of the universe.
So, while you might not be calculating these coefficients yourself (unless you're a chemist, of course!), knowing they exist adds a layer of wonder to the everyday. It’s a reminder that even the most common things, like the air we breathe, are governed by fascinating rules.
