What Is The Oxidation State Of Each Element In Febr2

Imagine you've got a little team of characters, each with their own special job. They're all hanging out together, forming a compound, and just like in any team, they have different roles. Today, we're going to peek behind the scenes of a compound called FeBr₂, which is basically iron hanging out with two bromines.
Think of this compound like a tiny drama where each element plays a part. Some are a bit more "giving," happily sharing their electrons, while others are more "taking," eager to snag them. It's all about balance, and understanding these roles helps us see how the whole team works together.
The Star of the Show: Iron (Fe)
Our main guy, the one in the spotlight, is Iron, or Fe for short. In the world of chemistry, iron is known for being a bit of a chameleon. It can change its "outfit" depending on who it's hanging out with.
In our FeBr₂ story, iron is playing a very specific role. It's the generous one, the one who's decided to share some of its "stuff" to make everyone else happy. This sharing is what gives iron a particular "score" or "oxidation state."
When iron is with bromine, it's usually feeling quite giving. It's like the friendly neighbor who always has a spare cup of sugar. This generosity allows the other members of the team to feel more stable.
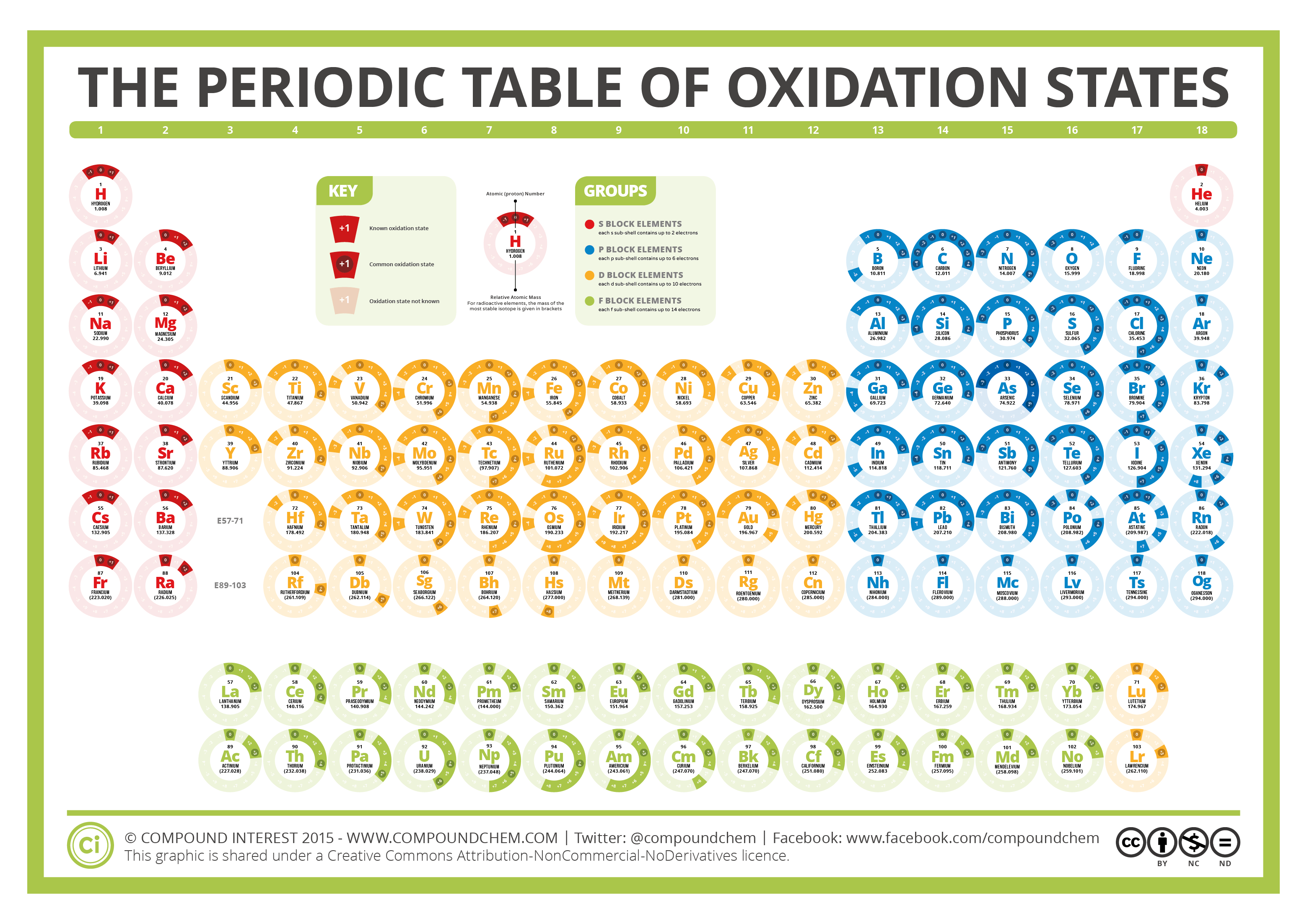
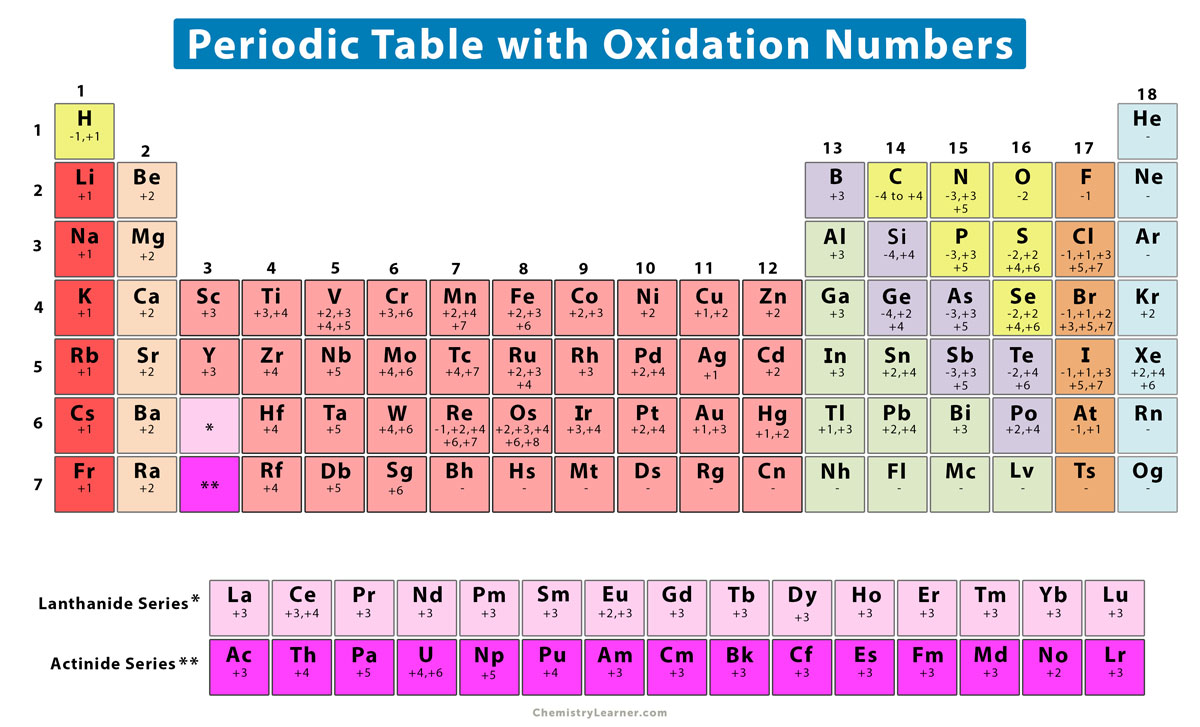
So, for Iron (Fe) in FeBr₂, its oxidation state is a nice, round +2. This tells us that iron has decided to let go of two of its little electron "treasures" to its teammates. It’s a sign of its willingness to cooperate and form a strong bond.
