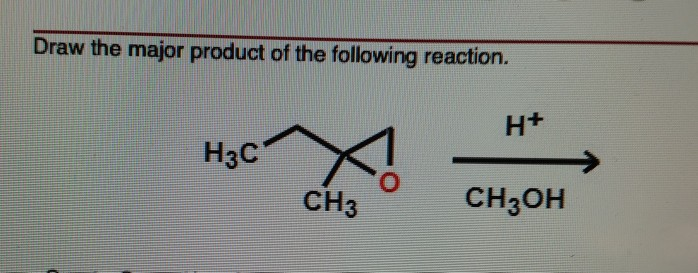
What Is The Product Of The Following Reaction Ch3oh H+
Ever wondered what happens when a simple alcohol like methanol meets a tiny spark of acidity? It’s a question that might sound a bit niche, but delving into it can be surprisingly fun and offers a peek into how many of the things we use every day are made. Think of it like a tiny chemical puzzle, and figuring out the answer unlocks a bit of the magic behind some familiar substances.
The reaction we’re talking about is the acid-catalyzed reaction of methanol (CH₃OH) with itself, often represented in a simplified way as CH₃OH + H⁺. The H⁺ here represents an acid catalyst – it's not actually consumed in the reaction but helps it along. The primary goal of this kind of reaction is to create ethers. In this specific case, with methanol, the main product we get is dimethyl ether (CH₃OCH₃).
Why is this interesting? Well, dimethyl ether is a fascinating molecule with some important applications. It’s a gas at room temperature and pressure, making it useful as an aerosol propellant, much like the stuff that sprays your hairspray or air freshener. Beyond that, it’s being explored as a clean-burning fuel, either on its own or as a blend with other fuels. It’s also a key intermediate in the production of other chemicals, like formaldehyde, which is a building block for plastics and resins. So, understanding this simple reaction is like understanding a fundamental step in creating a whole range of useful materials and energy sources.
In education, this reaction is a classic example used to teach about nucleophilic substitution reactions and the role of acid catalysts in organic chemistry. Students learn how functional groups can be modified and how new bonds are formed. In daily life, while you won't directly perform this reaction in your kitchen, you indirectly benefit from its products. As mentioned, aerosol cans use propellants derived from similar chemistry. The plastics in your phone case, your car dashboard, or even some furniture often trace their origins back to chemicals produced using etherification reactions like this one. It’s a subtle but pervasive part of modern life.
Exploring this concept further doesn't require a full laboratory. You can start by looking up the chemical structures of methanol, dimethyl ether, and the concept of acid catalysts online. Websites like Wikipedia, educational chemistry sites, or even YouTube channels dedicated to explaining science concepts are great resources. You can visually see how the molecules change. Another simple way to explore is by looking at the ingredients list on aerosol products and researching what those propellants are and how they’re made. It’s a journey of curiosity that can lead you to appreciate the intricate world of chemistry that underpins so much of our world. So next time you use an aerosol can, remember the simple magic of methanol and its acidic spark!
