What Is The Oxidizing Agent In The Following Reaction
Alright, let's dive into the wild and wonderful world of chemistry. Don't worry, we're not talking about anything too intense here. Think of it more like a friendly chat with a beaker, not a pop quiz from Professor Grumpy Pants. Today's topic? The star of the show, the MVP, the one and only... Oxidizing Agent!
Now, I know what you're thinking. "Oxidizing Agent? Is that some kind of fancy space dust or maybe a new energy drink?" Nope! It's much cooler, and frankly, a lot less likely to give you the jitters. Imagine a chemical reaction as a bit of a drama. There are characters, there's action, and there's always someone who's a little more... demanding. That's where our Oxidizing Agent struts onto the stage.
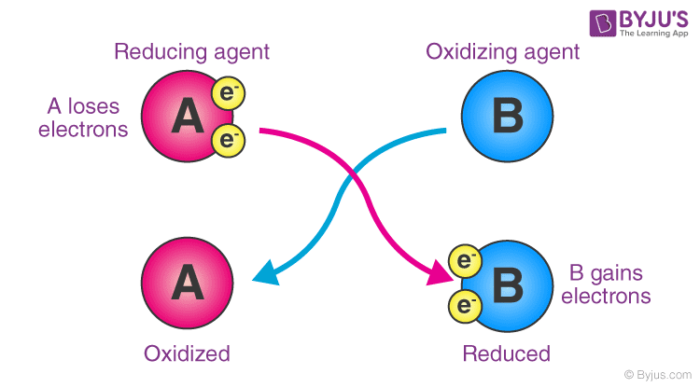
So, what exactly is this illustrious character? Think of it as the ultimate electron-taker. It strolls into a reaction, spots some poor, unsuspecting molecule with a spare electron, and goes, "Hey there, pal! Mind if I borrow that? Permanently?" It's the ultimate opportunist, the cosmic collector of electrons. And it does this by accepting them. It's like the person at a party who always ends up with everyone's spare change.
This act of electron-snatching is what we call oxidation. And our Oxidizing Agent? It's the one causing that oxidation to happen. It's the puppet master, the ringleader, the one pulling the strings. It's so good at its job, it practically forces other things to give up their electrons. It's a bit bossy, I'll admit, but hey, someone's gotta keep things moving, right?
Now, this is where things get a tiny bit juicy, and perhaps where my unpopular opinion comes in. While the Oxidizing Agent is busy being all electron-greedy, it's actually doing itself a favor. It's like when you go to a buffet and load up your plate. You're taking stuff, sure, but you're also getting fed. In chemistry terms, when the Oxidizing Agent takes electrons, it's actually getting reduced. Mind blown, right? It's the ultimate act of self-care disguised as a chemical heist.

It's the ultimate act of self-care disguised as a chemical heist.
So, while it's out there making other molecules lose electrons (oxidizing them, remember?), it's simultaneously getting electrons itself (getting reduced). It's a win-win, really. It's like that friend who always convinces you to try a new, slightly scary activity, and then ends up having the time of their life while you're just clinging on for dear life. But hey, at least they're having fun, and in this case, the entire reaction moves forward because of it.
Think about it in everyday terms. Ever had that friend who's just brimming with energy? They're always pulling you into their latest project, their grand adventure. They're the spark. They're the catalyst. In a way, they're acting a bit like an Oxidizing Agent in your social life. They're taking your "couch potato" electrons and oxidizing you into action! And in doing so, they're probably getting a kick out of seeing you participate. They're getting their dose of social reduction, if you will.
It's also worth noting that the universe is full of these electron-snatching characters. Things like oxygen itself are notoriously good at this. That's why we breathe it! It's constantly taking electrons from things, which is why things rust, and why campfires burn so merrily. Oxygen is the undisputed champion of electron collection in our everyday lives. It’s the ultimate electron vacuum cleaner.
