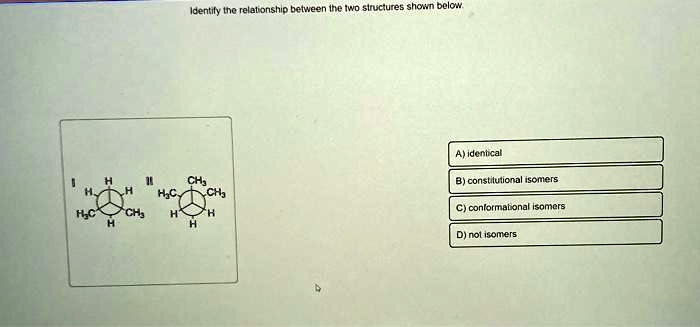
What Is The Relationship Between The Structures Shown Below
Ever looked at something and thought, "Wow, that's... similar!"? It's like spotting your favorite celebrity on the street and doing a double-take because they look exactly like your next-door neighbor. Well, prepare yourselves, because we're about to dive into a world of mind-blowing connections between things you might see every day, but never realized were practically kissing cousins!
Imagine you're baking a cake. You've got your flour, your sugar, your eggs – all separate ingredients. But when you mix them all up with a little love and heat, BAM! You get a fluffy, delicious masterpiece. These structures we're looking at? They're kind of like those ingredients, but instead of a cake, they bake up something way cooler: the very building blocks of life and the magnificent stuff that makes our world go 'round!
It's All About the Connections, Baby!
Think about it. What do you have when you have a whole bunch of tiny little things that are basically identical, just repeating themselves over and over and over? That, my friends, is the magic of polymers! You see them everywhere, whether you realize it or not. That comfy sweater you're wearing? Likely made of a polymer. The plastic bottle you're sipping your water from? Yep, a polymer. Even the DNA inside your cells, the instruction manual for you, is a super-duper special kind of polymer called a nucleic acid.
Now, let's get specific. When you see something like a long chain of identical little units linked together, like a train with hundreds, even thousands, of identical carriages, you're looking at a monomer that has decided to get very social and form a polymer. These monomers are the individual Lego bricks, and the polymer is the epic castle you build with them. It's all about these repeating units, these tiny building blocks, joining hands and creating something much bigger and much more awesome.
Consider the humble glucose molecule. This is a sugar, a simple little guy. But when a gazillion of these glucose molecules decide to have a massive party and link up end-to-end, what do they form? They can form starch, which is how plants store energy (think potatoes!). Or, if they link up in a slightly different, more organized way, they can form cellulose, which is what gives plants their structure (think trees!). Same basic ingredient, wildly different jobs. It's like having the same set of crayons but drawing a fierce dragon one day and a fluffy cloud the next. Mind. Blown.
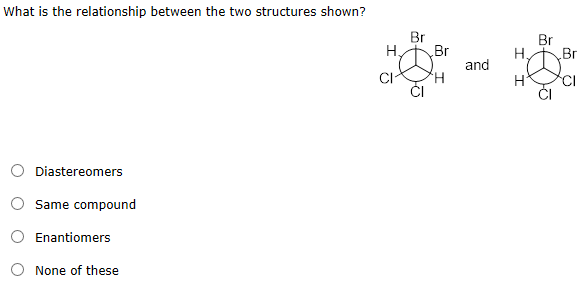
The DNA Connection: It's Not Just a Story in Your Genes!
And then there's the superstar of the show: DNA! You might think of DNA as just the stuff that makes you tall or gives you curly hair. But at its core, DNA is a magnificent polymer. It's a long, twisted ladder made up of repeating units called nucleotides. These nucleotides are the four letters of life's alphabet (A, T, C, and G), and the specific order of these letters tells your body how to build everything. It’s like a secret code, and the polymer structure is what holds that code in a stable, readable format.
Imagine trying to write a novel using only loose letters scattered on the floor. It's a mess! But when you put those letters into words, then sentences, then paragraphs, you get a story. DNA is like that story, and the nucleotides are the letters, all neatly arranged in a long, beautiful polymer chain.
