What Is The Relationship Between Metallic Character And Ionization Energy

Hey there, curious minds! Ever wonder why some stuff is super keen to hold onto its electrons, while other stuff just can't wait to let them go? It’s like a cosmic dance of atoms, and it all boils down to a really cool relationship between metallic character and ionization energy.
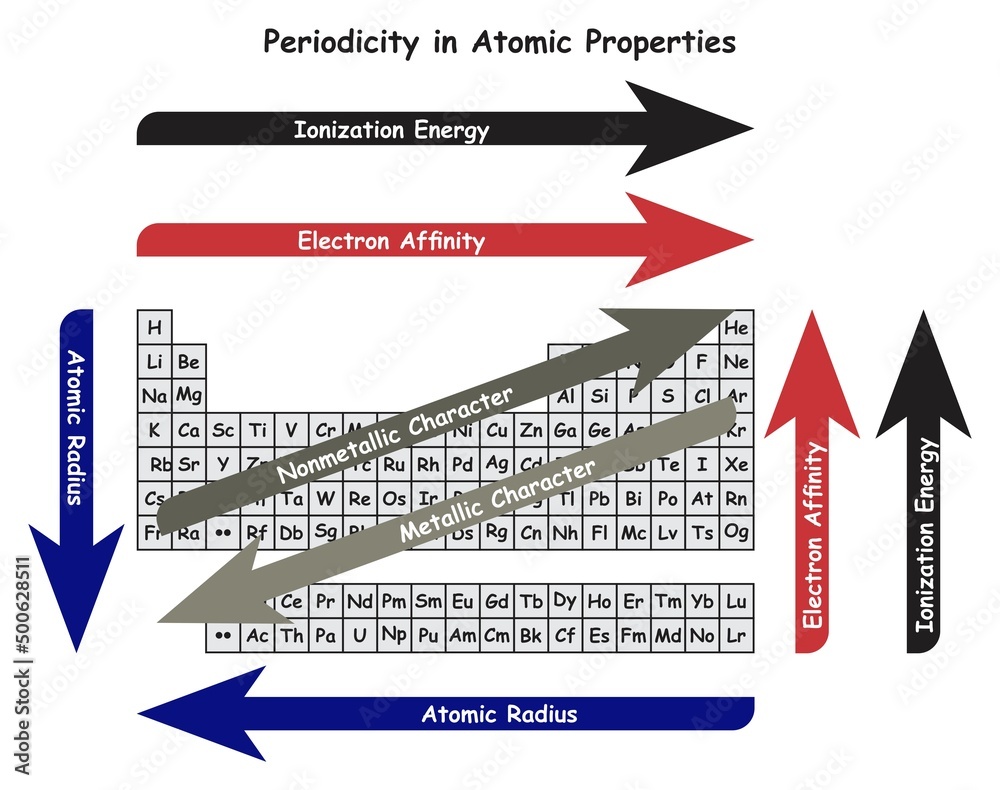
Think of metallic character as how much an element acts like a typical metal. You know, shiny, good at conducting electricity, and generally a bit… well, metallic! These guys love to lose electrons to become positively charged ions. It’s their happy place, where they feel complete and stable.
On the flip side, we have ionization energy. This is the energy it takes to actually pry one of those precious electrons away from an atom. Imagine it as the "stickiness factor" of an electron. High ionization energy means that electron is really, really stuck. Low ionization energy means it's practically begging to be set free!
So, what’s the connection? It’s actually super straightforward, like peas in a pod! Elements with high metallic character tend to have low ionization energy. They’re the generous types, always ready to share their electrons.
Why is this so neat? Because it helps us understand why different elements behave so differently! It’s like a secret code that tells us about their personality. Metals, with their low ionization energy, are the extroverts of the periodic table. They’re quick to bond and form new compounds, often by donating those electrons.
Imagine sodium, that friendly metal in salt. It has a super low ionization energy. It’s practically throwing its electron at chlorine to become the happy sodium ion (Na+) we see in table salt. It’s all about that electron liberation!
On the other hand, elements with low metallic character (think your nonmetals, like oxygen or fluorine) have high ionization energy. They cling to their electrons like a toddler to their favorite toy. They don’t want to give them up easily, if at all!
Fluorine, the most electronegative element out there, is a prime example. It has a ridiculously high ionization energy. It would rather hug its electron tightly than let it go. This is why fluorine is so reactive, always looking to take electrons from other atoms rather than give them away.
This relationship is fundamental to chemistry. It’s the engine that drives so many reactions we see every day. From the way batteries work to how plants photosynthesize, it’s all influenced by this electron-sharing dance.
Think about it: if metals had high ionization energies, they wouldn’t be such great conductors of electricity, right? The free-moving electrons are what make them so useful in our gadgets and wires. And that’s all thanks to their willingness to let go of those electrons.
The periodic table itself is a beautiful map of this relationship. As you move across a period from left to right, metallic character generally decreases, and ionization energy generally increases. It’s a consistent trend that chemists have relied on for ages!

Metals on the far left, like lithium and potassium, are super metallic and have very low ionization energies. They are practically begging for their electrons to be taken. They are the ultimate electron donors!
As you move towards the right, you encounter less metallic elements. Their atoms hold onto their electrons a bit tighter. Think of elements like silicon, which is a metalloid – it’s got some metallic properties, and its ionization energy is in the middle range.
Then you hit the nonmetals on the far right. These guys are the electron hoarders! They have high ionization energies and are not very metallic at all. They’d rather steal an electron than give one up.
And what about those noble gases, like helium and neon? They are the ultimate aloof characters of the periodic table. They have full outer electron shells, which makes them incredibly stable. Their ionization energies are through the roof! They have virtually no metallic character because they have zero interest in losing electrons.

This is what makes chemistry so fascinating – these predictable patterns! It’s like solving a puzzle where each element has its own unique set of rules. Understanding metallic character and ionization energy is like getting the cheat codes to that puzzle.
It’s not just about abstract science, either. This knowledge helps us develop new materials, design better medicines, and even understand the composition of distant stars. The universe is built on these fundamental interactions!
So, the next time you see a shiny piece of metal or a bubbling chemical reaction, remember the unseen ballet of electrons. Remember the generous metals happily parting with their electrons and the stubborn nonmetals guarding theirs fiercely.
The relationship between metallic character and ionization energy is a cornerstone of chemical understanding. It’s a beautiful illustration of how the structure of atoms dictates their behavior and interactions.
/PeriodicTable-Trends-56a1310e5f9b58b7d0bcea8a.png)
It’s the difference between something that readily forms a positive ion and something that fiercely resists losing an electron. It’s the reason why sodium (a metal with low ionization energy) forms NaCl (salt) so readily with chlorine (a nonmetal with high ionization energy).
And that’s the magic of it! It’s a concept that’s both incredibly powerful and surprisingly easy to grasp once you see the connection. It’s a little bit like learning a secret handshake for the elements.
So, if you’re looking for a little scientific wonder, dive into the world of periodic trends. You’ll find that the relationship between metallic character and ionization energy is a truly captivating part of the story of matter. It’s a reminder that even the smallest things, like electrons, play a massive role in shaping our world!
Isn’t that just the coolest? It’s a simple concept, but its implications are vast. It’s like finding out that your favorite superhero’s strength comes from a very specific, yet elegant, source.
Next time you’re pondering the elements, remember this dynamic duo. Their interplay is a fundamental aspect of chemistry that’s both fun to learn and incredibly important to understand. It’s a true testament to the order and beauty found in the atomic world. Happy exploring!
