What Is The Oxidation State Of Each Element In Febr3
Hey there, curious minds! Ever looked at a chemical formula and thought, "Whoa, what's going on in there?" Today, we're going to peek behind the curtain of a super cool compound called FeBr3. It might sound a bit science-y, but trust me, it's got some hidden charm that makes figuring out its secrets a real blast.
So, what's the deal with FeBr3? It's basically iron and bromine hanging out together. We call this an ionic compound. Think of it like a little chemical love story where one element gives away some of its tiny building blocks, called electrons, and another happily snatches them up. This giving and taking of electrons is what makes them stick together so tightly.
Now, to understand how they're behaving in this little partnership, we talk about their oxidation states. Imagine each element has a "score" that tells us how many electrons it's either lost or gained. It’s like a game, and everyone wants to reach a stable, happy state. And let me tell you, the oxidation states in FeBr3 are quite the fascinating characters!
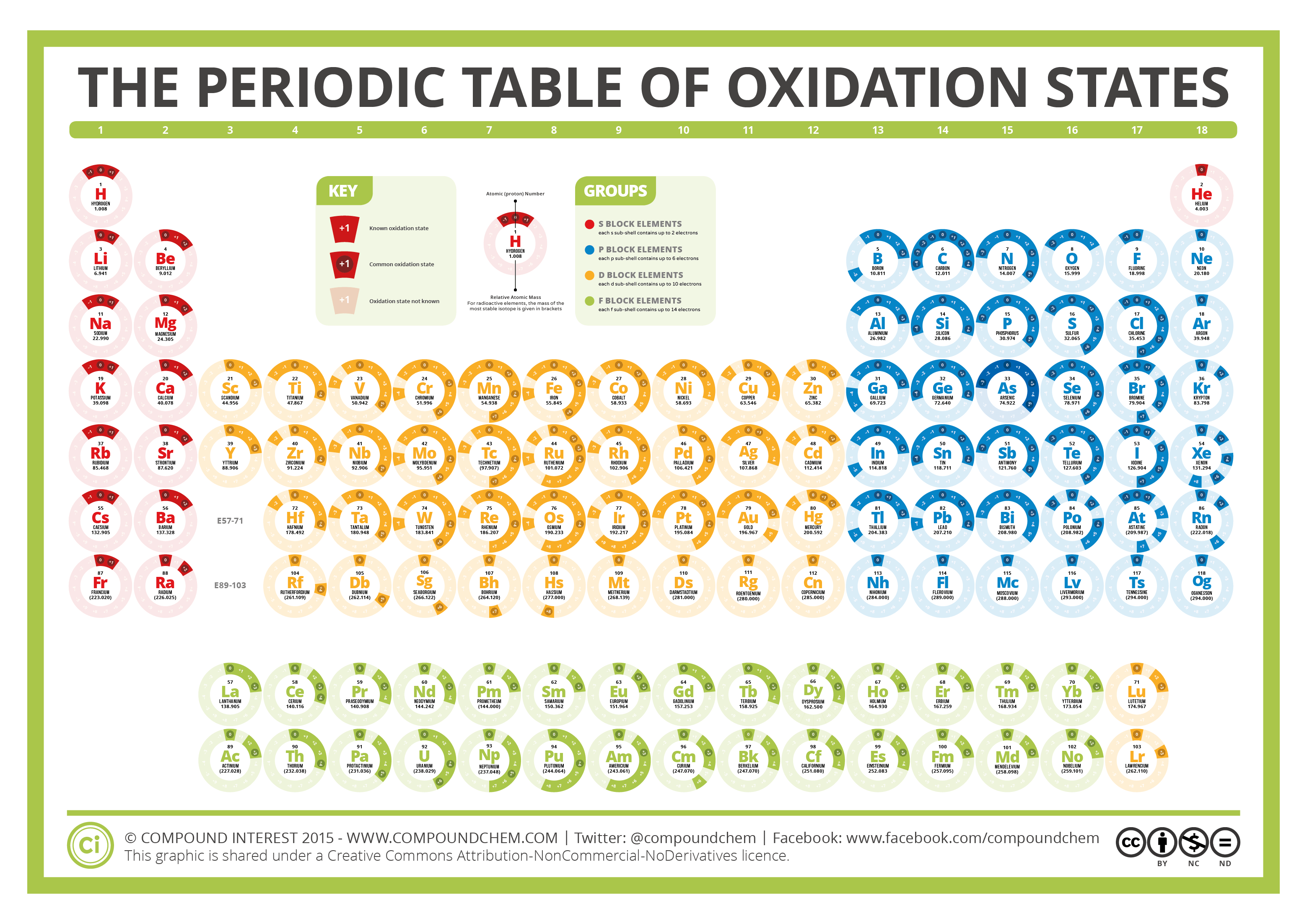
First up, let's meet Iron, or Fe. In FeBr3, our friend Iron is playing the generous giver. It's handed over three of its precious electrons. This means Iron's oxidation state here is a big, bold +3. Can you picture it? Iron strutting around, proud of its generosity, leaving behind a positive vibe. It’s like the star of the show, having made a significant contribution to the compound's stability.
Now, who's receiving those electrons? That would be our pals, the Bromine atoms, or Br. And guess what? We have three of them in this compound! Each Bromine atom is a bit of an electron-lover. They're eager to grab onto those electrons that Iron is so happily offering. So, each Bromine atom in FeBr3 has accepted one electron.

