What Is The Predominant Intermolecular Force In Cbr4

Have you ever found yourself gazing at a stunning piece of art, marveling at the intricate details and vibrant colors, and wondered, "How did they do that?" Often, the secret lies not just in talent, but in mastering specific techniques and understanding the fundamental building blocks of creative expression. Today, we’re diving into a topic that might sound a tad scientific but holds immense creative potential, especially for those who love to tinker and explore: the predominant intermolecular force in CBr4.
Now, before your eyes glaze over, let’s reframe this. Think of understanding intermolecular forces as a secret ingredient for artists and hobbyists. For anyone delving into materials science, chemistry, or even just curious about the physical world, grasping these forces can unlock a deeper appreciation for how substances behave. For those who enjoy DIY projects, particularly those involving the creation or manipulation of materials, knowing the forces at play can help predict outcomes and troubleshoot issues. It’s like knowing how different paints will mix or how clay will dry – a foundational knowledge that enhances your creative control.
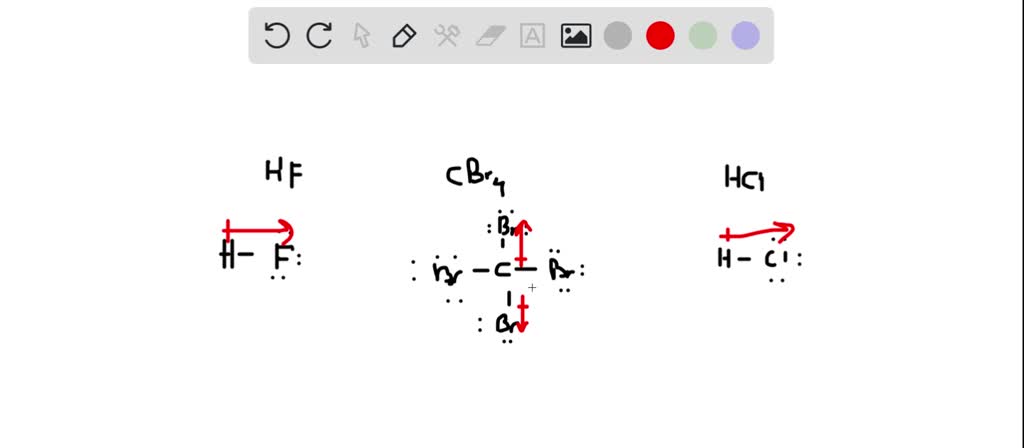
So, what exactly is this predominant force in CBr4 (carbon tetrabromide)? CBr4 is a molecule where carbon is bonded to four bromine atoms. Because the molecule is symmetrical, the individual bond polarities cancel each other out, making the overall molecule nonpolar. The predominant intermolecular force, therefore, is the London dispersion force, also known as van der Waals forces. These are weak, temporary attractive forces that arise from the random movement of electrons within molecules. While weak individually, they become significant when you have many molecules interacting.
How can this knowledge be creatively applied? Imagine artists exploring different types of inks or solvents for their work. Understanding the polarity of a solvent and the substances it needs to dissolve can prevent clumping or ensure smooth application. For hobbyists building models or working with resins, knowing how different components will interact at a molecular level can lead to stronger bonds and more durable creations. Even for something as simple as choosing a cleaning agent for a delicate surface, this knowledge can be invaluable.
For those eager to explore this at home, you don’t need a laboratory! Start by observing everyday substances. Notice how oil and water don’t mix – that’s due to differences in their intermolecular forces. You can experiment with different solvents and solutes (safely, of course, with adult supervision if needed). Look up the chemical structures of common household substances and try to predict their interactions based on whether they are polar or nonpolar. Think about how sticky notes work (adhesion, another intermolecular force) or why certain fabrics repel water.
The enjoyment comes from seeing the world around you with new eyes. It’s about the thrill of discovery, the satisfaction of solving a puzzle, and the added layer of understanding that enriches any creative pursuit. So, the next time you’re looking at a material, remember that behind its appearance and texture lies a fascinating dance of intermolecular forces, waiting to be understood and, perhaps, even creatively harnessed!