The Supportive Duo: Bromine (Br)
Now, let's meet the two Bromine atoms, or Br. These guys are like the eager helpers, always looking for a chance to complete their own electron "sets." They’re a bit more like the collectors, happily accepting any spare electrons offered.
In this team, bromine is playing the role of the electron acceptor. It’s always on the lookout to make itself feel complete and stable. This is a common trait for many elements that form compounds.

Since each bromine atom is so keen to grab an electron, they usually come with a price tag of -1 for each one. Think of it as a "thank you" fee for the electrons they receive. This makes them feel nice and complete.
So, we have two Bromine (Br) atoms, and each one has an oxidation state of -1. That means together, they’re looking to get a total of -2 worth of electrons. It's like a package deal, with each bromine contributing to the overall "electron debt."
The Team's Balancing Act
Now, let's put it all together. We have our generous Iron (Fe) with its +2 score, and our two electron-loving Bromine (Br) atoms, each with a -1 score. When you add up all these scores, something magical happens.
The total score for our FeBr₂ team is zero! That's right, the positive "giving" from iron perfectly balances out the negative "taking" from the two bromines. This is the secret to how they stay together, like a perfectly matched pair.
This balancing act is what makes compounds stable. It's like a seesaw that’s perfectly level because the weights on both sides are equal. Without this balance, they wouldn’t be able to form such a cozy little group.

It's a heartwarming tale of cooperation! The iron, feeling a bit like it has too much, finds two bromines who are just the right size to help it feel balanced. And the bromines, always wanting a bit more, are thrilled to receive what iron is happily offering.
So, in FeBr₂, we see Iron (Fe) rocking an oxidation state of +2, and each of the two Bromine (Br) atoms is at -1. It's a simple equation, but it unlocks a whole world of how these elements interact and form the materials we see around us every day.
Next time you hear about iron and bromine, you can imagine this little team, working together in perfect harmony. It's not just numbers; it's a story of elements finding their perfect balance and creating something new.
It’s a reminder that even in the smallest particles, there’s a whole lot of chemistry happening, a constant dance of giving and taking, all in pursuit of stability and togetherness. And it’s pretty fascinating when you think about it!
So, the next time you encounter FeBr₂, whether it's in a lab or a textbook, remember the story of the generous iron and its two grateful bromine friends. They’re a perfect example of how elements form bonds, and how their "scores" tell a tale of their interactions.
The world of chemistry is full of these little dramas, and understanding the oxidation states is like getting a backstage pass to see how the actors interact. It's a fun way to see the invisible forces at play!
And remember, even though we use numbers like +2 and -1, it’s all about a shared desire for stability. The elements are just trying to find their happy place, and sometimes that means sharing or taking a little bit.
This simple compound, FeBr₂, is a microcosm of a much larger universe of chemical interactions. It's a testament to the fundamental principles that govern how matter behaves, all explained through the concept of oxidation states.
So, let the numbers be a guide, but let the story of cooperation and balance be what truly captivates you. The chemistry behind FeBr₂ is as much about teamwork as it is about electron counts.
It’s a beautiful dance of charge, where the positive and negative charges meet in the middle, creating a stable and happy compound. Each element plays its part, contributing to the overall stability of the team.
And that, in a nutshell, is the story of the oxidation states in FeBr₂. A tale of giving, taking, and finding perfect equilibrium, all within a tiny, invisible world.
It’s a reminder that even the most complex compounds have roots in simple, fundamental interactions. The journey of an electron can lead to a stable bond, and that’s pretty remarkable.
So, we can confidently say that Iron is playing the generous role with a +2, and each of the two Bromines is happily accepting with a -1. The team is balanced, and the compound is stable.
The next time you see FeBr₂, you’ll know the secret behind its stability: a perfect partnership, orchestrated by the principles of oxidation states. It’s a story that repeats itself across the periodic table, in countless compounds.
And that, dear reader, is the charming, albeit numerical, story of how Iron and Bromine come together to form FeBr₂. A simple compound with a complex, yet elegant, internal dynamic.
So, let the numbers speak, but also let them remind you of the cooperative spirit of chemistry. It's a world where elements find their perfect match, and stability reigns supreme.