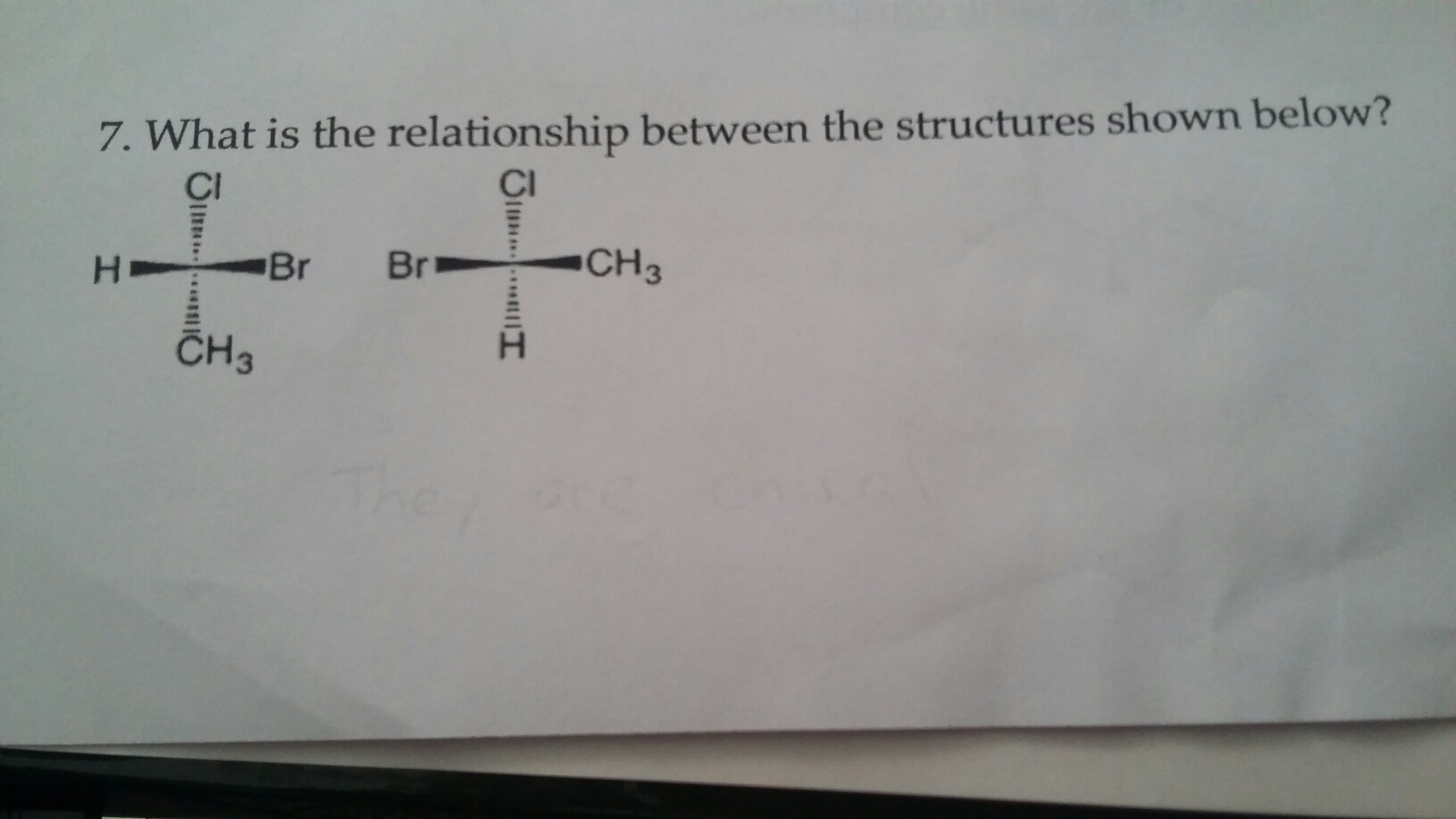
Identify the relationship between the two structures shown below. CH3
So, what's the relationship between a simple sugar like glucose, a structural component like cellulose, and the complex blueprint of life that is DNA? It’s the fundamental principle of polymerization! It's the idea that small, repeating units can join together to form much larger, more complex structures with entirely new properties. It’s nature’s way of building big things from small, simple parts. It’s efficient, it’s elegant, and it’s happening all around us, all the time.
Think about it like this: you wouldn't build a house with just one giant brick, right? You'd use lots and lots of smaller bricks. These monomers are those smaller bricks. When they come together, they form something incredible. It’s the same underlying idea, whether you're talking about the energy storage in a potato, the strength of a redwood tree, or the intricate instructions within your own body.
So next time you're munching on a piece of fruit (full of sugars that might be polymerized!), or marveling at the strength of a spider's web (made of protein polymers!), or even just looking in the mirror and thinking about your unique awesomeness (thanks, DNA!), remember this incredible connection. It’s the power of the repeat, the beauty of the chain, the fundamental relationship that makes life, well, life!