Then there are other heavy hitters, like halogens – think chlorine or bromine. They're practically salivating at the thought of snagging an electron. They’re the ultimate hoarders of the chemical world, always looking for that next shiny electron to add to their collection. They’re like that person who has to have all the best collectibles, leaving everyone else to sort through the leftovers.

And let's not forget peroxides. They're like the overachievers of the chemical world. They have an extra oxygen atom just waiting to cause some electron-related chaos. They're the ones you call when you need a serious oxidation done. They're not subtle, they're not shy, they just get straight to the electron-taking business.
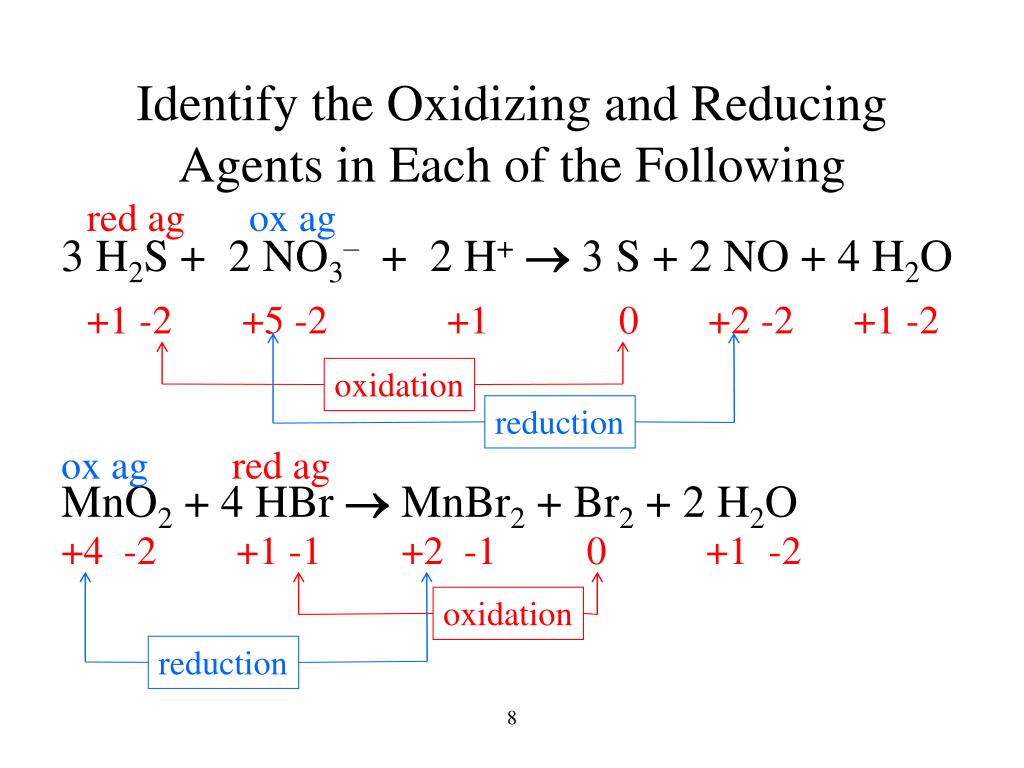
The thing is, identifying the Oxidizing Agent is like playing detective. You have to look for who is gaining electrons, or in other words, who is getting reduced. It's the substance that's doing the oxidizing. It's the electron thief. It’s the catalyst of change. It's the silent (or sometimes not-so-silent) force that drives a whole bunch of chemical reactions forward.
So, the next time you hear the term "Oxidizing Agent," don't picture some scary, lab-coat-wearing villain. Picture a busybody, a collector, a catalyst for change. Picture that friend who pulls you out of your comfort zone. Picture the very air you breathe, busy at work. They might be taking something, but in the grand scheme of things, they're making the world go 'round, one electron at a time. And honestly, I think that's pretty darn cool. It’s the unsung hero of so many chemical transformations, and frankly, it deserves a little more fan club.