Because each Bromine atom has gained one electron, their oxidation state is -1. See how it's the opposite of Iron? That's the beauty of this electron exchange! You've got Iron being a +3 boss, and then you have three Bromine atoms, each being a -1 buddy, collectively balancing out that big positive charge. It's a perfectly orchestrated dance of positive and negative, making the whole compound electrically neutral. Pretty neat, right?
So, to sum up our little adventure into FeBr3: Iron (Fe) has an oxidation state of +3, and each Bromine (Br) atom has an oxidation state of -1. It's like Iron is the ultimate philanthropist, and Bromine is the happy recipient, and together they create something new and stable.

Why is this so entertaining? Well, it's like solving a tiny puzzle! You see the formula, and then you use your knowledge of how elements like to behave to figure out their individual roles. It's like being a detective in the microscopic world. And when you crack the code and understand the oxidation states, you unlock a deeper appreciation for how these seemingly simple formulas are actually intricate arrangements of atoms working together.
What makes FeBr3 special? It's a classic example, a go-to for understanding these concepts. It’s like the friendly face of chemistry that invites you to explore further. It’s not some super rare or complicated molecule; it’s accessible, and its oxidation states are clear and easy to follow once you get the hang of it.
Imagine Iron as the generous friend who's always treating everyone to coffee, and each Bromine atom is the friend who gratefully accepts, making sure the total "bill" is always zero. That's the vibe of FeBr3!
Redox Recap. - ppt download
The beauty lies in the balance. The positive charge from the iron perfectly cancels out the negative charge from the three bromine atoms. This balance is crucial for the compound to exist. Without it, things would be a bit chaotic, and the compound wouldn't be as stable.
Thinking about oxidation states is more than just assigning numbers. It tells us a story about the chemical personalities of the elements. Iron, in this case, is showing its more "cationic" side – its tendency to lose electrons and become positively charged. Bromine, on the other hand, is showcasing its "anionic" nature – its desire to gain electrons and become negatively charged.

It’s this interaction, this transfer of tiny electron particles, that forms the very bonds holding FeBr3 together. It’s a fundamental concept in chemistry, and FeBr3 offers a straightforward window into it. You don't need a fancy lab coat or years of study to start appreciating this. Just a little bit of curiosity!
So, the next time you see FeBr3, don't just see letters and numbers. See the dynamic interplay of elements, the generous Iron with its +3 swagger, and the three receptive Bromines, each with a -1 glow. It’s a little chemical drama playing out, and it’s incredibly cool to understand your part in it.
This kind of understanding opens doors to so many other fascinating chemical phenomena. It’s like learning the alphabet before you can read a novel. And the world of chemistry is a truly vast and engaging novel, just waiting for you to dive in. Give FeBr3 a second look – it might just be the spark that ignites your interest in the amazing world of chemistry!

